Advanced Chemo-Enzymatic Synthesis of Tenofovir for Commercial Scale-Up
Introduction to Next-Generation Tenofovir Manufacturing
The global demand for effective antiretroviral therapies continues to drive the need for efficient, scalable, and cost-effective manufacturing processes for key pharmaceutical intermediates. Patent CN102899367B introduces a groundbreaking hybrid methodology that merges biological fermentation with traditional organic synthesis to produce Tenofovir (PMPA), a critical active pharmaceutical ingredient (API) intermediate used in the treatment of HIV and Hepatitis B. This technical breakthrough addresses long-standing challenges in chirality control and process economy by leveraging the stereoselective power of Saccharomyces cerevisiae yeast to generate the crucial chiral building block. By integrating this biocatalytic step with a streamlined chemical sequence involving phosphonation and nucleophilic substitution, the technology offers a robust alternative to purely synthetic routes, promising enhanced purity profiles and reduced environmental impact for reliable pharmaceutical intermediates supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tenofovir has been plagued by significant technical hurdles that impact both cost and quality. Early literature, such as the work by Holy et al., describes synthetic pathways that are excessively long and operationally cumbersome, involving multiple protection and deprotection steps that drastically lower the overall yield. Alternatively, other established methods, like those cited in US5935946A, attempt to shorten the route but often fail to deliver the requisite optical purity, typically achieving only 90-94% enantiomeric excess. This level of purity is insufficient for clinical applications, necessitating additional, costly purification steps such as chiral chromatography or recrystallization, which further erode profit margins and complicate the supply chain. These conventional chemical-only approaches often rely on harsh reagents and difficult-to-control reaction conditions, making them less ideal for modern green chemistry standards.
The Novel Approach
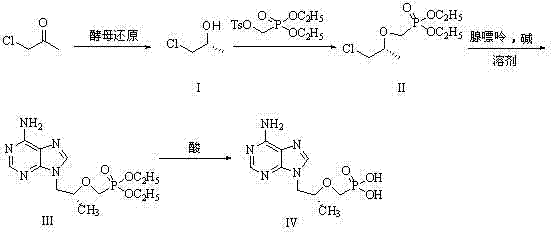
The patented innovation fundamentally reimagines the synthetic strategy by introducing a biocatalytic entry point. Instead of relying on asymmetric chemical catalysis or resolution of racemates, the process begins with the fermentation of chloroacetone using dry yeast. This biological transformation efficiently produces chiral chloropropanol with high stereoselectivity under mild conditions (approximately 35°C). Following this, the chiral alcohol undergoes a clever condensation reaction with diethyl p-toluenesulfonyloxymethylphosphonate. This specific reagent choice is pivotal as it simultaneously installs the phosphonate moiety and protects the hydroxyl group, effectively merging two synthetic steps into one. The resulting intermediate is then coupled with adenine and subsequently hydrolyzed to yield the final Tenofovir product. This hybrid route not only shortens the reaction sequence but also ensures superior product quality from the outset.
Mechanistic Insights into Chemo-Enzymatic Cascade Synthesis
The core of this technological advancement lies in the precise orchestration of enzymatic and chemical mechanisms. In the initial biocatalytic phase, the yeast cells act as a source of carbonyl reductases, enzymes that facilitate the hydride transfer from NADPH to the carbonyl carbon of chloroacetone. The enzyme's active site imposes a strict steric environment, ensuring that the hydride attacks from a specific face of the planar carbonyl group, thereby generating the (R)-chloropropanol with high enantiomeric excess. This biological precision eliminates the formation of the unwanted (S)-enantiomer, which would otherwise act as a difficult-to-remove impurity later in the synthesis. The mild aqueous environment of the fermentation also prevents side reactions such as elimination or polymerization that might occur under harsher chemical reduction conditions.
Following the isolation of the chiral alcohol, the chemical mechanism shifts to nucleophilic substitution. The hydroxyl group of the chloropropanol is deprotonated by a strong base, such as sodium hydride or sodium hydroxide, generating a reactive alkoxide species. This alkoxide then attacks the methylene carbon adjacent to the sulfonate leaving group in the diethyl p-toluenesulfonyloxymethylphosphonate reagent via an SN2 mechanism. This step is critical as it forms the stable ether linkage connecting the chiral backbone to the phosphonate group. Subsequent coupling with adenine involves another nucleophilic attack, where the nitrogen of the purine ring displaces the chloride on the side chain. The final hydrolysis step cleaves the ethyl ester groups of the phosphonate using hydrobromic acid, revealing the free phosphonic acid functionality essential for the antiviral activity of Tenofovir, while the careful control of pH during workup ensures the precipitation of the pure product.
How to Synthesize Tenofovir Efficiently
The implementation of this hybrid synthesis requires careful attention to the interface between the biological and chemical domains. The process begins with the activation of dry yeast in a buffered solution, followed by the controlled addition of chloroacetone to maintain cell viability and enzymatic activity. Once the chiral alcohol is secured, the transition to anhydrous organic conditions is necessary for the phosphonation step. Detailed operational parameters, including specific molar ratios of base to substrate and precise temperature controls during the adenine coupling, are critical for maximizing yield and minimizing byproduct formation. For a comprehensive understanding of the exact experimental conditions and stoichiometry required to replicate this high-efficiency pathway, please refer to the standardized technical guide below.
- Perform asymmetric reduction of chloroacetone using Saccharomyces cerevisiae yeast fermentation to obtain chiral chloropropanol.
- Condense the chiral alcohol with diethyl p-toluenesulfonyloxymethylphosphonate under basic conditions to form the phosphonate intermediate.
- Couple the phosphonate intermediate with adenine followed by acid hydrolysis to yield the final Tenofovir product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chemo-enzymatic route presents a compelling value proposition centered on cost efficiency and supply security. By substituting expensive chiral catalysts and cryogenic reagents with commodity-grade yeast and ambient temperature fermentation, the raw material cost profile is significantly optimized. The reduction in the total number of synthetic steps directly translates to lower consumption of solvents, energy, and labor, driving down the overall cost of goods sold (COGS). Furthermore, the reliance on widely available starting materials like chloroacetone and adenine mitigates the risk of supply disruptions associated with specialized fine chemicals, ensuring a more resilient and continuous supply chain for cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The elimination of complex chiral resolution steps and the telescoping of protection-phosphorylation reactions remove significant cost centers from the production budget. Traditional methods often require expensive chiral auxiliaries or precious metal catalysts, along with the associated costs of removing trace metals to meet regulatory limits. This bio-based approach bypasses these requirements entirely. Additionally, the high yield and purity achieved in the initial fermentation step reduce the load on downstream purification units, leading to substantial savings in chromatography media and solvent recovery operations.
- Enhanced Supply Chain Reliability: The robustness of the yeast fermentation process allows for flexible production scheduling and easier scale-up compared to sensitive organometallic reactions. Since the key chiral intermediate is generated biologically, manufacturers are less dependent on volatile markets for chiral building blocks. The use of standard chemical reagents in the subsequent steps ensures that sourcing remains straightforward and competitive. This stability is crucial for maintaining consistent lead times and meeting the rigorous delivery schedules demanded by global pharmaceutical clients seeking high-purity Tenofovir.
- Scalability and Environmental Compliance: The process operates under mild conditions, avoiding the safety hazards and energy intensity associated with high-pressure hydrogenation or ultra-low temperature reactions. This makes the technology inherently safer and easier to scale from pilot plant to commercial tonnage. From an environmental perspective, the use of biocatalysis aligns with green chemistry principles by reducing waste generation and energy consumption. The simplified workup procedures minimize the volume of hazardous waste streams, facilitating easier compliance with increasingly stringent environmental regulations and supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is essential for R&D teams evaluating process feasibility and procurement officers assessing vendor capabilities. The answers provided are derived directly from the technical specifications and advantageous effects described in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of purity, cost, and operational simplicity for the commercial scale-up of complex nucleotide analogs.
Q: How does the chemo-enzymatic route improve optical purity compared to traditional chemical synthesis?
A: Traditional chemical routes often struggle to achieve high enantiomeric excess without expensive chiral catalysts or resolution steps. The patented method utilizes specific yeast strains (Saccharomyces cerevisiae) which naturally possess ketoreductase enzymes capable of highly stereoselective reduction of chloroacetone. This biological step inherently produces the desired chiral chloropropanol with superior optical purity, eliminating the need for downstream chiral separation and ensuring the final Tenofovir meets stringent clinical specifications.
Q: What are the primary cost drivers reduced in this hybrid manufacturing process?
A: The process significantly lowers costs by replacing expensive chiral chemical reagents and cryogenic conditions with inexpensive, readily available dry yeast and mild fermentation conditions (around 35°C). Furthermore, the strategy of using diethyl p-toluenesulfonyloxymethylphosphonate serves a dual purpose: it introduces the phosphonate group while simultaneously protecting the hydroxyl functionality. This telescoping of protection and phosphorylation steps reduces the total number of unit operations, solvent consumption, and labor hours required for the overall synthesis.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the methodology is explicitly designed for industrial scalability. The fermentation step avoids extreme temperatures or pressures, utilizing standard bioreactor equipment. The subsequent chemical steps employ common solvents like THF and DMF and standard bases such as sodium hydride or cesium carbonate. The robustness of the yeast reduction and the simplified workup procedures, such as direct filtration and distillation without complex chromatography in early stages, make the transfer from laboratory bench to multi-ton commercial production highly feasible and reliable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of integrating biocatalysis into small molecule synthesis to create superior pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Tenofovir adheres to the highest international standards required for antiviral drug formulation.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized processes can enhance your project's economic and technical success while reducing lead time for high-purity antiviral intermediates.
