Optimizing Testosterone Enanthate Production: A Technical Breakthrough in Hormone Synthesis
The pharmaceutical landscape for hormone replacement therapies relies heavily on the efficient and pure synthesis of long-acting esters like Testosterone Enanthate. A pivotal advancement in this domain is detailed in patent CN102558267A, which outlines a robust preparation method that fundamentally alters the traditional synthetic pathway. This technology addresses long-standing inefficiencies in the production of this critical API intermediate, specifically targeting the issues of by-product formation, expensive reagent usage, and environmental pollution. By introducing a strategic etherification protection step prior to reduction, the process effectively eliminates the formation of difficult-to-remove diol impurities that have historically plagued manufacturers. Furthermore, the substitution of heptanoic anhydride with heptanoyl chloride in the final esterification stage represents a significant optimization in both cost structure and waste management. For global procurement and R&D teams, understanding this mechanistic shift is essential for securing a reliable testosterone enanthate supplier capable of delivering high-purity materials at a competitive operational cost.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Testosterone Enanthate from 4-Androstenedione (4-AD) has been fraught with selectivity challenges and economic inefficiencies. The conventional route typically involves a direct reduction of 4-AD to obtain a mixture of testosterone and a glycol by-product, followed by an oxidation step to recover testosterone, and finally esterification. As illustrated in the reaction scheme below, this lack of chemoselectivity during the initial reduction phase inevitably leads to the co-production of 3-beta, 17-beta-dihydroxy-androst-5-ene (the diol). This by-product is structurally similar to the target molecule, making its removal via crystallization or chromatography extremely difficult and yield-destructive. Additionally, the traditional esterification process relies on heptanoic anhydride, a reagent that is not only costly due to import dependencies but also generates substantial quantities of heptanoic acid as a waste by-product. This results in a complex wastewater treatment burden and limits the overall process yield to approximately 90.0%, creating a bottleneck for large-scale commercial manufacturing.
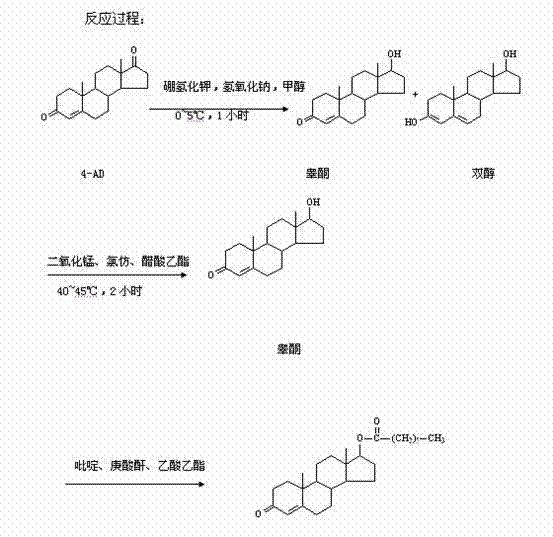
The Novel Approach
In stark contrast to the legacy methods, the novel approach described in the patent introduces a sophisticated protection-deprotection strategy that dramatically enhances process control and product quality. The core innovation lies in the initial etherification of the 4-AD starting material. By reacting 4-AD with absolute ethanol and triethyl orthoformate in the presence of a pyridinium tribromide hydrobromide catalyst, the C3 carbonyl group is temporarily protected as an enol ether. This structural modification effectively masks the C3 position, forcing the subsequent reduction reaction to occur exclusively at the C17 carbonyl group. Following the reduction, a mild acidic hydrolysis step removes the ether protecting group, yielding testosterone with exceptional purity and negligible diol contamination. The final esterification utilizes heptanoyl chloride instead of the anhydride, a change that accelerates reaction kinetics and simplifies the workup procedure. This comprehensive redesign of the synthetic pathway ensures a streamlined operation with significantly improved yields and a much cleaner impurity profile.
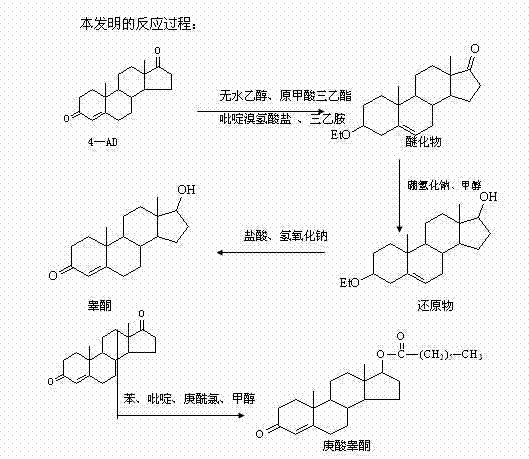
Mechanistic Insights into Etherification-Protection and Selective Reduction
The success of this synthesis hinges on the precise manipulation of electronic environments within the steroid backbone during the etherification and reduction phases. The use of pyridinium tribromide hydrobromide as a catalyst in the etherification step is particularly noteworthy; it facilitates the formation of the 3-ethoxy-3,5-androstadien-17-one intermediate under mild thermal conditions (42-45°C). This intermediate serves as a crucial shield, preventing the nucleophilic attack of the hydride reducing agent at the C3 position. When sodium borohydride or potassium borohydride is subsequently introduced in methanol at low temperatures (0-5°C), the hydride ion selectively targets the exposed C17 ketone. This chemoselectivity is the key to avoiding the diol by-product entirely. The subsequent hydrolysis step, conducted by adjusting the pH to approximately 2.0 with hydrochloric acid, cleaves the enol ether bond, regenerating the conjugated enone system characteristic of testosterone while maintaining the newly formed 17-beta-hydroxyl group intact. This mechanism ensures that the stereochemistry at the C17 position is preserved, which is vital for the biological activity of the final hormone.
Furthermore, the transition to heptanoyl chloride for the final acylation step offers distinct mechanistic advantages over the anhydride route. Acid chlorides are inherently more electrophilic than acid anhydrides, allowing the esterification to proceed rapidly at lower temperatures (5-10°C) in the presence of pyridine as a base. This milder condition minimizes the risk of epimerization or degradation of the sensitive steroid nucleus. The by-product of this reaction is hydrogen chloride, which is neutralized by the pyridine to form pyridine hydrochloride, a solid salt that is easily removed during the aqueous workup. In contrast, the anhydride route produces heptanoic acid, a liquid organic acid that often requires extensive washing or distillation to remove completely from the lipophilic steroid product. By eliminating this persistent organic impurity at the source, the new method drastically simplifies the purification protocol, leading to a final product that consistently meets rigorous pharmacopoeial specifications for free acid content.
How to Synthesize Testosterone Enanthate Efficiently
The implementation of this patented methodology requires strict adherence to the specified stoichiometric ratios and thermal profiles to maximize the benefits of the protection strategy. The process begins with the dissolution of 4-AD in absolute ethanol and triethyl orthoformate, followed by the catalytic addition of pyridinium tribromide hydrobromide. Maintaining the reaction temperature between 42°C and 45°C for approximately 8 hours is critical to ensure complete conversion to the etherate intermediate without degrading the steroid skeleton. Once the etherification is complete, the mixture is cooled to near-freezing conditions before the controlled addition of the borohydride reducing agent. This exothermic reduction must be managed carefully to prevent runaway reactions that could compromise selectivity. Following the reduction, the pH is meticulously adjusted to facilitate hydrolysis and product precipitation. The final esterification step demands anhydrous conditions and precise temperature control to ensure the complete conversion of testosterone to the enanthate ester while minimizing the formation of free acid impurities. Detailed standardized operating procedures for these steps are essential for reproducibility.
- Perform etherification of 4-Androstenedione (4-AD) using absolute ethanol, triethyl orthoformate, and pyridinium tribromide hydrobromide catalyst at 42-45°C to protect the C3 carbonyl.
- Conduct reductive hydrolysis using sodium or potassium borohydride in methanol, followed by acid hydrolysis to remove the ether protecting group and yield high-purity Testosterone.
- Execute the final esterification reaction by reacting Testosterone with heptanoyl chloride in benzene and pyridine at 5-10°C to produce Testosterone Enanthate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates into tangible operational improvements and risk mitigation strategies. The primary economic driver is the substitution of heptanoic anhydride with heptanoyl chloride. Since heptanoic anhydride is often an imported specialty chemical with a volatile price point, replacing it with the more commodity-grade heptanoyl chloride stabilizes the raw material cost structure. Moreover, the higher selectivity of the new process means that less starting material is wasted on by-products that must be discarded or recycled. This improvement in atom economy directly correlates to a reduction in the cost of goods sold (COGS). Additionally, the simplified purification process reduces the consumption of solvents and energy associated with multiple recrystallizations or chromatographic separations, further driving down manufacturing expenses. These factors combined create a more resilient supply chain that is less susceptible to raw material price fluctuations and processing bottlenecks.
- Cost Reduction in Manufacturing: The elimination of expensive heptanoic anhydride in favor of heptanoyl chloride provides a direct reduction in reagent costs, while the high-yield protection strategy minimizes the loss of valuable 4-AD starting material. By avoiding the formation of the diol by-product, the need for costly oxidative recycling steps is completely removed, streamlining the production flow. The reduction in solvent usage for purification also contributes to lower utility and waste disposal costs, resulting in a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Relying on domestically available or widely sourced reagents like heptanoyl chloride and common borohydrides reduces dependency on single-source imported anhydrides. The robustness of the etherification protection step ensures consistent batch-to-batch quality, reducing the risk of production failures or off-spec material that could disrupt delivery schedules. This reliability is crucial for maintaining continuous supply to downstream formulation partners who depend on timely API deliveries for their own production planning.
- Scalability and Environmental Compliance: The process generates significantly less organic waste, particularly heptanoic acid, which simplifies wastewater treatment and reduces the environmental footprint of the facility. The mild reaction conditions and straightforward workup procedures make the process highly scalable from pilot plant to multi-ton commercial production without requiring specialized high-pressure or cryogenic equipment. This ease of scale-up allows manufacturers to respond quickly to market demand surges while maintaining compliance with increasingly stringent environmental regulations regarding organic effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these details is vital for technical teams assessing the transferability of this process to their existing infrastructure.
Q: Why is the etherification step critical in this synthesis route?
A: The etherification step protects the C3 carbonyl group of 4-Androstenedione. Without this protection, direct reduction leads to the formation of a diol by-product (3-beta, 17-beta-dihydroxy-androst-5-ene), which is difficult to separate and lowers overall yield. This protection ensures selective reduction at the C17 position.
Q: What are the advantages of using heptanoyl chloride over heptanoic anhydride?
A: Using heptanoyl chloride instead of heptanoic anhydride significantly reduces raw material costs as the chloride is generally less expensive and more reactive. Furthermore, it minimizes the generation of free heptanoic acid waste, simplifying wastewater treatment and improving the purity profile of the final API.
Q: What purity levels can be achieved with this patented method?
A: The patented process consistently achieves Testosterone Enanthate purity levels exceeding 98.0%, with free acid content controlled below 0.16%. This meets stringent pharmacopoeial standards such as USP28 without requiring extensive downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Testosterone Enanthate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of steroid synthesis demands a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of temperature and stoichiometry required for this etherification-reduction sequence is maintained at every scale. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of Testosterone Enanthate meets the highest international standards, including USP and EP monographs. Our capability to implement the protection-group strategy described in CN102558267A allows us to offer a product with superior impurity profiles compared to conventional market offerings.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic advantages of switching to our high-efficiency manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless integration of our high-quality intermediates into your pharmaceutical development pipeline.
