Advanced Green Esterification Technology for Commercial Scale Production of Testosterone Derivatives
The pharmaceutical industry is constantly seeking robust, environmentally sustainable pathways for the synthesis of critical hormonal active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is documented in patent CN111848713A, which discloses a novel preparation method for alkyl acid testosterone derivatives. This technology addresses the longstanding inefficiencies associated with traditional steroid esterification by shifting from water-soluble solvent systems to water-insoluble organic media. By utilizing solvents such as dichloromethane or dichloroethane, the process fundamentally alters the post-reaction workup, enabling direct phase separation rather than the water-intensive precipitation methods of the past. This strategic modification not only aligns with modern green chemistry principles but also delivers a total molar yield higher than 85 percent, establishing a new benchmark for efficiency in hormone manufacturing. For global procurement and R&D teams, this patent represents a critical opportunity to optimize supply chains for high-value intermediates like testosterone propionate and enanthate.
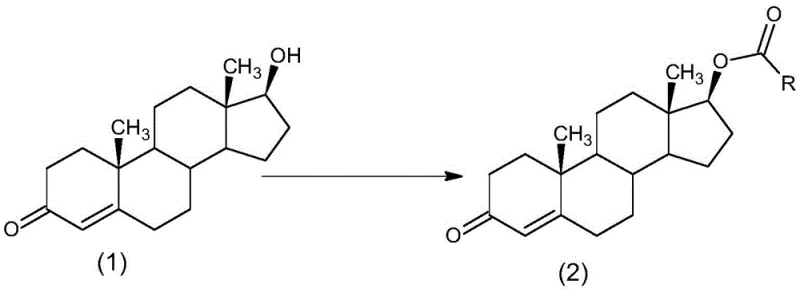
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of testosterone esters has been plagued by significant environmental and operational drawbacks inherent to older synthetic protocols. As highlighted in prior art such as patent CN105732754A, conventional methods frequently rely on water-soluble organic solvents to facilitate the reaction between steroidal alcohols and acyl chlorides. While chemically feasible, this approach necessitates the addition of substantial volumes of water post-reaction to precipitate the product, a step that generates massive quantities of acidic wastewater containing dissolved organic solvents. Furthermore, alternative methods described in patents like CN103910777A utilize chloroform but require complex post-treatment procedures involving acid scavengers such as alumina, calcium oxide, or sodium methoxide. These additional purification steps are not only operationally cumbersome and time-consuming but also introduce potential points of failure where residual acids or scavenger particulates might contaminate the final API, compromising quality control standards.
The Novel Approach
The methodology presented in CN111848713A offers a transformative solution by replacing problematic solvent systems with water-insoluble alternatives like dichloromethane or dichloroethane. This seemingly simple substitution creates a profound impact on the process workflow; upon completion of the esterification, the reaction mixture can be quenched with water and immediately separated into distinct organic and aqueous layers. This eliminates the need for energy-intensive distillation or filtration steps required to recover products from water-soluble matrices. Moreover, because the solvent remains in the organic phase, it can be easily recovered and recycled for subsequent batches, drastically reducing raw material consumption. The elimination of solid acid scavengers further streamlines the operation, removing the filtration bottlenecks that often limit throughput in batch reactors. This streamlined approach ensures that the production of reliable hormone intermediates is both economically superior and environmentally compliant.
Mechanistic Insights into DMAP-Catalyzed Esterification
The core chemical transformation driving this process is a nucleophilic acyl substitution, specifically an esterification reaction catalyzed by 4-dimethylaminopyridine (DMAP). In this mechanism, the steroid substrate, 17β-hydroxyandrost-4-en-3-one, acts as the nucleophile, attacking the electrophilic carbonyl carbon of the alkyl acyl chloride. The presence of DMAP is critical; it functions as a super-nucleophile that initially attacks the acyl chloride to form a highly reactive acyl-pyridinium intermediate. This intermediate is significantly more electrophilic than the original acyl chloride, thereby lowering the activation energy required for the sterically hindered 17-hydroxyl group of the testosterone scaffold to attack. This catalytic cycle allows the reaction to proceed rapidly and efficiently at mild temperatures ranging from 5°C to 40°C, preserving the integrity of the sensitive conjugated enone system within the steroid A-ring which might otherwise be susceptible to degradation under harsher thermal conditions.
Simultaneously, the inclusion of a stoichiometric organic base, such as pyridine, triethylamine, or diisopropylethylamine, serves a dual purpose in maintaining reaction fidelity. Primarily, the base acts as an acid scavenger, neutralizing the hydrogen chloride (HCl) byproduct generated during the formation of the ester bond. If left unchecked, accumulated HCl could catalyze unwanted side reactions, such as the dehydration of the tertiary alcohol or the hydrolysis of the newly formed ester. By effectively trapping the acid as an ammonium salt, the base drives the equilibrium toward product formation and ensures a clean impurity profile. The synergy between the nucleophilic catalyst (DMAP) and the stoichiometric base creates a robust chemical environment that consistently delivers purities exceeding 99 percent, meeting the rigorous specifications required for pharmaceutical grade hormones.
How to Synthesize Testosterone Esters Efficiently
The practical execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize impurities. The protocol begins by establishing an inert atmosphere using nitrogen, argon, or helium to prevent moisture ingress, which could hydrolyze the acyl chloride reagent. The substrate is dissolved in the water-insoluble solvent, followed by the sequential addition of the organic base and the DMAP catalyst. Once the mixture is homogenized and cooled to the target temperature between 5°C and 40°C, the alkyl acyl chloride is introduced. The reaction is monitored until completion, after which a controlled water quench initiates the phase separation. This standardized approach ensures reproducibility across different scales of operation, from laboratory optimization to multi-ton commercial campaigns.
- Under an inert atmosphere (nitrogen, argon, or helium), suspend 17β-hydroxyandrost-4-en-3-one in a water-insoluble organic solvent such as dichloromethane or dichloroethane.
- Introduce an organic base (pyridine, TEA, or DIPEA) and a catalytic amount of DMAP to the reaction mixture to facilitate nucleophilic activation.
- Add the corresponding alkyl acyl chloride while maintaining the temperature between 5°C and 40°C, then quench with water and separate layers to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates directly into tangible operational efficiencies and risk mitigation. The shift away from water-soluble solvents and solid acid scavengers removes several cost centers from the manufacturing budget. By eliminating the need for expensive filtration aids and the disposal of hazardous acidic sludge, the overall cost of goods sold (COGS) is substantially reduced. Furthermore, the ability to recycle the organic solvent creates a closed-loop system that insulates the production process from volatility in solvent pricing. This economic resilience is crucial for maintaining stable pricing structures for long-term supply contracts of critical hormone intermediates.
- Cost Reduction in Manufacturing: The elimination of water-soluble solvents removes the necessity for large-scale wastewater treatment infrastructure dedicated to recovering organics from aqueous streams. Additionally, the removal of solid acid scavengers like alumina or calcium oxide cuts out the cost of these reagents entirely, along with the labor and equipment time associated with their filtration and disposal. This streamlined material flow results in significant cost savings per kilogram of finished product, enhancing the margin profile for manufacturers of testosterone derivatives.
- Enhanced Supply Chain Reliability: Simplified workup procedures inherently reduce the cycle time for each batch. Without the need for prolonged filtration of scavengers or complex precipitation steps, the turnaround time between batches is drastically shortened. This increased throughput capacity allows suppliers to respond more agilely to market demand spikes, ensuring continuity of supply for downstream formulators who rely on consistent availability of testosterone propionate and enanthate for their own production schedules.
- Scalability and Environmental Compliance: The process is explicitly designed with industrial scale-up in mind, evidenced by the high molar yields reported in the patent data. The reduction in wastewater volume simplifies regulatory compliance, as facilities face less scrutiny regarding effluent discharge limits. This environmental advantage future-proofs the supply chain against tightening global environmental regulations, ensuring that production can continue uninterrupted without the risk of shutdowns due to non-compliance with waste management protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this green synthesis route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios. These insights provide a foundational understanding of why this method is becoming the preferred standard for high-quality steroid ester production.
Q: How does the new solvent system improve environmental compliance compared to traditional methods?
A: Traditional methods often utilize water-soluble solvents that require massive amounts of water for product precipitation, generating large volumes of acidic wastewater. The patented method employs water-insoluble solvents like dichloromethane, allowing for direct phase separation upon quenching. This drastically reduces wastewater generation and enables solvent recycling, significantly lowering the environmental footprint.
Q: What represents the key advantage of using DMAP in this esterification process?
A: 4-Dimethylaminopyridine (DMAP) acts as a potent nucleophilic catalyst that accelerates the acylation reaction under mild conditions. Its presence allows the reaction to proceed efficiently at temperatures ranging from 5°C to 40°C, ensuring high conversion rates without the need for harsh thermal conditions that could degrade the sensitive steroid backbone.
Q: Can this process be scaled for industrial production of testosterone undecanoate?
A: Yes, the process is explicitly designed for industrial scalability. The patent reports total molar yields exceeding 85% and purities above 99% for various esters including testosterone undecanoate. The simplified workup procedure, which eliminates cumbersome acid scavenging steps found in prior art, makes it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Testosterone Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is vital for the long-term sustainability of the pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111848713A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of testosterone esters meets the highest international pharmacopeial standards. Our commitment to technical excellence allows us to deliver high-purity hormone intermediates that support the development of safe and effective therapeutic products globally.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the partner of choice for complex steroid synthesis.
