Optimizing Levetiracetam Synthesis: Advanced Resolution of S-2-Aminobutanamide Hydrochloride
Optimizing Levetiracetam Synthesis: Advanced Resolution of S-2-Aminobutanamide Hydrochloride
The pharmaceutical industry's relentless pursuit of cost-effective and sustainable manufacturing processes has brought significant attention to the synthesis of chiral intermediates for antiepileptic drugs. Patent CN109503408B introduces a groundbreaking resolution method for (S)-(+)-2-aminobutanamide hydrochloride, a critical precursor in the production of Levetiracetam. This technology addresses the longstanding inefficiencies of traditional chiral splitting by integrating a novel racemization strategy directly into the resolution workflow. By leveraging the unused enantiomer found in resolution mother liquors, this approach transforms a typically wasteful 50% yield process into a highly efficient, near-quantitative recovery system. For global supply chain leaders and R&D directors, this represents a pivotal shift towards greener chemistry and reduced raw material dependency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Levetiracetam intermediates has been plagued by economic and technical bottlenecks inherent to classical resolution techniques. Traditional routes often rely on expensive chiral pool starting materials like L-2-aminobutyric acid, which drives up the cost of goods significantly due to the premium pricing of natural amino acids. Alternatively, synthetic routes starting from achiral materials like n-propionaldehyde require a resolution step that theoretically discards half of the produced material. In standard diastereomeric salt formation, the unwanted (R)-enantiomer is typically isolated as waste or requires complex, multi-step recycling procedures that degrade overall efficiency. Furthermore, prior art methods, such as those utilizing acetic acid as a solvent, introduce severe downstream processing challenges, including difficult solvent recovery and high energy consumption for distillation, making them less attractive for large-scale commercial production.
The Novel Approach
The methodology disclosed in CN109503408B fundamentally reengineers the resolution landscape by turning waste into value. Instead of discarding the mother liquor containing the unwanted (R)-enantiomer, this process subjects it to an in-situ racemization using catalytic amounts of aromatic or aliphatic aldehydes. This clever integration allows the L-tartaric acid already present in the mother liquor to be reused indefinitely for subsequent resolution cycles. The result is a dramatic increase in the total recovery rate of the desired (S)-enantiomer without the need for additional resolving agents. Moreover, the substitution of harsh organic solvents with a benign water and alcohol system not only lowers operational costs but also aligns with modern environmental, health, and safety (EHS) standards, facilitating easier regulatory approval and plant operation.
Mechanistic Insights into Aldehyde-Catalyzed Racemization and Resolution
At the heart of this technological advancement lies a sophisticated understanding of stereochemical equilibrium and salt stoichiometry. The process exploits the specific interaction between 2-aminobutanamide and L-tartaric acid, where the optimal molar ratio is maintained at 2:1. In this configuration, a single molecule of tartaric acid effectively complexes with two molecules of the amine, forming a stable diastereomeric salt that precipitates out of the solution. The key innovation, however, is the management of the supernatant. By introducing a weak base and a catalytic aldehyde—such as salicylaldehyde or butyraldehyde—the uncrystallized (R)-enantiomer in the mother liquor undergoes rapid racemization. This converts the optically pure waste back into a racemic mixture, which can then re-enter the resolution cycle.
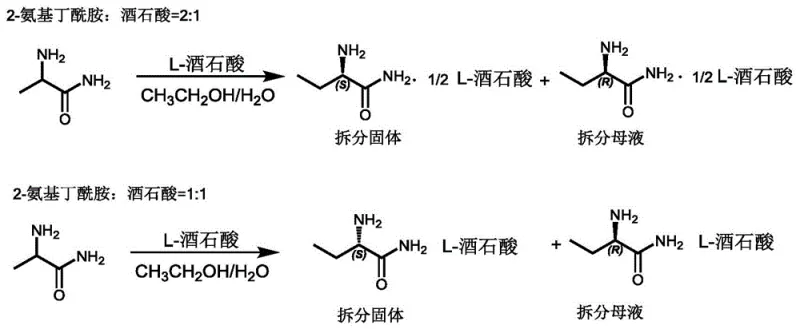
This mechanistic loop ensures that the chiral information provided by the L-tartaric acid is maximized. Unlike dynamic kinetic resolution methods that require expensive transition metal catalysts or enzymes, this organic catalytic system operates under mild thermal conditions, typically between 70°C and 100°C. The use of weak bases like ammonia or triethylamine facilitates the formation of the Schiff base intermediate necessary for racemization without degrading the sensitive amide functionality. Consequently, the optical purity of the final product remains consistently high, exceeding 99% ee, while the cumulative yield surpasses the theoretical limits of static resolution. This robust mechanism provides a reliable foundation for scaling up the production of high-purity pharmaceutical intermediates.
How to Synthesize S-2-Aminobutanamide Hydrochloride Efficiently
Implementing this resolution strategy requires precise control over reaction parameters to ensure optimal crystal growth and complete racemization. The process begins with the preparation of the resolution mixture, followed by the critical recycling of the mother liquor. Operators must carefully monitor the pH and temperature during the racemization phase to prevent side reactions while ensuring complete conversion of the R-isomer. The following guide outlines the standardized operational procedure derived from the patent examples, designed to assist process engineers in replicating these high-yield results in a pilot or production setting.
- Prepare the resolution mixture by combining 2-aminobutanamide and L-tartaric acid in a 2: 1 molar ratio within an ethanol/water solvent system.
- Separate the solid S-enantiomer tartrate salt, then treat the remaining mother liquor containing the R-enantiomer with a weak base and catalytic aldehyde.
- Heat the mother liquor to induce racemization, allowing the recycled tartrate to resolve the newly formed racemic mixture in subsequent cycles.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution technology translates directly into tangible operational improvements and risk mitigation. The primary value proposition lies in the drastic reduction of raw material waste. By recycling the mother liquor instead of disposing of it, manufacturers can effectively double the output from the same amount of initial racemic feedstock. This efficiency gain significantly lowers the cost per kilogram of the final active pharmaceutical ingredient (API) intermediate, providing a competitive edge in price-sensitive markets. Furthermore, the elimination of acetic acid and the reduction of organic solvent usage simplify the waste treatment process, reducing the burden on environmental compliance teams and lowering disposal costs.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of fresh resolving agents in every cycle. Since the L-tartaric acid is recovered and reused from the mother liquor, the recurring cost of chiral auxiliaries is virtually eliminated after the initial charge. Additionally, the use of low-loading aldehyde catalysts means that expensive reagents are not consumed in stoichiometric quantities. This lean reagent profile, combined with the ability to use cheaper solvents like ethanol and water, results in substantial cost savings across the entire production batch, enhancing the overall margin profile for the final drug product.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the volatility of chiral raw material markets. This method mitigates that risk by reducing dependence on external suppliers of expensive chiral starting materials like L-2-aminobutyric acid. By enabling the use of cheaper, readily available racemic mixtures and maximizing their conversion, manufacturers can secure a more stable and predictable supply of intermediates. The robustness of the aqueous solvent system also reduces the likelihood of production delays caused by solvent shortages or strict transportation regulations associated with hazardous volatile organic compounds.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen complexities, particularly regarding heat transfer and waste management. This resolution method is inherently scalable because it avoids exothermic hazards associated with strong acids or reactive metals. The aqueous workup simplifies the isolation of the product, making filtration and drying steps more efficient on a tonnage scale. From an environmental perspective, the reduction in organic waste and the avoidance of heavy metal catalysts align perfectly with green chemistry principles, ensuring that the manufacturing facility remains compliant with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this resolution method is essential for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding the practical application, purity profiles, and scalability of the technology. These insights are drawn directly from the experimental data and technical disclosures within the patent documentation to provide clarity on its industrial viability.
Q: How does this resolution method improve yield compared to traditional diastereomer splitting?
A: Traditional resolution typically caps yield at 50% because the unwanted enantiomer is discarded. This patented method utilizes aldehyde-catalyzed racemization to convert the unwanted R-enantiomer in the mother liquor back into a racemic mixture, allowing for infinite recycling and significantly higher total recovery rates.
Q: What are the environmental benefits of using water and alcohol solvents in this process?
A: Unlike previous methods that relied on acetic acid or large amounts of organic solvents which are difficult to recycle and pose disposal challenges, this process utilizes a green solvent system of water and methanol or ethanol. This simplifies waste treatment and reduces the environmental footprint of the manufacturing process.
Q: Why is the molar ratio of amine to tartaric acid critical in this resolution?
A: The process relies on a specific 2:1 molar ratio where one molecule of L-tartaric acid binds with two molecules of the amine. This stoichiometry allows the tartaric acid present in the mother liquor from the initial split to be fully utilized for resolving the racemized material, eliminating the need for fresh resolving agents in every cycle.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobutanamide Hydrochloride Supplier
As the demand for high-quality antiepileptic medications continues to grow globally, the need for efficient and sustainable intermediate synthesis has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced resolution technologies like the one described in CN109503408B to deliver superior value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-aminobutanamide hydrochloride meets the exacting standards required for GMP pharmaceutical manufacturing.
We invite pharmaceutical companies and contract manufacturers to explore how our optimized processes can enhance your supply chain resilience. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to drive down costs and accelerate the delivery of life-saving medications to patients worldwide through superior chemical engineering.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
