Scalable Manufacturing of 2-Aminobutanamide Hydrochloride via Bis(trichloromethyl)carbonate Route
The pharmaceutical industry continuously seeks robust synthetic routes for key intermediates like 2-aminobutanamide hydrochloride, a critical precursor in the manufacturing of the antiepileptic drug levetiracetam. Patent CN101684078A introduces a transformative chemical synthesis method that utilizes bis(trichloromethyl)carbonate (BTC) as a chlorinating agent to construct a stable cyclic intermediate, 4-ethyl-2,5-oxazolidinedione. This approach represents a significant departure from traditional methodologies, addressing long-standing issues regarding safety, environmental impact, and overall process efficiency. By leveraging the unique reactivity of BTC, the process achieves high conversion rates under relatively mild thermal conditions, typically ranging from -5 to 110°C, which facilitates easier temperature control during exothermic phases. The strategic formation of the oxazolidinedione ring serves as a protective and activating group, streamlining the subsequent ammoniation steps and ensuring a cleaner reaction profile with minimal byproduct formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aminobutanamide hydrochloride has relied on pathways that pose significant operational and environmental challenges for large-scale manufacturers. One common legacy method involves the use of butyric acid as a starting material, necessitating a cumbersome four-step sequence that includes bromination, esterification, ammoniation, and acidification. This route is particularly problematic due to the utilization of elemental bromine, a hazardous reagent that requires stringent safety protocols and specialized containment infrastructure to prevent exposure and corrosion. Furthermore, the ammoniation step in these older processes often demands high-pressure conditions, which increases capital expenditure for reactor vessels and introduces substantial safety risks related to pressure containment failures. Another prevalent method employs thionyl chloride for chlorination, which generates sulfur dioxide and hydrogen chloride gases; while HCl can be scrubbed, the efficient absorption of SO2 is difficult, leading to sulfur-containing wastewater that is costly and complex to treat according to modern environmental regulations.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes bis(trichloromethyl)carbonate to effect chlorination and cyclization in a single, streamlined operation. This reagent acts as a solid source of phosgene equivalents but is significantly safer to handle, transport, and store compared to gaseous phosgene or liquid thionyl chloride. The reaction proceeds through the formation of 4-ethyl-2,5-oxazolidinedione, a cyclic intermediate that exhibits superior stability compared to linear acyl chloride intermediates found in other routes. This stability allows for effective purification via distillation and recrystallization before the final ring-opening ammoniation, thereby preventing the carryover of impurities into the final API intermediate. The process operates at atmospheric pressure and moderate temperatures, eliminating the need for expensive high-pressure autoclaves and reducing the energy footprint of the manufacturing suite. Additionally, the absence of sulfur and bromine atoms in the reagent system drastically simplifies waste stream management, aligning perfectly with green chemistry principles and reducing the burden on effluent treatment plants.
Mechanistic Insights into BTC-Mediated Cyclization and Ring Opening
The core of this synthetic innovation lies in the efficient activation of the carboxylic acid group of 2-aminobutyric acid using bis(trichloromethyl)carbonate in the presence of an organic base catalyst. Mechanistically, the BTC reacts with the amino acid to generate an activated mixed anhydride or acyl chloride species in situ, which immediately undergoes intramolecular nucleophilic attack by the amine nitrogen to close the five-membered oxazolidinedione ring. The addition of organic amines such as triethylamine, pyridine, or 1,3-dimethyl-2-imidazolidinone plays a crucial catalytic role by scavenging the generated hydrogen chloride and enhancing the nucleophilicity of the reacting species. This cyclization is highly selective, minimizing the formation of polymeric byproducts or oligomers that often plague peptide coupling reactions. The resulting 4-ethyl-2,5-oxazolidinedione is a robust electrophile that resists racemization and degradation during isolation, providing a high-quality substrate for the subsequent transformation. The ability to isolate this intermediate with purities exceeding 98% ensures that the downstream chemistry begins with a well-defined starting material, which is essential for maintaining consistent quality in the final pharmaceutical product.
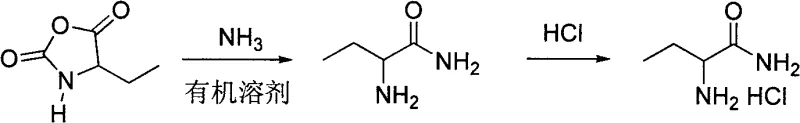
Following the isolation of the cyclic intermediate, the mechanism shifts to a nucleophilic ring-opening ammoniation. When the 4-ethyl-2,5-oxazolidinedione is exposed to saturated ammonia gas in a solvent like methanol or ethanol, the ammonia molecule attacks the carbonyl carbon of the cyclic carbonate moiety. This attack breaks the C-O bond of the ring, leading to the formation of a carbamate intermediate which spontaneously decomposes or rearranges to yield the primary amide functionality while releasing carbon dioxide. The final step involves acidification with hydrochloric acid, which protonates the alpha-amino group to form the stable hydrochloride salt. This salt formation is critical for the isolation of the product, as it allows for crystallization from the reaction mixture, effectively purifying the compound from any unreacted starting materials or soluble organic impurities. The entire sequence is designed to maximize atom economy and minimize the generation of hazardous waste, making it a superior choice for sustainable chemical manufacturing.
How to Synthesize 2-Aminobutanamide Hydrochloride Efficiently
To implement this advanced synthesis protocol effectively, manufacturers must carefully control the stoichiometry and reaction parameters to optimize yield and purity. The process begins with the precise weighing of 2-aminobutyric acid and bis(trichloromethyl)carbonate, typically in a molar ratio ranging from 1:0.33 to 1:1.5, with a preferred range around 1:0.5 to 1.2 to ensure complete conversion without excessive reagent waste. The reaction is conducted in a suitable organic solvent such as tetrahydrofuran, dichloromethane, or ethyl acetate, with the solvent mass being 3 to 30 times that of the starting acid to ensure proper mixing and heat dissipation. A catalytic amount of an organic amine is added to facilitate the reaction kinetics, and the mixture is heated to reflux or maintained at a controlled temperature between 25 and 100°C for a duration of 2 to 16 hours. Detailed standard operating procedures for the workup, including distillation, recrystallization, and final salt formation, are provided in the technical guide below.
- React 2-aminobutyric acid with bis(trichloromethyl)carbonate in an organic solvent at -5 to 110°C to form 4-ethyl-2,5-oxazolidinedione.
- Purify the cyclic intermediate via distillation and recrystallization to ensure high purity before the next step.
- Perform ammoniation with ammonia gas followed by acidification with hydrochloric acid to precipitate the final 2-aminobutanamide hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this BTC-based synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. By eliminating the need for hazardous bromine and high-pressure equipment, the process significantly reduces the capital and operational expenditures associated with safety compliance and specialized infrastructure maintenance. The substitution of thionyl chloride with bis(trichloromethyl)carbonate removes the complexity of treating sulfur-laden wastewater, leading to substantial cost savings in waste disposal and environmental remediation efforts. Furthermore, the high stability of the intermediate allows for flexible production scheduling, as the cyclic compound can be stockpiled if necessary without significant degradation, thereby enhancing supply chain resilience against demand fluctuations. The overall simplification of the process flow, reducing the number of unit operations and handling steps, translates into shorter batch cycle times and improved throughput capacity for existing manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like elemental bromine and the avoidance of high-pressure reactors result in a drastic simplification of the production infrastructure requirements. By utilizing solid bis(trichloromethyl)carbonate, companies can reduce storage costs and mitigate the risks associated with handling corrosive liquids, leading to lower insurance premiums and safety training costs. The high yield of the reaction minimizes raw material consumption per kilogram of finished product, directly improving the gross margin of the manufacturing operation. Additionally, the reduced burden on waste treatment facilities lowers the variable costs associated with effluent processing, contributing to a more lean and efficient cost structure for the production of this key pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The use of widely available and stable reagents ensures a consistent supply of raw materials, reducing the risk of production stoppages due to reagent shortages or delivery delays. The robustness of the intermediate 4-ethyl-2,5-oxazolidinedione allows for decoupling the synthesis stages, enabling manufacturers to produce and store the intermediate in bulk during periods of low demand to buffer against future supply shocks. This flexibility is crucial for maintaining continuous supply to downstream API manufacturers, especially in the context of global health demands for antiepileptic medications. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites with varying levels of equipment sophistication, further diversifying the supply base and reducing single-point failure risks.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard atmospheric pressure reactors and moderate temperature ranges, which are common features in most fine chemical plants. This ease of scale-up allows for rapid expansion of production capacity to meet growing market needs without requiring extensive new construction or engineering modifications. From an environmental perspective, the absence of sulfur and bromine byproducts simplifies regulatory compliance, making it easier to obtain and maintain operating permits in jurisdictions with strict environmental laws. The generation of less hazardous waste aligns with corporate sustainability goals and enhances the brand reputation of the manufacturer as a responsible supplier of pharmaceutical ingredients, which is increasingly valued by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and comparative analysis. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source. The answers reflect the specific advantages of the BTC route over legacy methods, focusing on safety, purity, and operational efficiency. Stakeholders are encouraged to review these points to gain a comprehensive understanding of the value proposition offered by this advanced manufacturing technique.
Q: Why is bis(trichloromethyl)carbonate preferred over thionyl chloride for this synthesis?
A: Bis(trichloromethyl)carbonate avoids the generation of sulfur dioxide and difficult-to-treat sulfur-containing wastewater associated with thionyl chloride, significantly reducing environmental compliance costs and safety hazards.
Q: What are the purity specifications achievable with this novel route?
A: The patent data indicates that the intermediate 4-ethyl-2,5-oxazolidinedione can achieve purities exceeding 98%, leading to a final product purity of approximately 98.5%, which is critical for downstream API synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (ranging from -5 to 110°C) and do not require high-pressure equipment, making the process inherently safer and easier to scale up compared to traditional high-pressure amination methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobutanamide Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain and have invested heavily in mastering advanced synthetic routes like the one described in patent CN101684078A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to verify the identity and purity of every batch before release. Our commitment to quality assurance means that you receive a product that is fully compliant with international standards, ready for immediate use in your API synthesis processes without the need for additional purification steps.
We invite you to collaborate with us to optimize your supply chain for levetiracetam production and other related therapeutic areas. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and logistical constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical expertise can drive efficiency and reliability in your manufacturing operations. Together, we can build a resilient and cost-effective supply partnership that supports your long-term business goals.
