Advanced CuI-Catalyzed Coupling Technology for Scalable Production of High-Purity Alkyne Intermediates
Introduction to Next-Generation Alkyne Coupling Technology
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more sustainable, cost-effective, and robust methodologies. A pivotal advancement in this domain is documented in Chinese Patent CN1634810A, which introduces a novel coupling reaction method for terminal alkynes and aryl halides. This technology leverages a copper(I) iodide (CuI) catalyst system augmented by amino acid additives, specifically N-substituted alpha-amino acids, to facilitate cross-coupling under remarkably mild conditions. Unlike traditional methods that often rely on expensive and toxic palladium catalysts, this approach utilizes earth-abundant copper, significantly altering the economic and environmental profile of producing aryl alkyne derivatives. The patent highlights that this catalytic system operates effectively at temperatures around 100°C, which is notably lower than many conventional protocols, thereby enhancing energy efficiency and safety profiles for industrial applications.
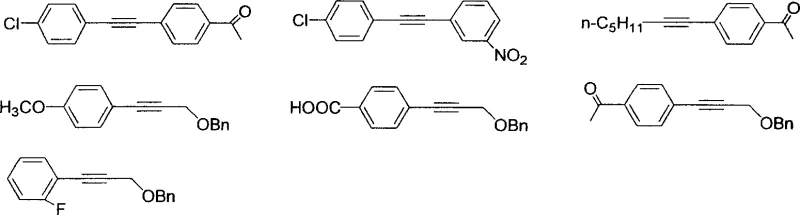
For R&D directors and process chemists, the implications of this technology are profound. It offers a viable pathway to synthesize complex molecular architectures found in active pharmaceutical ingredients (APIs) and agrochemicals without the burden of heavy metal residue removal associated with palladium. The versatility of the reaction is evidenced by its compatibility with a wide range of substrates, including those bearing electron-withdrawing and electron-donating groups. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative catalytic cycles is crucial for maintaining a competitive edge in the global market, ensuring that the supply chain remains resilient against fluctuations in precious metal prices and regulatory pressures regarding heavy metal limits in drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-carbon bonds between alkynes and aryl halides has been dominated by the Sonogashira coupling reaction, which typically employs a dual catalytic system of palladium and copper. While effective, this conventional approach presents several significant drawbacks for large-scale manufacturing. Firstly, the reliance on palladium catalysts introduces substantial cost volatility, as palladium is a precious metal with fluctuating market prices that can drastically impact the cost of goods sold (COGS). Secondly, the removal of trace palladium from the final product is a critical and often expensive downstream processing step, required to meet stringent regulatory limits for heavy metals in pharmaceuticals. Furthermore, traditional protocols often necessitate harsh reaction conditions, including high temperatures and the use of hazardous solvents or bases, which can compromise the integrity of sensitive functional groups and increase the generation of hazardous waste. These factors collectively contribute to longer lead times and reduced overall process efficiency.
The Novel Approach
The methodology described in patent CN1634810A represents a paradigm shift by replacing the palladium component with a robust copper-only catalytic system stabilized by amino acid ligands. This novel approach effectively mitigates the economic and environmental burdens associated with precious metal catalysis. By utilizing CuI in conjunction with ligands such as N,N-dimethylglycine hydrochloride, the reaction achieves high conversion rates at moderate temperatures, typically around 100°C. This reduction in thermal demand not only lowers energy consumption but also minimizes the risk of thermal degradation for thermally labile substrates. The use of inexpensive and air-stable reagents simplifies the handling and storage requirements, making the process inherently safer and more accessible for commercial scale-up. Moreover, the absence of palladium eliminates the need for specialized scavenging resins or complex purification steps to remove trace metals, streamlining the workflow and reducing the overall environmental footprint of the synthesis.
Mechanistic Insights into CuI-Amino Acid Catalyzed Coupling
The efficacy of this coupling reaction hinges on the synergistic interaction between the copper catalyst and the amino acid ligand. Mechanistically, the amino acid acts as a bidentate or monodentate ligand that coordinates to the copper center, stabilizing the active catalytic species and preventing the formation of inactive copper aggregates or precipitates. This stabilization is crucial for maintaining catalytic turnover over the extended reaction periods often required for less reactive aryl bromides. The proposed cycle involves the oxidative addition of the aryl halide to the copper center, facilitated by the electron-donating properties of the ligand, followed by the transmetallation with the terminal alkyne which has been activated by the base (typically K2CO3 or Cs2CO3). The final reductive elimination step releases the coupled product and regenerates the active copper catalyst. The presence of the amino acid ligand lowers the activation energy for these steps, allowing the reaction to proceed smoothly at 100°C, a temperature significantly lower than what is typically required for ligand-free copper catalysis.
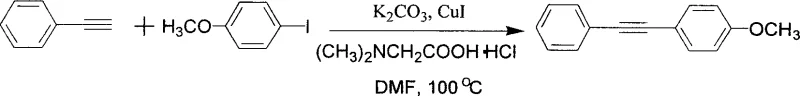
From an impurity control perspective, this mechanism offers distinct advantages. The mild conditions reduce the likelihood of side reactions such as homocoupling of the alkyne (Glaser coupling) or dehalogenation of the aryl ring, which are common pitfalls in harsher coupling environments. The specificity of the ligand-copper complex ensures that the reaction proceeds with high chemoselectivity, tolerating functional groups like nitro, ketone, and ester moieties that might otherwise be susceptible to reduction or hydrolysis under more aggressive conditions. For quality control teams, this translates to a cleaner crude reaction profile, simplifying the purification process and resulting in a final product with a superior impurity profile. The ability to consistently produce high-purity intermediates is paramount for downstream API synthesis, where impurity carryover can complicate registration and approval processes.
How to Synthesize Aryl Alkynes Efficiently
To implement this technology in a laboratory or pilot plant setting, the procedure involves charging a reaction vessel with the terminal alkyne and aryl halide substrates in a polar aprotic solvent such as DMF or DMSO. A base like potassium carbonate is added to deprotonate the alkyne, generating the nucleophilic acetylide species in situ. The catalytic system, consisting of CuI and the amino acid ligand (e.g., N,N-dimethylglycine hydrochloride), is then introduced. The mixture is heated to approximately 100°C under an inert atmosphere to prevent oxidation of the copper species. Detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with terminal alkyne, aryl halide (iodide or bromide), and potassium carbonate base.
- Add the catalytic system comprising CuI and N,N-dimethylglycine hydrochloride ligand in a molar ratio of approximately 1: 3.
- Heat the mixture in DMF solvent at 100°C under inert atmosphere for 24 hours, followed by aqueous workup and silica gel purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CuI-amino acid catalytic technology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs. By eliminating the need for palladium catalysts, which are subject to significant price volatility and supply constraints, manufacturers can stabilize their cost structures and improve margin predictability. The reagents used in this process, including copper iodide and simple amino acid derivatives, are commodity chemicals that are readily available from multiple global suppliers, reducing the risk of supply chain disruptions. This abundance ensures a reliable supply of critical intermediates, which is essential for maintaining continuous production schedules in the fast-paced pharmaceutical and agrochemical industries.
- Cost Reduction in Manufacturing: The economic impact of switching to this copper-based system is substantial. Palladium catalysts are not only expensive to purchase but also incur additional costs related to recovery and recycling or disposal. By utilizing a non-precious metal catalyst, the direct material cost per kilogram of product is significantly lowered. Furthermore, the simplified workup procedure, which does not require expensive metal scavengers to meet residual metal specifications, reduces the consumption of auxiliary materials and lowers waste disposal costs. The energy savings achieved by operating at lower temperatures (around 100°C compared to higher temperatures for other methods) also contribute to a leaner manufacturing budget, enhancing the overall cost competitiveness of the final product in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable reagents. Unlike specialized phosphine ligands or sensitive palladium complexes that may require cold chain shipping or have limited shelf lives, the amino acid ligands and copper salts used in this process are air-stable and easy to handle. This robustness simplifies logistics and inventory management, allowing for larger batch sizes and longer storage periods without degradation. The ability to source key materials from diverse geographic regions mitigates the risk of geopolitical supply shocks, ensuring that production timelines are met consistently. This reliability is a critical factor for long-term contracts with major pharmaceutical clients who prioritize supply security above all else.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the benign nature of the reagents and the moderate reaction conditions. The absence of toxic heavy metals like palladium simplifies environmental compliance and waste treatment protocols. Effluent streams are easier to treat, and the solid waste generated does not require classification as hazardous heavy metal waste, leading to lower disposal fees and a reduced environmental footprint. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for stakeholders and customers focused on environmental, social, and governance (ESG) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this coupling technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making.
Q: What are the advantages of using amino acid ligands in Cu-catalyzed coupling compared to traditional Pd systems?
A: The use of amino acid ligands like N,N-dimethylglycine allows the reaction to proceed at significantly lower temperatures (around 100°C vs higher temps for some Pd systems) and eliminates the need for expensive palladium catalysts, reducing heavy metal contamination risks in the final product.
Q: Can this coupling method tolerate sensitive functional groups like nitro or ketone moieties?
A: Yes, the patent data demonstrates excellent tolerance for various functional groups including nitro (-NO2), ketone (-COCH3), carboxylic acid (-COOH), and halogens (F, Cl), yielding products with high purity and minimal side reactions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Absolutely. The reagents used (CuI, K2CO3, amino acids) are inexpensive and air-stable, and the reaction conditions are mild (100°C), making it highly scalable and safer for industrial production compared to pyrophoric or highly toxic alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terminal Alkynes Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the CuI-amino acid coupling system described in patent CN1634810A. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this method are fully realized in a manufacturing environment. We are committed to delivering high-purity intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to verify that every batch conforms to the highest industry standards. Our infrastructure is designed to handle complex synthetic challenges, providing a seamless transition from process development to commercial supply.
We invite you to collaborate with us to optimize your supply chain for aryl alkyne derivatives. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you leverage this efficient, cost-effective technology to accelerate your drug development timelines and secure a competitive advantage in the marketplace.
