Advanced DMAP-Catalyzed Synthesis of 2-Chloroadenosine for Commercial API Manufacturing
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing critical anticancer intermediates. Patent CN112159447B introduces a groundbreaking synthesis process for 2-chloroadenosine, a pivotal precursor for the anti-leukemia drug cladribine and a potent adenosine receptor agonist. This technology addresses the longstanding challenges associated with nucleoside synthesis, specifically the reliance on toxic heavy metal catalysts and harsh reaction conditions that plague conventional methods. By leveraging 4-dimethylaminopyridine (DMAP) as an organocatalyst, this novel route achieves exceptional yields exceeding 90% and purity levels above 99%, representing a significant leap forward in process chemistry. For R&D directors and procurement managers alike, this patent offers a compelling solution that aligns with modern green chemistry principles while ensuring robust supply chain reliability for high-value API intermediates.
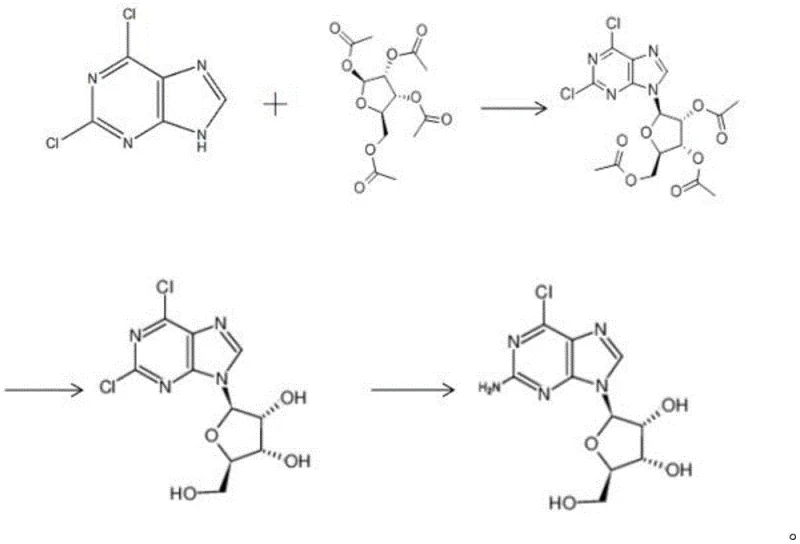
The strategic implementation of this DMAP-catalyzed pathway allows manufacturers to bypass the regulatory and operational hurdles associated with heavy metal residues. In the context of global regulatory tightening, the ability to produce high-purity 2-chloroadenosine without tin or other metallic contaminants is a distinct competitive advantage. This report analyzes the technical merits of this innovation, demonstrating how it serves as a reliable pharmaceutical intermediate supplier solution that drastically simplifies downstream processing and enhances overall process economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloroadenosine and related nucleosides has been fraught with inefficiencies, primarily due to the reliance on Lewis acid catalysts such as tin tetrachloride (SnCl4) or strong acids like trifluoromethanesulfonic acid. These traditional methodologies present severe drawbacks for commercial manufacturing. Firstly, the use of tin tetrachloride introduces significant heavy metal contamination into the reaction matrix, necessitating rigorous and costly purification steps to meet stringent pharmaceutical specifications. Secondly, these reactions often proceed under harsh conditions that can degrade sensitive sugar moieties, leading to complex impurity profiles and reduced overall yields, often hovering around merely 62%. Furthermore, the starting materials traditionally employed, such as 2-chloroadenine, are not only expensive but also difficult to source in bulk quantities, creating bottlenecks in the supply chain. The environmental footprint of disposing of heavy metal waste further exacerbates the cost burden, making these legacy processes unsustainable for modern, large-scale API production.
The Novel Approach
In stark contrast, the methodology disclosed in CN112159447B utilizes a transition-metal-free organocatalytic system centered on DMAP. This approach fundamentally shifts the paradigm of nucleoside glycosylation by employing mild, non-toxic reagents that are readily available on the global market. The process initiates with the condensation of 2,6-dichloropurine and tetraacetyl ribose in an aromatic solvent, typically toluene, under reflux conditions. The use of DMAP as a nucleophilic catalyst facilitates the formation of the glycosidic bond with remarkable selectivity and efficiency, driving yields to over 90%. This novel route eliminates the need for expensive and hazardous Lewis acids, thereby simplifying the workup procedure to basic crystallization steps. By replacing scarce starting materials with cost-effective 2,6-dichloropurine, the process ensures a stable and economical supply chain, directly addressing the pain points of cost reduction in API manufacturing faced by procurement teams worldwide.
Mechanistic Insights into DMAP-Catalyzed Condensation
The core of this technological breakthrough lies in the mechanistic role of 4-dimethylaminopyridine (DMAP) during the glycosylation step. Unlike traditional Lewis acids that activate the sugar donor through coordination, DMAP acts as a potent nucleophilic catalyst. It likely attacks the anomeric center of the tetraacetyl ribose to form a highly reactive glycosyl pyridinium intermediate. This activated species is then susceptible to nucleophilic attack by the N9 nitrogen of the 2,6-dichloropurine base. This mechanism proceeds under neutral to slightly basic conditions, which is crucial for preserving the integrity of the acid-sensitive acetyl protecting groups on the ribose ring. The reaction is optimized at temperatures between 100°C and 130°C in toluene, ensuring sufficient energy for the condensation while maintaining a homogeneous reaction phase that promotes high conversion rates. The precise control of the catalyst loading, with a mass ratio of 2,6-dichloropurine to DMAP ranging from 1:0.004 to 1:0.02, is critical; too little catalyst results in incomplete conversion, while excessive amounts do not significantly improve yield but increase purification load.
Impurity control is another hallmark of this mechanistic design. In conventional acid-catalyzed routes, the harsh acidic environment often leads to the hydrolysis of acetyl groups or the formation of N7-isomers, which are difficult to separate. The DMAP-catalyzed pathway, operating under milder thermal conditions without strong acids, minimizes these side reactions. The subsequent hydrolysis step utilizes sodium methoxide in methanol at a gentle 20-30°C, selectively removing the acetyl groups without affecting the chloro-substituents on the purine ring. Finally, the ammoniation step replaces the C6-chlorine atom with an amino group using aqueous ammonia. This sequential specificity ensures that the final product, 2-chloroadenosine, is obtained with purity levels exceeding 99%, significantly reducing the burden on quality control laboratories and ensuring a cleaner impurity profile for regulatory filings.
How to Synthesize 2-Chloroadenosine Efficiently
The synthesis of 2-chloroadenosine via this patented route is designed for operational simplicity and robustness, making it highly attractive for process chemists aiming for rapid technology transfer. The protocol involves three distinct stages: the initial condensation to form the protected nucleoside intermediate, followed by deprotection, and finally, amination. Each step utilizes standard laboratory equipment and commercially available solvents, removing the need for specialized high-pressure reactors or exotic reagents. The detailed standardized synthesis steps below outline the precise stoichiometry and conditions required to replicate the high yields and purity reported in the patent data, serving as a foundational guide for scaling this chemistry from benchtop to pilot plant.
- Condensation Reaction: React 2,6-dichloropurine with tetraacetyl ribose in toluene using DMAP as a catalyst at 100-130°C to form the triacetyl intermediate.
- Hydrolysis Reaction: Treat the intermediate with sodium methoxide in methanol at 20-30°C to remove acetyl protecting groups.
- Ammoniation Reaction: React the deprotected nucleoside with aqueous ammonia at 20-30°C to substitute the chlorine at the C6 position, yielding 2-chloroadenosine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMAP-catalyzed process translates into tangible strategic benefits beyond mere chemical yield. The elimination of heavy metal catalysts like tin tetrachloride removes a major cost center associated with waste treatment and metal scavenging resins. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates the risk of supply disruptions caused by the volatility of specialty metal prices. Furthermore, the use of commodity chemicals such as toluene, methanol, and aqueous ammonia ensures that the raw material supply chain is resilient and geographically diverse, reducing the risk of single-source dependency.
- Cost Reduction in Manufacturing: The transition from expensive, hazardous Lewis acids to catalytic amounts of DMAP represents a substantial cost saving. DMAP is a widely produced industrial chemical with a stable price point, unlike specialized fluorinated acids or metal salts. Additionally, the simplified purification process, which relies on crystallization rather than complex chromatography or multiple extraction steps to remove metals, drastically reduces solvent consumption and processing time. This streamlined workflow lowers utility costs and labor hours, contributing to a more lean and efficient manufacturing operation that enhances overall profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on 2,6-dichloropurine and tetraacetyl ribose as starting materials secures a robust supply chain. These precursors are manufactured by multiple global suppliers, ensuring competitive pricing and consistent availability. In contrast, legacy methods relying on 2-chloroadenine face supply constraints due to its niche production volume. By decoupling the synthesis from scarce reagents, manufacturers can guarantee continuous production schedules, effectively reducing lead time for high-purity intermediates and ensuring that downstream API production lines remain uninterrupted even during market fluctuations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently safer and easier to scale. The absence of corrosive strong acids and toxic heavy metals simplifies reactor maintenance and reduces the hazard classification of the waste stream. This facilitates easier permitting for commercial scale-up of complex nucleosides in regulated jurisdictions. The mild reaction temperatures (20-30°C for hydrolysis and amination) also reduce energy consumption for heating and cooling, aligning with corporate sustainability goals. The ability to handle larger batches with consistent quality makes this route ideal for meeting the growing global demand for anticancer therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on why this method outperforms traditional alternatives in both yield and purity metrics.
Q: Why is the DMAP-catalyzed route superior to traditional tin tetrachloride methods?
A: Traditional methods using tin tetrachloride (SnCl4) suffer from heavy metal contamination, requiring complex and costly removal steps. The DMAP route is metal-free, operates under milder conditions, and achieves significantly higher yields (over 90% vs. ~62%) and purity (>99%).
Q: What are the critical reaction conditions for the condensation step?
A: The condensation requires refluxing in an aromatic solvent like toluene at temperatures between 100°C and 130°C. The mass ratio of 2,6-dichloropurine to the DMAP catalyst is optimized between 1:0.004 and 1:0.02 to maximize conversion while minimizing catalyst load.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes common solvents like toluene and methanol, avoids hazardous heavy metals, and relies on simple crystallization for purification, making it ideal for commercial scale-up of complex nucleosides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloroadenosine Supplier
The technical superiority of the DMAP-catalyzed synthesis route positions 2-chloroadenosine as a highly viable candidate for commercial development. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory protocols into robust industrial realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and crystallization requirements of this process, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity 2-chloroadenosine that adheres to the highest international pharmacopeial standards.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for your pipeline. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of this critical oncology intermediate, ensuring your drug development programs proceed without interruption.
