Advanced Refining Technology for High-Purity Chiral Alcohols in Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust methodologies to enhance the purity and stereochemical integrity of critical intermediates, particularly chiral alcohols which serve as foundational building blocks for numerous active pharmaceutical ingredients (APIs). Patent CN112624901B introduces a groundbreaking refinement strategy that addresses the persistent challenges of residual impurities and low stereoselectivity often encountered in traditional synthesis routes. This innovation combines a specialized asymmetric catalytic hydrogenation step utilizing a unique polymer-supported ruthenium catalyst with a subsequent precision recrystallization process using specific ether-based solvents. By integrating these advanced catalytic systems with optimized downstream purification techniques, manufacturers can achieve exceptional purity profiles while maintaining high yields, a balance that is notoriously difficult to strike in complex organic synthesis. The technology represents a significant leap forward for producers aiming to supply high-purity pharmaceutical intermediates to global markets.
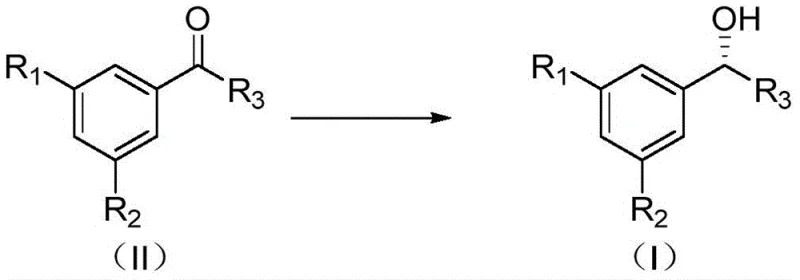
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing chiral alcohols often rely on standard asymmetric catalytic hydrogenation using homogeneous catalysts, which frequently suffer from suboptimal stereoselectivity and difficult product isolation. A major bottleneck in these conventional processes is the inability to effectively remove unreacted starting materials and enantiomeric impurities, which possess physicochemical properties strikingly similar to the target product. Consequently, standard solvent washing or basic recrystallization techniques often fail to reduce raw material residuals below 0.9% and other impurities below 0.5%, necessitating multiple purification cycles that drastically erode overall process yield. Furthermore, the use of homogeneous catalysts introduces significant challenges regarding heavy metal contamination, requiring expensive and time-consuming metal scavenging steps that complicate the supply chain and increase production costs. These inefficiencies create substantial barriers for scaling up production to meet the rigorous quality standards demanded by modern regulatory bodies.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles by employing a synergistic combination of a tailored solid-phase catalyst and a specifically selected refining solvent system. Unlike traditional methods, this process utilizes a polymeric bisphosphine dinitrogen ruthenium catalyst that not only enhances asymmetric conversion rates but also allows for facile separation via simple filtration, thereby minimizing metal leaching into the product stream. Following the hydrogenation step, the crude product undergoes a targeted recrystallization using solvents such as isopropyl ether or diethyl ether, which are uniquely capable of differentiating between the target chiral alcohol and its stubborn impurities. This dual-strategy ensures that process impurities are controlled below 0.3% without the need for repetitive purification steps that compromise yield. The result is a streamlined, industrially viable process that delivers high-purity chiral alcohols suitable for direct use in sensitive API manufacturing workflows.
Mechanistic Insights into Polymer-Supported Ruthenium Catalysis
The core of this technological advancement lies in the sophisticated design of the catalyst, specifically the incorporation of a polyethylene glycol (PEG) chain into the ligand architecture of the ruthenium complex. This structural modification serves a dual mechanistic purpose: firstly, the specific chain length of the PEG moiety acts as a guide to assist the catalytic reaction, effectively enhancing the catalytic activity and driving higher asymmetric conversion rates. Secondly, the presence of the polymer backbone enables the solid-phase immobilization of the catalyst, which fundamentally alters the interaction between the heavy metal ions and the reaction medium. This immobilization prevents metal ions from freely entering the solution phase, thereby drastically reducing the risk of heavy metal residues in the final product, a critical parameter for pharmaceutical compliance. The catalyst structure, often represented by Formula (C) in the patent data, demonstrates superior selectivity towards the substrate, ensuring that the desired enantiomer is produced with high fidelity.
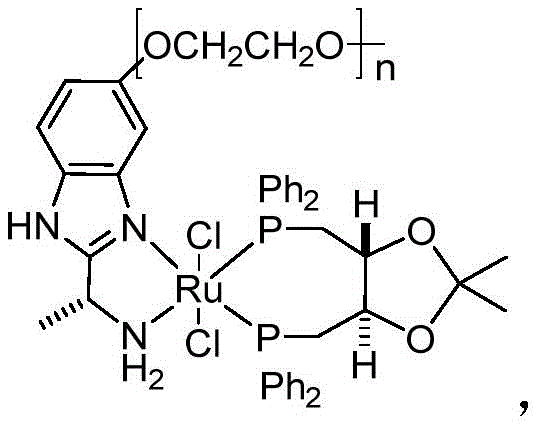
Regarding impurity control mechanisms, the refining solvent plays a pivotal role in the thermodynamic equilibrium of the crystallization process. Solvents like isopropyl ether exhibit specific solvation properties that favor the precipitation of the target chiral alcohol while keeping structurally similar impurities, such as the starting ketone or the opposite enantiomer, dissolved in the mother liquor. This selective crystallization is achieved by carefully controlling the dissolution temperature at 50-60°C followed by a gradual cooling to 0-5°C, which promotes the formation of a pure crystal lattice. The efficiency of this mechanism is evidenced by the ability to reduce single impurity levels to below 0.05% and total impurities to under 0.3% in a single pass. This level of control eliminates the need for aggressive chromatographic separations, making the process not only chemically elegant but also economically superior for large-scale operations.
How to Synthesize High-Purity Chiral Alcohol Efficiently
The synthesis of these high-value intermediates begins with the preparation of the crude chiral alcohol via asymmetric hydrogenation of the corresponding ketone precursor using the specialized polymer-supported catalyst under hydrogen pressure. Once the crude material is obtained, the critical refining stage involves dissolving the solid in a precise volume of refining solvent, such as isopropyl ether, at elevated temperatures to ensure homogeneity. The subsequent cooling phase must be managed carefully to induce nucleation and crystal growth without trapping impurities within the lattice. For a comprehensive, step-by-step technical guide including exact reagent ratios, pressure settings, and safety protocols, please refer to the standardized operating procedure outlined below.
- Dissolve the crude chiral alcohol product in a refined solvent such as isopropyl ether or diethyl ether at a temperature range of 50-60°C to ensure complete solubilization.
- Gradually cool the solution to a temperature between 0-5°C to induce controlled crystallization, allowing impurities to remain in the mother liquor.
- Perform solid-liquid separation via suction filtration, collect the crystals, and dry under reduced pressure at 25-40°C to obtain the final high-purity refined product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refining technology translates into tangible strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the drastic simplification of the purification workflow, which removes the need for multiple recrystallization cycles or complex chromatographic separations that traditionally bottleneck production timelines. By achieving high purity in fewer steps, manufacturers can significantly reduce the consumption of auxiliary materials and energy, leading to a more sustainable and cost-effective production model. Furthermore, the use of a solid-phase catalyst simplifies the recovery and recycling of expensive precious metals, directly contributing to cost reduction in API manufacturing by lowering the overall catalyst loading requirements over time.
- Cost Reduction in Manufacturing: The elimination of extensive metal scavenging processes and the reduction in solvent usage due to higher single-pass efficiency result in substantial operational savings. The ability to recover and reuse the polymer-supported catalyst further enhances the economic viability of the process, ensuring that the cost of goods sold remains competitive even for complex chiral intermediates. Additionally, the high yield retention during purification means that less raw material is wasted, optimizing the overall material balance of the production facility.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in crude quality ensures a consistent supply of high-purity intermediates, reducing the risk of batch failures that can disrupt downstream API synthesis. The simplicity of the equipment requirements, which do not demand specialized instrumentation, allows for flexible manufacturing across different sites, thereby diversifying supply sources and mitigating geopolitical or logistical risks. This reliability is crucial for maintaining uninterrupted production schedules for life-saving medications.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial application in mind, allowing for seamless transition from pilot batches to multi-ton commercial production without loss of efficiency. The reduction in solvent waste and the minimization of heavy metal discharge align perfectly with increasingly stringent environmental regulations, positioning manufacturers as responsible partners in the green chemistry initiative. This compliance reduces the administrative burden associated with waste disposal and environmental reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology, derived directly from the patent specifications and experimental data. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this process into their existing manufacturing lines. The answers provided reflect the specific capabilities and limitations observed during the development of this method.
Q: What specific solvents are most effective for refining these chiral alcohols?
A: The patent identifies diethyl ether, n-heptane, methyl tert-butyl ether, and isopropyl ether as highly effective refining solvents. Among these, isopropyl ether is particularly noted for achieving optimal balance between yield and impurity removal.
Q: How does the polymer-supported catalyst improve the process compared to homogeneous catalysts?
A: The polymer-supported ruthenium catalyst facilitates easy separation via simple filtration, significantly reducing heavy metal residues in the final product and simplifying the downstream purification workflow.
Q: What level of impurity control can be achieved with this method?
A: This refining method effectively controls process impurities to below 0.3%, with single impurities often maintained under 0.05%, meeting stringent requirements for pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering intermediates that meet the highest standards of purity and consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative refining techniques described in patent CN112624901B can be effectively deployed at an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to verify that every batch of chiral alcohol meets the exacting requirements of our global clientele. Our commitment to quality assurance ensures that the complex stereochemical integrity of your intermediates is preserved throughout the manufacturing journey.
We invite you to collaborate with us to leverage these advanced processing capabilities for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized refining methods can enhance the efficiency and reliability of your supply chain.
