Advanced Synthetic Route for Cyproheptadine Hydrochloride: Enabling Scalable Pharmaceutical Manufacturing
Introduction to the Novel Synthetic Breakthrough
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antihistamine agents, and the recent disclosure in patent CN112552230B presents a transformative approach to producing cyproheptadine hydrochloride. This potent antihistamine, widely utilized for treating allergic conditions and stimulating appetite, has traditionally been synthesized via hazardous Grignard reactions that demand rigorous exclusion of moisture and oxygen. The new methodology fundamentally shifts the paradigm by employing a low-valent titanium-mediated reductive coupling strategy, which not only simplifies the operational workflow but also drastically enhances the safety profile of the manufacturing process. By reacting dibenzosuberiene-5-ketone with N-methylpiperidine-4-ketone under mild thermal conditions, this innovation delivers a high-purity intermediate ready for subsequent salification. 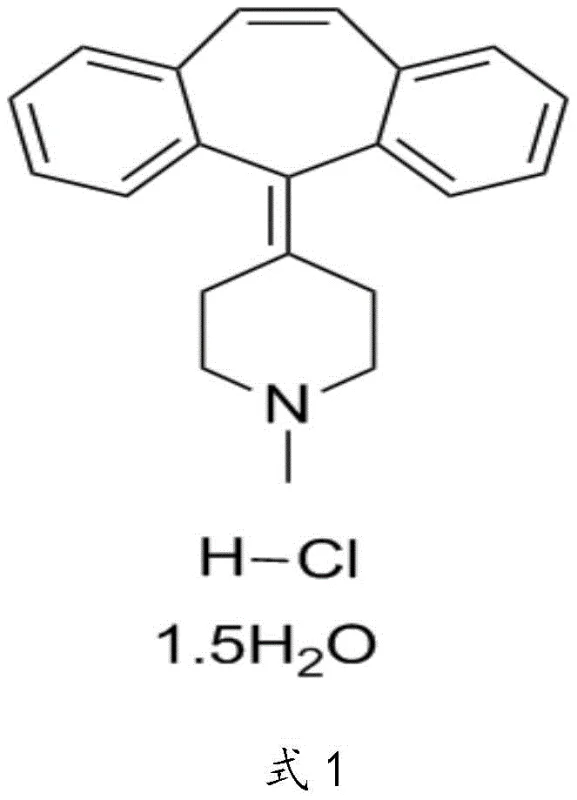 This structural integrity is maintained throughout the process, ensuring that the final active pharmaceutical ingredient meets the stringent quality standards required for global regulatory compliance and patient safety.
This structural integrity is maintained throughout the process, ensuring that the final active pharmaceutical ingredient meets the stringent quality standards required for global regulatory compliance and patient safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of cyproheptadine hydrochloride has been plagued by the inherent dangers and complexities associated with Grignard chemistry. The traditional route necessitates the preparation of a Grignard reagent from 1-methyl-4-chloropiperidine and magnesium chips, a step that requires absolutely anhydrous and oxygen-free conditions to prevent catastrophic failure or fire. This imposes a heavy burden on infrastructure, as all equipment, pipelines, and nitrogen systems must be meticulously dried in advance, leading to prolonged setup times and increased energy consumption. Furthermore, the multi-step nature of the conventional process, involving subsequent dehydration with acetic anhydride, results in lower overall yields and generates significant chemical waste. These operational bottlenecks make the traditional method economically inefficient and logistically challenging for manufacturers aiming to secure a reliable supply chain for high-volume production.
The Novel Approach
In stark contrast, the innovative process detailed in the patent utilizes a low-valent titanium system that operates effectively under much more forgiving conditions. By dissolving the ketone precursors in solvents such as tetrahydrofuran or pyridine and introducing titanium tetrachloride alongside a reducing agent like zinc powder, the reaction proceeds smoothly at temperatures between 80°C and 110°C. This elimination of the Grignard step removes the critical dependency on ultra-dry environments, thereby simplifying the reactor requirements and reducing the risk of batch loss due to moisture intrusion. The streamlined workflow consolidates several transformation steps into a more direct coupling reaction, which not only accelerates the production cycle but also minimizes the formation of complex byproducts. Consequently, this approach offers a pathway to significantly higher throughput and operational stability, making it an ideal candidate for modernizing existing manufacturing facilities.
Mechanistic Insights into Low-Valent Titanium Mediated Coupling
The core of this technological advancement lies in the mechanistic efficiency of the low-valent titanium species generated in situ, which facilitates the reductive coupling of the two ketone moieties. Unlike nucleophilic additions that are highly sensitive to proton sources, the titanium-mediated mechanism tolerates a broader range of reaction conditions while maintaining high chemoselectivity for the carbonyl groups involved. The reaction likely proceeds through the formation of a pinacol-like intermediate which subsequently undergoes deoxygenation to form the exocyclic double bond characteristic of the cyproheptadine scaffold. This specific pathway avoids the harsh acidic conditions often required for dehydration in older methods, thereby preserving the integrity of the sensitive piperidine ring and preventing ring-opening side reactions. The careful control of the titanium-to-ketone ratio, typically around 6:10 by weight, ensures complete conversion of the starting materials while minimizing the presence of unreacted precursors in the crude mixture.
Impurity control is further enhanced through a sophisticated purification sequence that leverages differential solubility and crystallization dynamics. Following the initial coupling, the crude product is subjected to a rigorous extraction protocol using toluene and water, which effectively partitions organic impurities and inorganic salts away from the desired intermediate. The subsequent crystallization from an ethanol-water mixture is a critical step that refines the molecular lattice, excluding structurally similar analogs and ensuring a sharp melting point profile. Finally, the salification with hydrochloric acid is performed in a controlled manner to precipitate the hydrochloride salt, followed by a decolorization step using activated carbon and titanium rod filtration. This multi-barrier purification strategy guarantees that the final product achieves a purity level of 99.9%, effectively eliminating potential genotoxic impurities or heavy metal residues that could compromise regulatory approval.
How to Synthesize Cyproheptadine Hydrochloride Efficiently
The implementation of this synthetic route requires precise adherence to the optimized parameters established in the patent examples to ensure reproducibility and maximum yield. Operators must carefully manage the addition rates of the reducing agents and maintain the reaction temperature within the narrow window of 80°C to 110°C to balance reaction kinetics with byproduct suppression. The workup procedure involves distinct phase separations and solvent swaps that are critical for removing the titanium residues before the final crystallization. For a comprehensive understanding of the exact stoichiometric ratios, solvent volumes, and timing for each unit operation, please refer to the standardized technical guide below which outlines the step-by-step execution plan for pilot and commercial batches.
- Perform reductive coupling of dibenzosuberiene-5-ketone and N-methylpiperidine-4-ketone using low-valent titanium and a reducing agent at 80-110°C.
- Execute a multi-stage workup involving filtration, solvent recovery, and toluene extraction to isolate the crude cyproheptadine intermediate.
- Purify via ethanol-water crystallization, followed by salification with hydrochloric acid and final decolorization to obtain the finished product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthetic method offers profound advantages in terms of cost structure and supply security. By eliminating the need for Grignard reagents, manufacturers can bypass the procurement of highly reactive and hazardous materials that often command premium pricing and require specialized storage logistics. The simplified reaction conditions mean that production can be scheduled with greater flexibility, as there is no longer a need for extensive pre-drying campaigns that tie up reactor capacity for days. This operational agility translates directly into reduced lead times and a more responsive supply chain capable of meeting fluctuating market demands for antihistamine APIs. Furthermore, the use of common, commercially available reagents like zinc powder and titanium tetrachloride ensures a stable raw material supply base that is less susceptible to geopolitical disruptions or vendor shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of expensive and hazardous Grignard preparation steps, which significantly lowers both raw material costs and safety compliance expenditures. The milder reaction conditions reduce energy consumption associated with heating and cooling cycles, while the higher overall yield means less starting material is wasted per kilogram of final product. Additionally, the simplified purification train reduces the volume of solvents required for washing and recrystallization, leading to substantial savings in solvent procurement and waste disposal fees. These cumulative efficiencies result in a drastically improved cost of goods sold (COGS), allowing suppliers to offer more competitive pricing without compromising on quality margins.
- Enhanced Supply Chain Reliability: The robustness of the low-valent titanium chemistry ensures consistent batch-to-batch performance, which is critical for maintaining uninterrupted supply to downstream formulation partners. Because the process does not rely on moisture-sensitive reagents, the risk of batch failure due to environmental factors is minimized, thereby enhancing the predictability of delivery schedules. The scalability of the method allows for seamless transition from pilot plant quantities to multi-ton commercial production without the need for fundamental process redesign. This reliability fosters stronger long-term partnerships between chemical suppliers and pharmaceutical companies, as it mitigates the risk of stockouts that can halt the production of life-saving allergy medications.
- Scalability and Environmental Compliance: Environmental sustainability is increasingly a key criterion for supplier selection, and this green chemistry approach aligns perfectly with modern ESG goals. The process generates fewer hazardous byproducts and eliminates the need for quenching large quantities of reactive magnesium, which simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. The ability to recycle solvents like toluene and ethanol further contributes to a circular economy model within the plant. Moreover, the use of standard stainless steel equipment rather than specialized glass-lined or Hastelloy reactors for Grignard chemistry lowers the capital expenditure barrier for scaling up, enabling faster deployment of new production lines to meet global health needs.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their supply chains, we have compiled answers to the most pertinent questions regarding the process specifications and quality outcomes. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a transparent view of the method's capabilities. Understanding these technical nuances is essential for R&D directors and procurement managers who are tasked with validating new vendors or optimizing existing manufacturing protocols for cyproheptadine hydrochloride.
Q: How does the new titanium-mediated route improve safety compared to the traditional Grignard method?
A: The novel process eliminates the need for preparing Grignard reagents, which require strictly anhydrous and oxygen-free environments and pose significant fire hazards. By utilizing low-valent titanium in standard solvents like THF, the reaction conditions become significantly milder and safer for large-scale operations.
Q: What purity levels can be achieved with this optimized synthetic method?
A: Through optimized crystallization steps using ethanol-water mixtures and final decolorization with activated carbon, the process consistently yields cyproheptadine hydrochloride with a purity of 99.9% as verified by HPLC analysis.
Q: Is this process suitable for industrial scale-up without specialized equipment?
A: Yes, the method is specifically designed for scalability. It removes the requirement for specialized drying of pipelines and reactors needed for Grignard reactions, allowing production in standard stainless steel reactors under nitrogen protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyproheptadine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and a proven track record in process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this low-valent titanium method are fully realized in a GMP-compliant environment. We are committed to delivering high-purity cyproheptadine hydrochloride that adheres to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex organic syntheses with the utmost attention to safety and quality, making us an ideal strategic partner for your antihistamine API requirements.
We invite you to engage with our technical procurement team to discuss how this innovative manufacturing process can optimize your supply chain and reduce overall costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our commitment to excellence in fine chemical manufacturing.
