Advanced Manufacturing of Cyproheptadine Hydrochloride for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, a challenge vividly addressed in the recent intellectual property disclosure CN110407737B. This patent introduces a revolutionary preparation method for cyproheptadine hydrochloride, a critical antihistaminic agent, which fundamentally restructures the traditional manufacturing landscape. Unlike legacy protocols that rely on hazardous reagents and complex multi-stage extractions, this novel approach leverages a streamlined Grignard-based sequence followed by a direct acid-catalyzed dehydration. For R&D directors and procurement specialists alike, this represents a significant pivot towards greener chemistry, offering a pathway that not only enhances the chemical integrity of the final active pharmaceutical ingredient but also drastically simplifies the logistical burden of raw material handling. The technical breakthrough lies in the elimination of volatile organic solvents like toluene during the intermediate isolation phase and the substitution of dangerous acetic anhydride with safer aqueous acid systems, setting a new benchmark for efficiency in pharmaceutical intermediates production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyproheptadine hydrochloride, as detailed in older patents such as US3014911, has been plagued by significant operational inefficiencies and safety hazards that hinder cost-effective commercial scale-up of complex antihistamine intermediates. The conventional route necessitates the use of glacial acetic acid and acetic anhydride for the dehydration step, alongside the handling of gaseous hydrogen chloride, which imposes stringent requirements on reactor materials and ventilation systems to prevent corrosion and exposure risks. Furthermore, the purification process traditionally relies heavily on toluene extraction, a practice that not only inflates solvent recovery costs but also introduces substantial environmental liabilities due to the toxicity and volatility of aromatic hydrocarbons. These factors collectively contribute to a fragmented production workflow where yield losses are common, often resulting in final product purities hovering around 97% with overall yields struggling to exceed 35% in comparative industrial settings, thereby eroding profit margins and supply reliability.
The Novel Approach
In stark contrast, the methodology disclosed in CN110407737B offers a transformative solution by integrating the dehydration and salt formation steps into a single, cohesive operation using concentrated hydrochloric acid in an alcoholic medium. This innovation effectively bypasses the need for anhydrous conditions and hazardous acylating agents, allowing the reaction to proceed smoothly at moderate temperatures between 80°C and 90°C. By replacing the cumbersome toluene extraction with a straightforward aqueous quench and pH adjustment strategy, the new process minimizes solvent usage and eliminates the generation of acidic wastewater streams associated with acetic anhydride hydrolysis. This streamlined workflow not only accelerates the production cycle but also ensures that the intermediate 1-methyl-4-(5-hydroxy-5-dibenzo[a,e]cycloheptatrienyl)piperidine is converted directly into the target hydrochloride salt with exceptional efficiency, demonstrating yields that can reach up to 75% before final refinement.
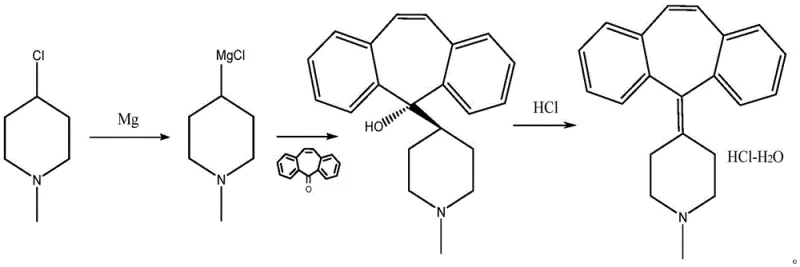
Mechanistic Insights into Grignard Addition and Acid-Catalyzed Dehydration
The core of this synthetic advancement rests on the precise control of the Grignard reagent formation and its subsequent nucleophilic attack on the dibenzo[a,e]cycloheptatriene-5-ketone backbone. In the initial phase, 1-methyl-4-chloropiperidine reacts with activated magnesium chips in tetrahydrofuran at reflux temperatures of 60-65°C to generate the organomagnesium species. This step is critical, as the quality of the Grignard reagent directly influences the impurity profile of the downstream alcohol intermediate. The patent emphasizes the importance of using freshly polished magnesium to ensure rapid initiation and complete conversion, thereby minimizing the presence of unreacted starting materials that could complicate later purification stages. Following this, the addition of the ketone is conducted under strictly controlled thermal conditions, typically between 0°C and 25°C, to manage the exothermic nature of the addition and prevent side reactions such as enolization or over-addition, which are common pitfalls in large-scale Grignard chemistry.
Following the formation of the tertiary alcohol intermediate, the mechanism shifts to an acid-catalyzed dehydration coupled with immediate salt formation. Unlike traditional methods that isolate the free base before salt formation, this process utilizes concentrated hydrochloric acid (30-36%) in an ethanol-water mixture to drive the elimination of the hydroxyl group. The protonation of the hydroxyl group creates a good leaving group (water), facilitating the formation of the stable carbocation which then loses a proton to form the conjugated tricyclic system characteristic of cyproheptadine. Simultaneously, the basic nitrogen of the piperidine ring is protonated by the excess acid, precipitating the product as the hydrochloride salt. This tandem mechanism is highly advantageous for impurity control, as the crystallization of the salt from the alcoholic solution inherently excludes non-basic organic impurities and residual magnesium salts, leading to the reported purity levels of over 99.8% after a simple recrystallization step.
How to Synthesize Cyproheptadine Hydrochloride Efficiently
Implementing this optimized synthetic route requires careful attention to thermal management and stoichiometry, particularly during the exothermic Grignard formation and the subsequent acidification steps. The process is designed to be robust, allowing for flexibility in solvent ratios while maintaining high reproducibility across different batch sizes. Operators must ensure that the addition of distilled water during the workup phase is performed slowly to control the temperature rise, preventing local overheating that could degrade the sensitive tricyclic structure. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Formation of 1-methyl-4-chloropiperidine magnesium chloride by reacting the chloropiperidine precursor with magnesium chips in tetrahydrofuran at reflux temperatures.
- Nucleophilic addition of the Grignard reagent to dibenzo[a,e]cycloheptatriene-5-ketone at controlled low temperatures to form the tertiary alcohol intermediate.
- Aqueous quenching and pH adjustment to neutral followed by crystallization to isolate the crude alcohol without toxic solvent extraction.
- Direct dehydration and salt formation using concentrated hydrochloric acid in alcoholic solution, followed by recrystallization to achieve >99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. By fundamentally altering the reagent profile, the process eliminates the dependency on controlled substances like acetic anhydride and the logistical complexities associated with transporting gaseous hydrogen chloride. This shift significantly reduces the regulatory overhead and safety compliance costs, allowing manufacturing facilities to operate with greater agility and lower insurance premiums. Furthermore, the removal of toluene from the extraction protocol simplifies waste management procedures, as the aqueous mother liquors are easier to treat and dispose of compared to halogenated or aromatic solvent waste streams, aligning perfectly with modern environmental, social, and governance (ESG) mandates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of unit operations and the reduction in raw material costs. By consolidating the dehydration and salt formation into a single step using inexpensive concentrated hydrochloric acid, the method removes the need for costly acylating agents and the energy-intensive distillation steps required to recover large volumes of toluene. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final API, effectively lowering the cost of goods sold (COGS) per kilogram. The elimination of complex purification sequences also reduces labor hours and equipment occupancy time, further enhancing the overall cost efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on commodity chemicals such as magnesium, tetrahydrofuran, and hydrochloric acid ensures a stable and resilient sourcing strategy. Unlike specialized reagents that may be subject to market volatility or geopolitical supply disruptions, these foundational chemicals are widely available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the simplified process flow reduces the number of intermediate storage requirements, allowing for a more continuous and Just-In-Time production model that can respond rapidly to fluctuations in market demand for antihistaminic medications.
- Scalability and Environmental Compliance: The inherent safety and simplicity of this route make it exceptionally well-suited for scaling from pilot batches to multi-ton commercial production. The absence of highly corrosive gases and the use of standard stainless steel reactors for the acidification step lower the capital expenditure barriers for capacity expansion. Environmentally, the process generates significantly less hazardous waste, reducing the burden on effluent treatment plants and ensuring compliance with increasingly strict discharge regulations. This 'green' advantage not only protects the company from potential fines but also enhances the brand reputation among downstream pharmaceutical partners who are increasingly prioritizing sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis protocol. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the transition from legacy methods to this improved manufacturing standard.
Q: How does the new synthesis method improve upon the traditional US3014911 process?
A: The new method eliminates the use of hazardous acetic anhydride and gaseous hydrogen chloride, replacing them with safer concentrated hydrochloric acid solutions. It also removes the need for extensive toluene extraction, significantly reducing solvent consumption and environmental burden while increasing overall yield from approximately 33% to over 60%.
Q: What purity levels can be achieved with this optimized protocol?
A: Through the implementation of controlled crystallization and a simplified refining step using ethanol or methanol aqueous solutions, the process consistently achieves purity levels exceeding 99.8%, which is superior to the 97% purity typically associated with legacy manufacturing techniques.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. By removing complex extraction steps and utilizing standard reagents like concentrated hydrochloric acid and ethanol, the operational difficulty is drastically reduced, making it highly viable for multi-ton commercial manufacturing without requiring specialized corrosion-resistant equipment for gas handling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyproheptadine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN110407737B are fully realized in practical application. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cyproheptadine hydrochloride meets the highest international pharmacopoeia standards. We are committed to delivering high-purity pharmaceutical intermediates that support the development of safe and effective antihistaminic therapies globally.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this optimized synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable manufacturing strategy available in the market.
