Scalable Synthesis of Aromatic Carboxylic Acids via Dual-Catalytic CO2 Fixation
The chemical industry is currently witnessing a paradigm shift towards sustainable carbon utilization, driven by the urgent need to reduce reliance on fossil-derived feedstocks. Patent CN110724047B introduces a groundbreaking methodology for the synthesis of aromatic carboxylic acid compounds, leveraging carbon dioxide (CO2) as a direct C1 building block. This technology represents a significant advancement over traditional oxidation or stoichiometric metallation routes, offering a pathway to high-value fine chemicals with a markedly reduced environmental footprint. By employing a sophisticated dual-catalytic system involving copper and palladium, the process achieves efficient CO2 fixation under relatively mild thermal conditions. For R&D directors and process chemists, this patent provides a robust framework for accessing diverse benzoic acid derivatives, which are critical scaffolds in the manufacture of active pharmaceutical ingredients (APIs) and agrochemical intermediates. The ability to transform a greenhouse gas into valuable molecular architectures not only aligns with green chemistry principles but also opens new avenues for cost-effective manufacturing strategies in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of aryl carboxylic acids has relied heavily on two primary strategies: the oxidation of alkylbenzenes or the carboxylation of organometallic reagents. The oxidation route, while established for simple substrates like toluene, suffers from poor selectivity when dealing with complex molecules containing oxidizable functional groups, often leading to over-oxidation or ring degradation. Alternatively, the classical carboxylation using Grignard or organolithium reagents requires the generation of highly reactive aryl-metal species. These reagents are notoriously sensitive to moisture and oxygen, necessitating stringent anhydrous conditions and cryogenic temperatures that are energy-intensive and difficult to maintain on a large scale. Furthermore, these traditional methods typically demand stoichiometric or excess amounts of metal reagents, resulting in substantial quantities of metal salt waste during the quenching and workup phases. This not only increases the cost of raw materials but also creates significant challenges in waste disposal and environmental compliance, making these legacy processes increasingly untenable for modern sustainable manufacturing goals.
The Novel Approach
In stark contrast, the method disclosed in CN110724047B utilizes a catalytic cross-coupling strategy that circumvents the need for pre-formed organometallic reagents. The core innovation lies in the in situ generation of a reactive silyl species from CO2 and a hydrosilane, mediated by a copper catalyst, which then participates in a palladium-catalyzed coupling with readily available aryl halides. This approach allows for the direct use of stable and inexpensive aryl bromides or iodides as starting materials, significantly simplifying the supply chain logistics. The reaction conditions are remarkably mild, typically operating between 60°C and 150°C, which reduces energy consumption compared to cryogenic protocols. Moreover, the use of catalytic amounts of transition metals rather than stoichiometric quantities minimizes metal contamination in the final product, a critical parameter for pharmaceutical applications. This novel pathway effectively decouples the synthesis from the limitations of substrate oxidation states, enabling the preservation of sensitive functional groups that would otherwise be compromised in traditional oxidative processes.
Mechanistic Insights into Cu/Pd Dual-Catalytic Carboxylation
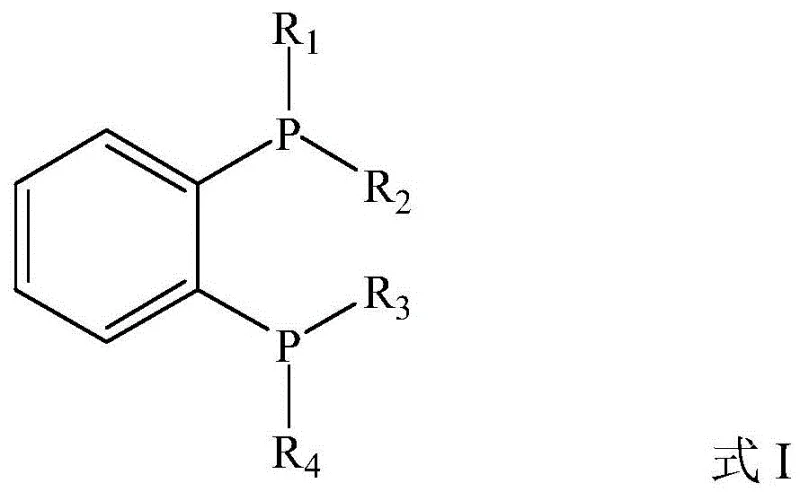
The mechanistic elegance of this transformation relies on the synergistic interplay between two distinct catalytic cycles. The first cycle involves the activation of the inert CO2 molecule. Under the influence of a copper catalyst coordinated with a specific bisphosphine ligand (Formula I), carbon dioxide undergoes hydrosilylation with a silane reagent such as polymethylhydrosiloxane (PMHS). This step generates a silyl formate or related silyl-carboxylate intermediate, effectively "fixing" the CO2 into a more reactive organic species. The choice of ligand is paramount here; the steric and electronic properties of the bisphosphine ligand modulate the electron density at the copper center, facilitating the insertion of CO2 into the Cu-H or Cu-Si bond. This activation step is crucial because free CO2 is thermodynamically stable and kinetically inert, requiring significant energy or highly reactive partners to participate in bond formation.
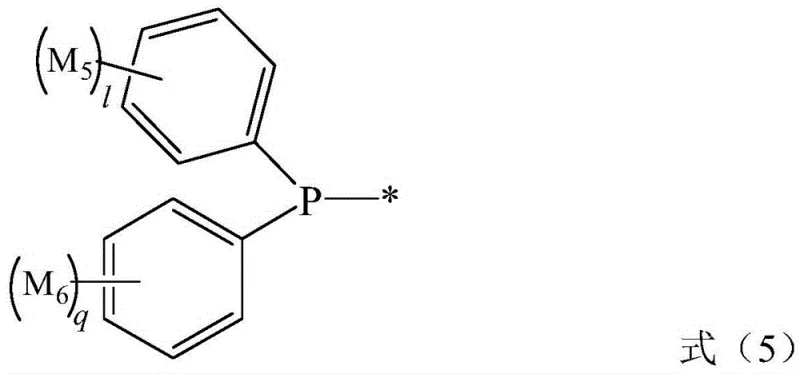
Following the activation of CO2, the second catalytic cycle takes over, driven by a palladium catalyst system. The aryl halide substrate undergoes oxidative addition to the palladium(0) species, forming an aryl-palladium(II) complex. This electrophilic intermediate then reacts with the nucleophilic silyl-carboxylate species generated in the first cycle. Through a transmetallation step, the carboxylate group is transferred to the palladium center, followed by reductive elimination to release the silyl ester of the target aromatic carboxylic acid. Finally, hydrolysis during the workup yields the free carboxylic acid. This dual-catalyst design ensures that the highly reactive intermediates are generated and consumed in situ, minimizing side reactions such as homocoupling of the aryl halide. The precise control over the catalytic environment also contributes to superior impurity profiles, as the mild conditions prevent the decomposition of thermally labile functional groups often present in complex drug intermediates.
How to Synthesize Aromatic Carboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and the maintenance of an inert atmosphere to protect the catalytic species. The process begins with the preparation of the CO2-activated species in a dedicated step, ensuring complete conversion of the silane before introducing the sensitive palladium catalyst. This staged approach prevents potential deactivation of the palladium center by the silane or copper species. Operators must strictly adhere to the temperature gradients specified, starting at lower temperatures for CO2 activation and ramping up for the cross-coupling event to maximize turnover frequency. The versatility of the method allows for a wide range of aryl halides, including heteroaromatics and poly-halogenated benzenes, to be converted into their corresponding acids with high fidelity. For detailed procedural specifics regarding molar ratios, solvent choices, and purification techniques, please refer to the standardized synthesis guide below.
- Activate carbon dioxide by heating it with a hydrosilane and a copper catalyst complex in an organic solvent A at 50-70°C to form a silyl ester intermediate.
- Introduce the aryl halide substrate, a palladium catalyst complex, and an organic base into the reaction mixture, then heat to 60-150°C to facilitate the cross-coupling reaction.
- Quench the reaction with an alkaline aqueous solution, separate the layers, acidify the aqueous phase to precipitate the product, and extract with organic solvent E.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative benefits that directly impact the bottom line and operational resilience. The shift from stoichiometric metal reagents to catalytic systems fundamentally alters the cost structure of aromatic carboxylic acid manufacturing. By eliminating the need for expensive and hazardous organolithium or Grignard reagents, manufacturers can achieve substantial cost savings on raw material acquisition. Furthermore, the stability of the starting materials—simple aryl halides and commodity silanes—ensures a reliable supply chain that is less susceptible to the volatility associated with specialized organometallic suppliers. This stability translates into enhanced supply security, allowing production planners to forecast inventory needs with greater confidence and reduce the safety stock buffers typically required for hazardous reagents.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the drastic reduction in waste treatment costs and raw material efficiency. Traditional methods generate massive amounts of inorganic salt waste during the quenching of excess metal reagents, which incurs high disposal fees and environmental levies. In contrast, this catalytic method produces minimal inorganic waste, significantly lowering the operational expenditure related to effluent treatment. Additionally, the high atom economy of using CO2 as a C1 source means that the carbon input is essentially free or very low-cost compared to purchasing pre-functionalized carboxylation reagents. The ability to recover and recycle the organic solvents used in the biphasic workup further contributes to the overall cost efficiency, making the process economically viable for large-scale commodity chemical production as well as high-value specialty synthesis.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable aryl halides mitigates the risks associated with the storage and transport of pyrophoric materials. Aryl bromides and iodides are shelf-stable solids or liquids that do not require specialized cold-chain logistics or inert atmosphere storage facilities. This simplification of logistics reduces the total cost of ownership and minimizes the risk of supply disruptions caused by transportation regulations on hazardous goods. Moreover, the robustness of the catalyst system means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain fluctuations. For global supply chain heads, this translates to a more resilient manufacturing network capable of maintaining continuous production even during periods of raw material scarcity or logistical bottlenecks.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this method is inherently designed for scalability. The mild reaction temperatures (60-150°C) are compatible with standard stainless steel reactors, eliminating the need for exotic alloys or cryogenic cooling infrastructure. This compatibility accelerates the technology transfer from laboratory to pilot plant and finally to commercial production. From an environmental compliance standpoint, the process aligns perfectly with increasingly stringent global regulations on volatile organic compounds (VOCs) and heavy metal discharge. The reduced metal loading in the final product simplifies the purification process, often removing the need for expensive scavenging resins to meet ppm-level metal specifications required by regulatory bodies for pharmaceutical ingredients. This inherent "greenness" serves as a strong value proposition for customers seeking to reduce their Scope 3 emissions and improve their sustainability ratings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CO2 fixation technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these nuances is essential for project managers evaluating the feasibility of adopting this route for their specific product portfolios. The answers reflect the consensus of the technical team regarding the practical application of the dual-catalytic system in an industrial setting.
Q: What are the safety advantages of this CO2 fixation method compared to traditional Grignard carboxylation?
A: Traditional methods often require stoichiometric amounts of highly reactive organolithium or Grignard reagents, which pose significant safety risks regarding moisture sensitivity and exothermic reactions. This patented method utilizes stable aryl halides and mild hydrosilanes under catalytic conditions, eliminating the need for hazardous stoichiometric metal reagents and significantly improving operational safety.
Q: Can this method tolerate functional groups like nitro or aldehyde groups on the aromatic ring?
A: Yes, the method demonstrates excellent functional group tolerance. Experimental data within the patent indicates successful carboxylation of substrates containing sensitive groups such as nitro (-NO2), aldehyde (-CHO), and ketone moieties, yielding products like 4-nitrobenzoic acid and 4-formylbenzoic acid with high purity and yield.
Q: Is this process suitable for large-scale industrial production of pharmaceutical intermediates?
A: The process is highly amenable to scale-up due to its use of common, stable catalysts (Cu and Pd complexes) and mild reaction conditions (50-150°C). The avoidance of cryogenic temperatures and pyrophoric reagents simplifies reactor requirements, while the straightforward aqueous workup facilitates efficient isolation of the final carboxylic acid product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic methodologies to maintain a competitive edge in the global fine chemical market. Our technical team has extensively evaluated the dual-catalytic carboxylation process described in CN110724047B and confirmed its potential for delivering high-purity aromatic carboxylic acids with exceptional consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging this advanced chemistry to provide our partners with superior quality intermediates that drive their own downstream success.
We invite you to collaborate with us to explore how this sustainable synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our aromatic carboxylic acid portfolio and to discuss route feasibility assessments for your custom synthesis projects. Together, we can build a more sustainable and efficient future for the chemical industry.
