Advanced Photoredox Decarboxylative Coupling for Scalable Aryl Ketone Manufacturing
The landscape of organic synthesis is continuously evolving towards more sustainable and atom-economical processes, particularly in the construction of carbonyl-containing scaffolds which are ubiquitous in active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN108912042B, which discloses a novel method for the direct preparation of aromatic ketones from aromatic carboxylic acids and olefins. This technology leverages visible-light photoredox catalysis to achieve decarboxylative coupling under exceptionally mild conditions, bypassing the need for harsh activating agents or stoichiometric organometallic reagents. By utilizing an Iridium-based photocatalyst system in a biphasic dichloromethane-water medium, this process offers a robust pathway for generating high-purity aryl ketones. For R&D directors and process chemists, this represents a paradigm shift from traditional acylation strategies, offering enhanced safety profiles and reduced environmental impact while maintaining high chemical selectivity.
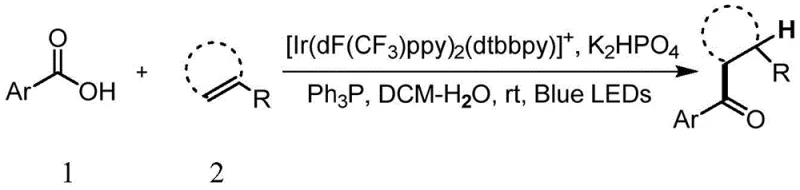
The limitations of conventional methods for ketone synthesis have long been a bottleneck in efficient process development. Traditional approaches often rely on Friedel-Crafts acylation or the coupling of nucleophilic organometallic reagents with electrophilic carboxylic acid derivatives such as acid chlorides or anhydrides. These classical routes frequently necessitate harsh reaction conditions, including strong Lewis acids, elevated temperatures, and strictly anhydrous environments, which can lead to poor chemoselectivity and the degradation of sensitive functional groups. Furthermore, the pre-activation of carboxylic acids into acid chlorides generates stoichiometric amounts of corrosive byproducts and requires additional synthetic steps, increasing both the operational complexity and the waste burden. In contrast, the novel approach described in the patent utilizes readily available aromatic carboxylic acids and olefins directly. This strategy eliminates the need for pre-functionalization of the acid component, thereby streamlining the synthetic sequence. The reaction proceeds at room temperature under blue LED irradiation, utilizing triphenylphosphine as a benign terminal reductant and dipotassium hydrogen phosphate as a mild base. This transition from activated electrophiles to stable feedstock chemicals significantly simplifies the operational workflow and enhances the overall safety of the manufacturing process.
Mechanistic Insights into Iridium-Catalyzed Photoredox Decarboxylation
The core of this transformative methodology lies in the sophisticated interplay between the photocatalyst and the substrate under visible light irradiation. The reaction employs [Ir(dF(CF3)ppy)2(dtbbpy)]PF6 as the photocatalyst, a complex known for its strong oxidizing power in the excited state and long-lived triplet state. Upon irradiation with blue LEDs, the Iridium center is excited to a high-energy state capable of engaging in single-electron transfer (SET) processes. The mechanism initiates with the oxidative quenching of the excited photocatalyst by the carboxylate anion, generated in situ from the aromatic acid and the phosphate base. This single-electron oxidation triggers the rapid decarboxylation of the carboxylate radical, releasing carbon dioxide and generating a nucleophilic benzyl or aryl radical species. This carbon-centered radical then adds across the double bond of the olefin partner, forming a new C-C bond and a transient alkyl radical intermediate.
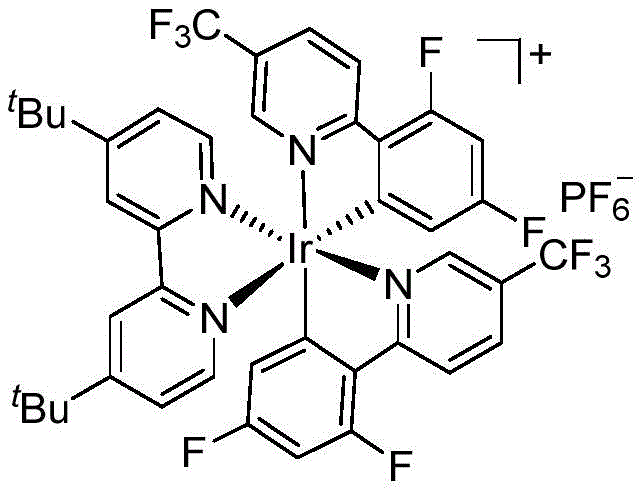
Following the radical addition, the resulting alkyl radical is oxidized by the reduced form of the Iridium catalyst or interacts with the phosphine reductant system to regenerate the ground-state catalyst and yield the final cationic or neutral ketone product after hydrolysis or elimination steps. The presence of triphenylphosphine is crucial as it acts as a sacrificial electron donor or oxygen scavenger, ensuring the catalytic cycle continues efficiently without being inhibited by oxygen or radical termination side reactions. This intricate redox manifold allows for the precise control of radical intermediates, minimizing homocoupling byproducts and ensuring high regioselectivity. The biphasic solvent system of DCM and water further aids in the solubility of inorganic bases and the stabilization of ionic intermediates, contributing to the robustness of the reaction. Understanding these mechanistic nuances is vital for scaling up the process, as it highlights the importance of maintaining strict inert conditions and optimal light penetration to maximize quantum efficiency and yield.
How to Synthesize Aryl Ketones Efficiently
Implementing this photocatalytic protocol requires careful attention to reaction setup and parameter control to ensure reproducibility and high yields. The process begins with the precise weighing of the aromatic carboxylic acid substrate and the olefin coupling partner, typically in a molar ratio ranging from 1:1 to 1:5, with 1:1.5 often proving optimal for balancing conversion and material efficiency. The photocatalyst is added at a low loading of 1 mol%, demonstrating the high turnover number achievable with this Iridium complex. Alongside the catalyst, triphenylphosphine (1.2 equivalents) and dipotassium hydrogen phosphate (20 mol%) are introduced to the reaction vessel containing a mixture of dichloromethane and water. The detailed standardized synthesis steps are outlined below.
- Prepare the reaction mixture by combining aromatic carboxylic acid, olefin, triphenylphosphine, dipotassium hydrogen phosphate, and the iridium photocatalyst in a DCM-water solvent system.
- Degas the reaction vessel thoroughly using a vacuum line and backfill with argon gas to maintain an inert atmosphere essential for radical stability.
- Irradiate the mixture with blue LEDs at room temperature for 36 to 60 hours, followed by standard aqueous workup and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this photocatalytic technology offers substantial strategic benefits for supply chain optimization and cost management in fine chemical manufacturing. The primary advantage lies in the utilization of commodity chemicals as starting materials. Aromatic carboxylic acids and styrene derivatives are produced on a massive industrial scale, ensuring a stable and reliable supply chain with minimal price volatility compared to specialized organometallic reagents or activated acid halides. By eliminating the need for pre-activation steps such as thionyl chloride treatment, the process reduces the number of unit operations required, which directly translates to lower capital expenditure on equipment and reduced labor costs. Furthermore, the mild reaction conditions operate at ambient temperature and pressure, removing the need for energy-intensive heating or cryogenic cooling systems, thereby significantly lowering the utility costs associated with production.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the atom-economical nature of the decarboxylative coupling. Since the carboxylic acid is used directly without conversion to acid chlorides, the costs associated with activating agents and the disposal of corrosive byproducts like HCl or SO2 are completely avoided. Although the Iridium catalyst is a precious metal, its usage at only 1 mol% combined with the potential for recycling or recovery makes the overall catalyst cost negligible relative to the value of the pharmaceutical intermediate produced. Additionally, the use of triphenylphosphine as a reductant is far more cost-effective than using silanes or metallic powders often found in alternative radical protocols. This streamlined approach minimizes raw material consumption and waste treatment expenses, leading to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials mitigates risks associated with the storage and handling of hazardous reagents. Acid chlorides, for instance, are moisture-sensitive and require specialized storage infrastructure, whereas carboxylic acids are generally stable solids that can be stored in standard warehouses. This stability simplifies logistics and reduces the risk of supply disruptions due to reagent degradation. Moreover, the broad substrate scope demonstrated in the patent means that a single platform technology can be applied to synthesize a wide variety of ketone intermediates. This versatility allows manufacturers to respond quickly to changing market demands for different API precursors without needing to retool facilities or qualify entirely new synthetic routes, thereby enhancing the agility and resilience of the supply chain.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration limits, but advancements in flow chemistry and LED reactor design have made this increasingly feasible. The mild conditions of this reaction reduce the thermal load on reactors, allowing for safer scale-up with lower risks of runaway exotherms. From an environmental standpoint, the process aligns with green chemistry principles by using water as a co-solvent and generating carbon dioxide as the only stoichiometric byproduct from the decarboxylation step. This reduces the E-factor (mass of waste per mass of product) significantly compared to traditional methods. The absence of heavy metal waste streams typical of stoichiometric organometallic couplings simplifies wastewater treatment and ensures compliance with stringent environmental regulations, facilitating smoother regulatory approvals for commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this photocatalytic decarboxylative coupling technology. These insights are derived directly from the experimental data and scope studies presented in the patent documentation, providing clarity on reaction parameters and substrate compatibility.
Q: What are the primary advantages of this photocatalytic method over traditional Friedel-Crafts acylation?
A: Unlike traditional methods requiring harsh Lewis acids and pre-activated acid chlorides, this protocol utilizes stable carboxylic acids and olefins under mild, neutral conditions, significantly reducing hazardous waste and improving functional group compatibility.
Q: Is the Iridium catalyst loading economically viable for large-scale production?
A: Yes, the process operates efficiently with a low catalyst loading of approximately 1 mol%, and the use of inexpensive triphenylphosphine as a terminal reductant helps maintain overall process cost-effectiveness despite the use of a precious metal catalyst.
Q: What is the substrate scope regarding heterocyclic compounds?
A: The method exhibits broad substrate tolerance, successfully coupling various substituted benzoic acids with olefins containing pyridyl, quinolyl, and other heterocyclic moieties, making it highly suitable for synthesizing complex pharmaceutical scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Ketone Supplier
The technological advancements described in patent CN108912042B underscore the immense potential of photoredox catalysis in modernizing the synthesis of complex organic molecules. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such innovative methodologies to meet the evolving needs of the global pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into robust manufacturing processes. Our facility is equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of handling sensitive catalytic systems, guaranteeing stringent purity specifications for every batch of aryl ketone intermediates we produce.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, evaluating the feasibility of adapting this decarboxylative protocol to your existing supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can drive efficiency and innovation in the production of high-value pharmaceutical intermediates.
