Advanced Manufacturing of High-Purity Tenofovir Intermediates for Global API Production
Introduction to Next-Generation Tenofovir Synthesis
The global demand for effective antiretroviral therapies continues to drive innovation in the manufacturing of key pharmaceutical intermediates, particularly for treatments targeting HIV and chronic Hepatitis B. Patent CN102060876A introduces a transformative preparation method for tenofovir and its prodrug forms, addressing critical bottlenecks in existing supply chains. This technology represents a significant leap forward in process chemistry, moving away from hazardous, low-yielding protocols toward a streamlined, high-purity manufacturing workflow. By leveraging a unique protection-deprotection strategy involving p-toluenesulfonyl groups, the process achieves superior control over impurity profiles, specifically targeting the persistent issue of monoethyl ester residues. For R&D directors and procurement leaders, understanding this patented route is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
The clinical significance of tenofovir disoproxil fumarate (TDF) cannot be overstated, given its status as a first-line therapy with a favorable resistance profile. However, the complexity of synthesizing the acyclic nucleotide phosphonate core has historically limited production capacity and inflated costs. The methodology outlined in this patent utilizes readily available starting materials such as adenine and (R)-propylene carbonate, bypassing the need for complex chiral pool precursors that often constrain supply. This shift not only democratizes access to raw materials but also stabilizes the supply chain against fluctuations in specialty chemical markets. As we delve deeper into the technical specifics, it becomes clear that this approach offers a robust solution for cost reduction in API manufacturing while maintaining stringent regulatory standards for purity and safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tenofovir has been plagued by reliance on dangerous and expensive reagents that complicate scale-up and compromise safety. Traditional routes frequently employ n-butyllithium as a base for the alkylation step, a reagent that is pyrophoric and requires storage at cryogenic temperatures, introducing severe operational hazards and necessitating specialized infrastructure. Furthermore, the deprotection of phosphonate esters in legacy processes often utilizes trimethylbromosilane (TMSBr), a costly reagent that frequently leads to incomplete hydrolysis. This incomplete reaction results in the retention of monoethyl ester impurities, which are structurally similar to the target molecule and notoriously difficult to separate, thereby capping the overall yield and purity of the final active pharmaceutical ingredient. These factors collectively drive up the cost of goods sold and create significant bottlenecks for commercial scale-up of complex nucleotide analogues.
The Novel Approach
In stark contrast, the methodology disclosed in CN102060876A pioneers a safer and more efficient pathway by substituting hazardous reagents with benign alternatives and introducing a strategic amino protection step. Instead of cryogenic n-butyllithium, the process utilizes potassium alkoxides, such as potassium tert-butoxide, which operate effectively at ambient or mildly controlled temperatures, drastically reducing energy consumption and safety risks. The core innovation lies in the temporary protection of the exocyclic amine on the adenine ring with a p-toluenesulfonyl (Ts) group prior to hydrolysis. This protective shield allows the use of strong mineral acids like concentrated hydrochloric acid or hydrobromic acid to aggressively cleave the ethyl esters from the phosphonate group without degrading the sensitive heterocyclic base. This ensures complete conversion to the free phosphonic acid, effectively eliminating the monoethyl ester impurity and delivering a product of exceptional purity suitable for direct salt formation.
Mechanistic Insights into Ts-Protection and Acid Hydrolysis
The chemical elegance of this synthesis lies in its precise management of chemoselectivity during the hydrolysis phase. In standard nucleotide synthesis, the exocyclic amine of adenine is nucleophilic and susceptible to side reactions under harsh acidic conditions. By installing the p-toluenesulfonyl group in an earlier step, the electron density on the nitrogen is significantly reduced, rendering the ring system inert to the aggressive conditions required for phosphonate ester cleavage. This allows the manufacturer to push the hydrolysis reaction to completion using inexpensive strong acids, a feat that is difficult to achieve with unprotected intermediates where milder, more expensive reagents like TMSBr are typically mandated to preserve the integrity of the base. The result is a cleaner reaction profile with fewer byproducts, simplifying downstream purification and crystallization processes.
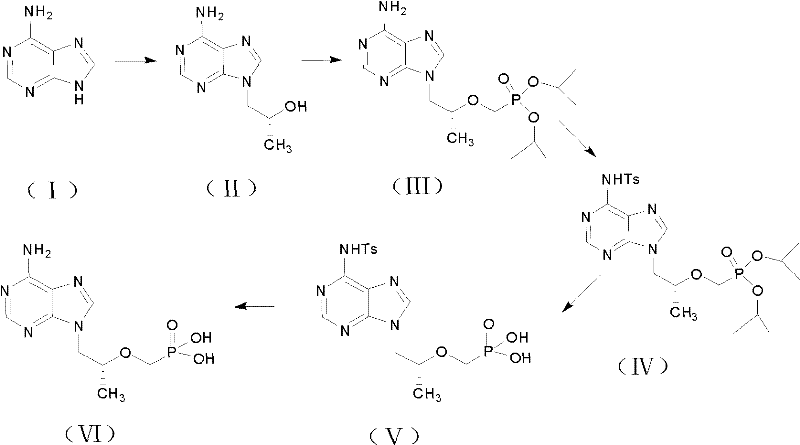
Following the hydrolysis, the removal of the Ts protecting group is achieved under mild alkaline conditions using thiophenol as a nucleophile. This step is highly specific and proceeds with high efficiency, regenerating the free amine necessary for biological activity without affecting the newly formed phosphonic acid moiety. The entire sequence demonstrates a sophisticated understanding of orthogonal protection strategies, ensuring that each functional group is manipulated only when intended. For technical teams evaluating process feasibility, this mechanistic robustness translates to higher reproducibility and lower batch-to-batch variability. The ability to monitor reaction progress via HPLC at each stage, particularly observing the disappearance of the diethyl ester peak and the emergence of the free acid, provides clear process control points that are vital for Good Manufacturing Practice (GMP) compliance.
How to Synthesize Tenofovir Efficiently
The synthesis of high-purity tenofovir via this patented route involves a logical sequence of condensation, coupling, protection, hydrolysis, and deprotection steps that can be executed in standard stainless steel reactors. The process begins with the alkylation of adenine using (R)-propylene carbonate in the presence of a strong base catalyst like potassium hydroxide or sodium hydroxide in polar aprotic solvents such as DMF. Following isolation of the hydroxy-propyl intermediate, the phosphonate side chain is installed using diethyl p-toluenesulfonyloxyphosphonate mediated by potassium tert-butoxide. The subsequent protection of the amine and acid hydrolysis steps are critical for ensuring the removal of ethyl esters, followed by the final thiol-mediated deprotection to yield the target molecule. For a detailed breakdown of reaction parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Condense adenine with (R)-propylene carbonate under strong base catalysis to form (R)-9-(2-hydroxypropyl)adenine.
- Couple the hydroxy-intermediate with diethyl p-toluenesulfonyloxyphosphonate using potassium alkoxide to install the phosphonate side chain.
- Protect the exocyclic amine with p-toluenesulfonyl chloride, followed by strong acid hydrolysis to cleave ethyl esters, and finally remove the protecting group with thiophenol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By eliminating the need for cryogenic reagents and expensive silylating agents, the operational expenditure (OpEx) associated with raw material procurement and waste disposal is significantly reduced. The substitution of n-butyllithium with potassium alkoxides removes the requirement for specialized low-temperature storage and handling equipment, thereby lowering capital expenditure (CapEx) for facility upgrades. Moreover, the use of common mineral acids for hydrolysis instead of proprietary silicon reagents creates a more transparent and competitive supply market for inputs, reducing the risk of vendor lock-in and price volatility. These factors combine to create a manufacturing environment that is not only safer but also economically superior for long-term production planning.
- Cost Reduction in Manufacturing: The elimination of high-cost reagents such as trimethylbromosilane and n-butyllithium results in a drastic reduction in direct material costs. Additionally, the ability to recycle solvents like toluene and dichloromethane, which are used extensively in the extraction and crystallization steps, further enhances the economic efficiency of the process. The improved yield resulting from complete hydrolysis means less raw material is wasted on off-spec batches, maximizing the output per kilogram of input. This holistic approach to cost optimization ensures that the final API can be produced at a highly competitive price point without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as adenine, propylene carbonate, and hydrochloric acid ensures a stable and diversified supply base. Unlike specialty catalysts that may have single-source suppliers and long lead times, the reagents used in this process are widely available from multiple global vendors. This diversification mitigates the risk of supply disruptions due to geopolitical issues or production outages at specific facilities. Furthermore, the robustness of the reaction conditions allows for flexible scheduling and faster turnaround times between batches, enabling manufacturers to respond more agilely to fluctuations in market demand for antiviral medications.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, avoiding unit operations that are difficult to translate from pilot plant to commercial scale, such as cryogenic reactions or high-pressure hydrogenations. The mild reaction temperatures and atmospheric pressure operations simplify engineering requirements and reduce energy consumption. From an environmental standpoint, the reduction in hazardous waste generation, particularly the avoidance of silicon-containing byproducts and lithium salts, simplifies wastewater treatment and disposal. The ability to recover and reuse solvents aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this method compares to industry standards. Understanding these nuances is critical for technical procurement teams evaluating potential technology transfers or licensing opportunities. The answers highlight the specific improvements in safety, purity, and cost-efficiency that define the value proposition of this manufacturing approach.
Q: How does this patent improve impurity control compared to conventional TMSBr hydrolysis?
A: Conventional methods using trimethylbromosilane often result in incomplete hydrolysis, leaving behind monoethyl ester impurities that are difficult to remove. This patented process utilizes a p-toluenesulfonyl (Ts) protection strategy on the adenine amine, which allows for vigorous hydrolysis using strong mineral acids like HCl or HBr. This ensures complete cleavage of the ethyl groups from the phosphonate moiety while the amine is shielded, significantly reducing the monoethyl ester content and enhancing overall product purity.
Q: What are the safety advantages of replacing n-butyllithium with potassium alkoxides?
A: Traditional routes often rely on n-butyllithium, a pyrophoric reagent requiring cryogenic storage and specialized handling equipment, which poses significant safety risks and increases operational costs. The novel method described in CN102060876A replaces this hazardous reagent with potassium alkoxides (such as potassium tert-butoxide) which can be handled at ambient or mildly controlled temperatures. This substitution drastically reduces the risk of fire and explosion, simplifies the reactor requirements, and lowers the barrier for safe commercial scale-up.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrialization. It avoids extreme conditions such as ultra-low temperatures or high-pressure hydrogenation. Furthermore, the solvents used, including DMF, toluene, and dichloromethane, are standard industrial solvents that can be efficiently recovered and recycled. The use of inexpensive, readily available raw materials like adenine and propylene carbonate, combined with robust reaction conditions, makes this route highly scalable and economically viable for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN102060876A can be seamlessly transitioned from the laboratory to the manufacturing floor. We are committed to delivering high-purity tenofovir intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance ensures that every batch delivered to our partners adheres to the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your manufacturing strategy, reduce costs, and secure a reliable supply of high-quality antiviral intermediates for the future.
