Advanced Synthesis of Maraviroc Intermediates via Gamma-Azide Precursors for Commercial Scale-Up
Introduction to Next-Generation Maraviroc Synthesis
The pharmaceutical landscape for HIV treatment continues to evolve, demanding more efficient and cost-effective pathways for critical antiretroviral agents like Maraviroc. Patent CN113549070A introduces a groundbreaking preparation method for Maraviroc and its derivatives, shifting the paradigm from traditional ester-based syntheses to a versatile gamma-position azide strategy. This innovation addresses long-standing challenges in organic synthesis by utilizing azide compounds containing halogens at the gamma position as key starting materials. The disclosed methodology enables a series of non-sequential reactions including reduction, hydrolysis, substitution, and condensation, ultimately yielding the target compound with exceptional purity. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for CCR5 antagonists, offering a route that is not only chemically robust but also operationally simpler than prior art.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Maraviroc has relied on pathways starting from 3-amino-3-phenyl methyl propionate, a route fraught with inefficiencies that hinder cost reduction in pharmaceutical manufacturing. As illustrated in the background art, the conventional process necessitates the initial protection of the amino group to prevent unwanted side reactions during subsequent transformations. Following protection, the ester group must be carefully reduced to an aldehyde, a step that often requires stringent control to avoid over-reduction to the alcohol. The resulting aldehyde is then reductively coupled with the complex tropane derivative, followed by a final deprotection step and acylation. This multi-step sequence involves significant handling of protected intermediates, increasing both material costs and processing time. Furthermore, the reliance on protection-deprotection chemistry introduces additional impurities that complicate downstream purification, thereby impacting the overall yield and economic viability of large-scale production.
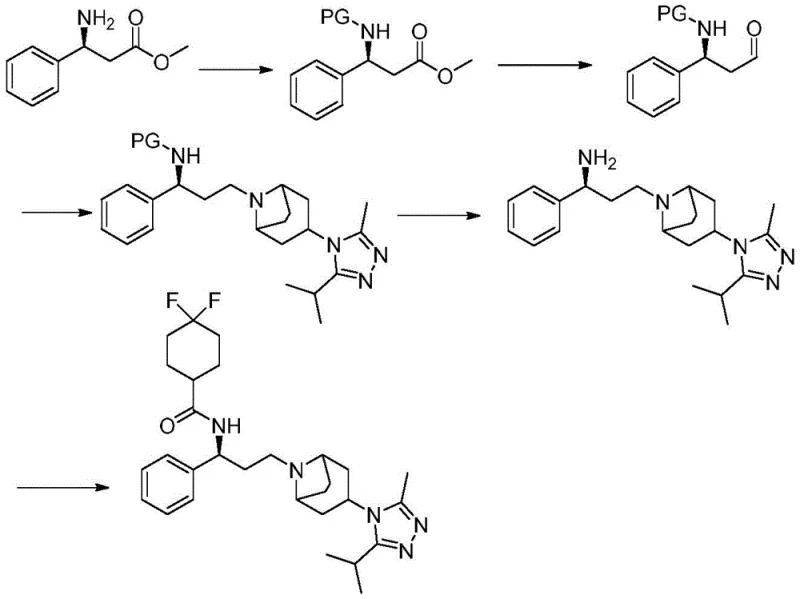
The Novel Approach
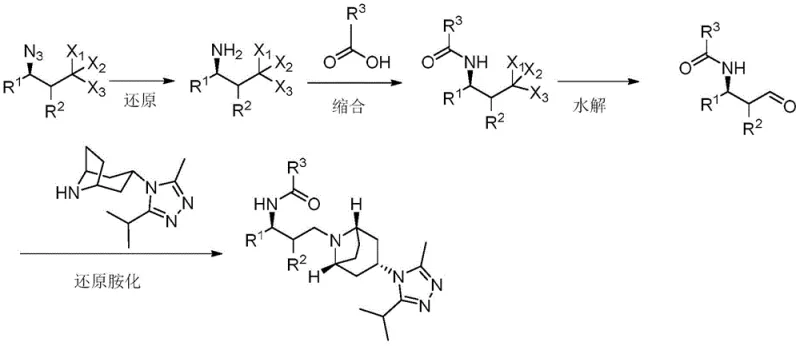
In stark contrast, the novel approach detailed in the patent leverages the unique reactivity of gamma-position azide compounds to streamline the entire synthetic sequence. By initiating the synthesis with an azide precursor, the need for early-stage amine protection is effectively eliminated, as the azide group serves as a stable, masked equivalent of the amine. This strategic shift allows for a more direct construction of the carbon-nitrogen framework. The process facilitates a flexible order of operations where reduction, hydrolysis, and condensation can be tailored based on specific substrate availability. For instance, Method A demonstrates a pathway where the azide is first reduced to an amine, condensed with a carboxylic acid, hydrolyzed to an aldehyde, and finally coupled with the tropane moiety. This modularity not only simplifies the workflow but also enhances the adaptability of the process to different derivative structures, making it a superior choice for reliable API intermediate supplier operations seeking to diversify their portfolio.
Mechanistic Insights into Gamma-Azide Functionalization
The core of this technological advancement lies in the sophisticated manipulation of the azide and halogen functionalities within the gamma-position scaffold. The reduction of the azide group, typically achieved using lithium aluminum hydride or sodium borohydride, proceeds with high chemoselectivity to generate the primary amine without disturbing other sensitive moieties such as the halogenated alkyl chain. This amine then undergoes condensation with substituted carboxylic acids, such as 4,4-difluorocyclohexanecarboxylic acid, to form a stable amide bond. A critical mechanistic feature is the subsequent hydrolysis step, where silver nitrate acts as a promoter to convert the terminal halogenated group into an aldehyde. This transformation is pivotal, as it generates the electrophilic center required for the final reductive amination. The use of silver salts in an acetone-water system ensures mild conditions that preserve the stereochemical integrity of the adjacent chiral centers, a crucial factor for maintaining the biological activity of the final antiviral agent.
Furthermore, the final reductive amination step utilizes sodium triacetoxyborohydride to couple the newly formed aldehyde with the bulky (1R,3S,5S)-3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octane. This reaction is highly efficient, driven by the formation of an iminium ion intermediate that is rapidly reduced to the secondary amine. The mechanism avoids the formation of stable emulsions often seen in aqueous workups, facilitating easier phase separation. From an impurity control perspective, the linear nature of this synthesis minimizes the generation of branched byproducts. The specific choice of reagents ensures that side reactions, such as the reduction of the triazole ring or the amide bond, are thermodynamically unfavorable, resulting in a crude product profile that is significantly cleaner than those obtained from traditional ester-based routes.
How to Synthesize Maraviroc Efficiently
The synthesis of Maraviroc via this patented gamma-azide route offers a standardized protocol that balances reaction efficiency with operational safety. The process begins with the careful reduction of the chiral azide starting material, followed by amidation and selective hydrolysis to install the aldehyde functionality. The final coupling with the tropane derivative completes the molecular architecture. Detailed standard operating procedures for each reaction step, including precise stoichiometric ratios, temperature controls, and workup protocols, are essential for reproducing the high yields reported in the patent examples. For a comprehensive guide on executing these transformations in a pilot or production setting, please refer to the structured synthesis guide below.
- Reduce the gamma-position azide compound containing halogen using lithium aluminum hydride or sodium borohydride to generate the primary amine intermediate.
- Perform a condensation reaction between the resulting amine and a substituted carboxylic acid (e.g., 4,4-difluorocyclohexanecarboxylic acid) to form the amide structure.
- Execute a hydrolysis reaction using silver nitrate in an acetone/water system to convert the halogenated moiety into a critical aldehyde group.
- Complete the synthesis via reductive amination with (1R,3S,5S)-3-(3-isopropyl-5-methyl-4H-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octane using sodium triacetoxyborohydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this gamma-azide synthesis route presents compelling economic and logistical benefits that directly impact the bottom line. The elimination of amine protection and deprotection steps fundamentally alters the cost structure of the synthesis by removing the requirement for expensive protecting group reagents and the associated waste disposal costs. This simplification translates into substantial cost savings in raw material procurement and reduces the overall number of unit operations required per batch. Additionally, the mild reaction conditions, typically ranging from 0°C to 50°C, significantly lower energy consumption compared to processes requiring cryogenic temperatures or prolonged reflux, further enhancing the economic feasibility of commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway drastically reduces the consumption of auxiliary chemicals and solvents associated with protection-deprotection cycles. By avoiding these extra steps, manufacturers can achieve a leaner process with higher atom economy. The use of common reducing agents like lithium aluminum hydride and sodium borohydride, which are readily available in bulk quantities, ensures stable pricing and reduces dependency on specialized catalysts. This operational efficiency allows for a more competitive pricing model for the final API, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically the gamma-position azide compounds and the tropane derivative, are accessible through established chemical supply networks, mitigating the risk of raw material shortages. The robustness of the reaction conditions means that the process is less susceptible to variations in utility supply, such as cooling water fluctuations, ensuring consistent batch-to-batch quality. This reliability is critical for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The simplicity of the workup procedures, which primarily involve extraction and chromatography, facilitates easy scaling from laboratory to industrial volumes without the need for specialized equipment. The reduction in synthetic steps inherently lowers the volume of chemical waste generated, aligning with increasingly stringent environmental regulations and sustainability goals. This eco-friendly profile not only reduces disposal costs but also enhances the corporate social responsibility standing of the manufacturing entity, making it a preferred partner for green-conscious drug developers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Maraviroc synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this technology. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: What is the primary advantage of the gamma-azide route over conventional Maraviroc synthesis?
A: The gamma-azide route eliminates the need for complex amine protection and deprotection sequences required in traditional methods starting from amino esters. This results in fewer synthetic steps, milder reaction conditions (0-50°C), and significantly simplified purification processes, leading to higher overall yields and purity.
Q: How does the hydrolysis step utilizing silver nitrate impact product quality?
A: The use of silver nitrate in an acetone/water mixture allows for the selective hydrolysis of the halogenated intermediate to the aldehyde under mild thermal conditions (50-70°C). This specificity minimizes the formation of over-oxidized byproducts or racemization, ensuring the stereochemical integrity of the chiral center is maintained throughout the synthesis.
Q: Is this synthesis method scalable for industrial API production?
A: Yes, the method is highly scalable due to the use of commercially available reagents like lithium aluminum hydride and sodium triacetoxyborohydride, and the avoidance of cryogenic temperatures. The workup procedures involve standard extractions and chromatography, which are easily adaptable from kilogram to multi-ton scales in a GMP environment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Maraviroc Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the gamma-azide synthesis route for producing high-quality Maraviroc intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Maraviroc intermediate delivered meets the highest standards of safety and efficacy required for antiviral drug development.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your budget. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about securing a stable and cost-effective supply of this critical HIV medication intermediate.
