Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles Using Iron Catalysis for Commercial API Production
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles Using Iron Catalysis for Commercial API Production
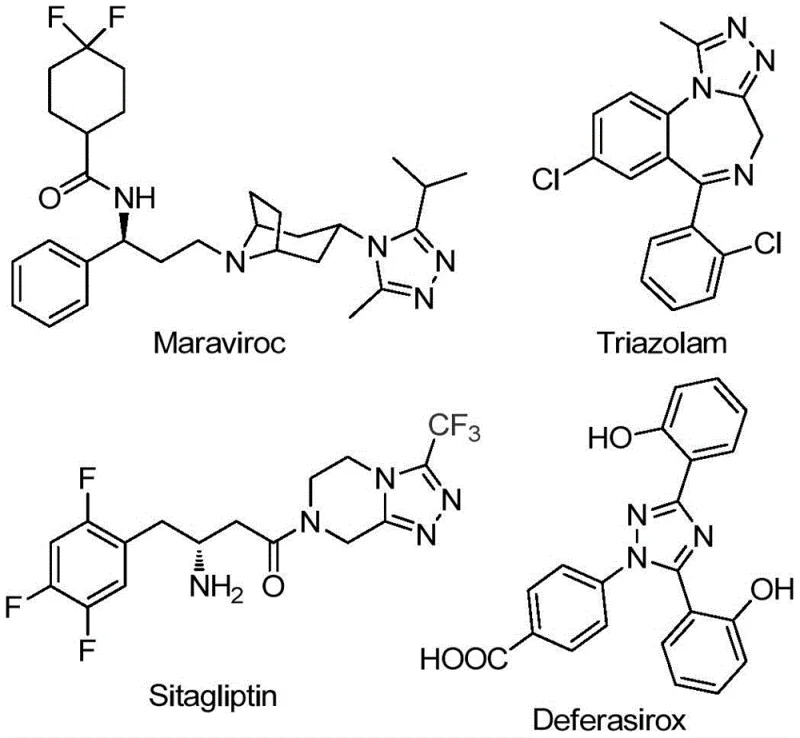
The pharmaceutical and agrochemical industries continuously seek robust methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which enhance metabolic stability and bioavailability. Patent CN111978265B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives, a structural core prevalent in blockbuster drugs such as Maraviroc, Triazolam, and Sitagliptin. This innovation addresses critical bottlenecks in existing synthetic routes by employing a dual-promoter system involving sodium bicarbonate and ferric chloride. For R&D directors and procurement managers, this technology represents a paradigm shift towards more sustainable and cost-effective manufacturing of high-value intermediates. The method eliminates the need for stringent anhydrous or oxygen-free environments, significantly lowering operational complexity while maintaining high yields across a broad spectrum of substrates. By leveraging readily available starting materials like hydrazides and trifluoroethylimidoyl chlorides, this process offers a reliable pathway for producing high-purity pharmaceutical intermediates essential for modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by significant technical hurdles that impede efficient commercial production. Traditional literature methods often rely on the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines, processes that frequently demand harsh reaction conditions and multi-step sequences. These legacy approaches suffer from narrow substrate scopes, particularly struggling with alkyl hydrazones, and often result in disappointing reaction yields that compromise overall process economics. Furthermore, many established protocols require specialized equipment to maintain inert atmospheres or strictly anhydrous conditions, driving up capital expenditure and operational costs for manufacturing facilities. The inability to efficiently synthesize 3-alkyl substituted variants limits the chemical space available to medicinal chemists, forcing reliance on more expensive or convoluted alternative routes. Consequently, there is a pressing industry demand for a streamlined methodology that can overcome these limitations while ensuring consistent quality and supply chain reliability for critical API intermediates.
The Novel Approach
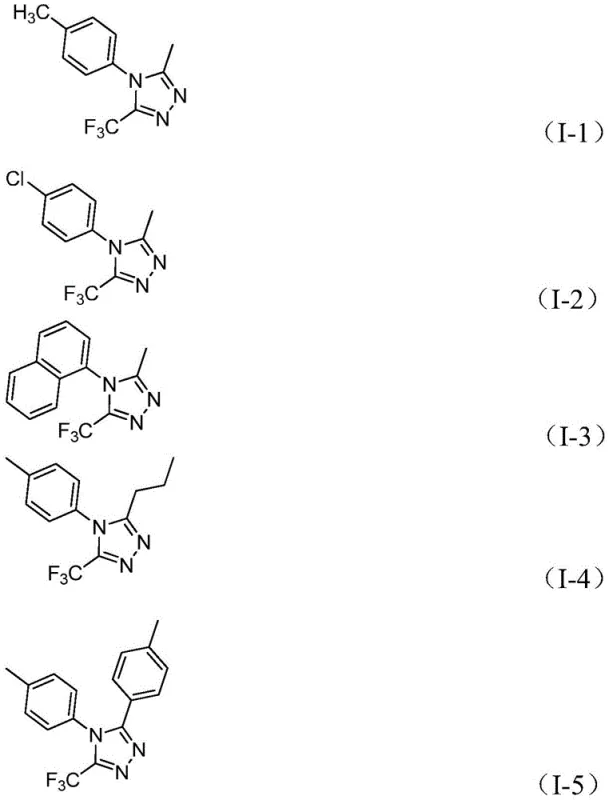
The patented methodology introduces a remarkably simple yet highly effective strategy utilizing iron catalysis to construct the triazole ring under mild conditions. By reacting trifluoroethylimidoyl chlorides with hydrazides in the presence of sodium bicarbonate and ferric chloride, the process achieves efficient cyclization without the need for exotic reagents or extreme parameters. This novel approach allows for the successful incorporation of diverse R2 groups, including alkyl, alkenyl, and various substituted aryl moieties, thereby vastly expanding the accessible chemical library for drug development. The reaction proceeds smoothly in common aprotic solvents like 1,4-dioxane, facilitating easy handling and workup procedures that are compatible with standard industrial infrastructure. Crucially, the tolerance for air and moisture simplifies the operational workflow, reducing the burden on technical teams and minimizing the risk of batch failures due to environmental factors. This technological advancement positions manufacturers as a reliable pharmaceutical intermediate supplier capable of delivering complex heterocyclic scaffolds with superior efficiency and reduced lead times.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between base-promoted nucleophilic attack and Lewis acid-catalyzed dehydration. Initially, sodium bicarbonate facilitates the deprotonation of the hydrazide, enhancing its nucleophilicity for an intermolecular attack on the imidoyl chloride to form a trifluoroacetamidine intermediate. This initial step is critical for establishing the carbon-nitrogen framework required for subsequent ring closure. Following this, the introduction of ferric chloride acts as a potent Lewis acid promoter, coordinating with nitrogen atoms to activate the intermediate for intramolecular cyclization. The metal center likely stabilizes the transition state during the dehydration condensation step, driving the equilibrium towards the formation of the aromatic 1,2,4-triazole ring. This dual-catalyst system ensures high conversion rates even with sterically hindered or electronically diverse substrates, providing a robust solution for synthesizing high-purity OLED material precursors and pharmaceutical building blocks. Understanding this mechanism allows process chemists to fine-tune reaction parameters for optimal impurity profiles and maximum yield recovery.
Impurity control is inherently managed through the specificity of the iron-catalyzed cycle, which minimizes side reactions common in harsher acidic or basic conditions. The mild thermal profile, ranging from 30°C to 90°C across two stages, prevents thermal degradation of sensitive functional groups often present in advanced intermediates. The use of ferric chloride, a cheap and abundant metal salt, avoids the contamination issues associated with precious metal catalysts like palladium or rhodium, simplifying downstream purification and heavy metal removal processes. This aspect is particularly vital for meeting stringent regulatory standards in API manufacturing, where residual metal limits are strictly enforced. The broad functional group tolerance means that substituents such as halogens, methoxy groups, and trifluoromethyl groups on the aromatic rings remain intact, preserving the desired physicochemical properties of the final molecule. Such precision in synthesis ensures that the resulting triazole derivatives possess the necessary purity and structural integrity for downstream biological evaluation and commercial application.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for seamless integration into existing laboratory and pilot plant workflows, emphasizing operational simplicity and safety. The process begins with the precise weighing of stoichiometric amounts of trifluoroethylimidoyl chloride, hydrazide, and sodium bicarbonate, which are then suspended in an appropriate volume of 1,4-dioxane. Detailed standardized operating procedures ensure reproducibility and safety during the handling of reactive chlorides and hydrazides. The reaction mixture is subjected to a controlled heating ramp, first holding at moderate temperatures to allow for the initial coupling, followed by the addition of the iron catalyst and a second heating phase to drive cyclization to completion. Post-reaction processing involves straightforward filtration to remove inorganic salts, followed by silica gel treatment and column chromatography to isolate the pure product. This streamlined protocol minimizes waste generation and solvent consumption, aligning with green chemistry principles while delivering high-quality outputs. For comprehensive step-by-step instructions and specific molar ratios tailored to different substrates, please refer to the detailed guide below.
- Mix sodium bicarbonate, trifluoroethylimidoyl chloride, and hydrazide in an aprotic organic solvent such as 1,4-dioxane.
- Heat the mixture to 30-50°C and stir for 8-16 hours to facilitate initial intermolecular bond formation.
- Add ferric chloride catalyst to the system, raise temperature to 70-90°C, and react for 6-10 hours to complete cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers substantial benefits that directly impact the bottom line and supply chain resilience for chemical manufacturers. The reliance on commodity chemicals such as ferric chloride and sodium bicarbonate drastically reduces raw material costs compared to processes requiring specialized organometallic catalysts or exotic reagents. Furthermore, the elimination of strict inert atmosphere requirements lowers the barrier to entry for contract manufacturing organizations, allowing for broader sourcing options and increased competition among suppliers. This flexibility translates into enhanced supply chain reliability, as production is less susceptible to disruptions caused by equipment failure or gas supply shortages. The ability to scale the reaction to gram levels and beyond without significant re-optimization provides confidence in the continuity of supply for long-term projects. Additionally, the simplified workup procedure reduces the time and resources needed for purification, accelerating the overall production cycle and enabling faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The substitution of expensive precious metal catalysts with inexpensive ferric chloride results in significant direct cost savings on reagent procurement. By avoiding the need for rigorous drying of solvents and glassware, energy consumption and labor costs associated with preparation are substantially lowered. The high atom economy and yield of the reaction minimize waste disposal costs and maximize the output per batch, further improving the overall cost efficiency of the manufacturing process. These cumulative savings allow for more competitive pricing strategies in the global market for pharmaceutical intermediates and fine chemicals.
- Enhanced Supply Chain Reliability: The use of widely available starting materials ensures that production schedules are not held hostage by the lead times of niche reagents. Since the reaction tolerates air and moisture, the risk of batch rejection due to environmental excursions is minimized, leading to more predictable delivery timelines. This robustness is crucial for maintaining just-in-time inventory levels and meeting the demanding schedules of downstream API producers. Manufacturers can confidently commit to larger volumes knowing that the process is stable and reproducible across different batches and scales.
- Scalability and Environmental Compliance: The straightforward nature of the reaction conditions facilitates easy scale-up from laboratory benchtop to industrial reactors without complex engineering modifications. The use of common solvents and the absence of highly toxic reagents simplify waste management and regulatory compliance, reducing the environmental footprint of the manufacturing operation. This alignment with sustainability goals enhances the corporate reputation of suppliers and meets the increasing demand for green chemistry solutions in the pharmaceutical industry. The process is well-suited for continuous flow chemistry adaptations, offering further potential for efficiency gains in large-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and experimental data. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs. The answers reflect the proven capabilities of the method in handling diverse substrates and its compatibility with standard industrial practices. This transparency ensures that partners have a clear understanding of the process capabilities and limitations before initiating collaboration.
Q: What are the primary advantages of this FeCl3-catalyzed method over traditional triazole synthesis?
A: Unlike conventional methods requiring harsh conditions or expensive reagents, this protocol operates under air without strict anhydrous requirements, utilizes cheap ferric chloride, and offers broad substrate tolerance for diverse R2 groups including alkyl and aryl moieties.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly demonstrates scalability to the gram level with simple post-processing involving filtration and column chromatography, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Q: How does the choice of solvent impact the reaction efficiency?
A: While various organic solvents can dissolve the reactants, aprotic solvents like 1,4-dioxane are preferred as they effectively promote the reaction progression and ensure higher conversion rates compared to other solvent systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and securing supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative patents like CN111978265B are translated into tangible commercial success. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-trifluoromethyl-1,2,4-triazole derivatives meets the highest international standards. Our commitment to quality and consistency makes us a trusted partner for multinational pharmaceutical companies seeking reliable sources for complex heterocyclic intermediates. By leveraging our technical expertise and state-of-the-art facilities, we help clients navigate the complexities of chemical manufacturing with confidence and ease.
We invite you to engage with our technical procurement team to discuss how this FeCl3-catalyzed route can optimize your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits of switching to this robust synthesis method. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to drive innovation and efficiency in your supply chain, ensuring timely delivery of high-quality intermediates for your next breakthrough therapy.
