Advanced Continuous Flow Synthesis of Betamipron: Enhancing Purity and Scalability for Global Pharmaceutical Manufacturing
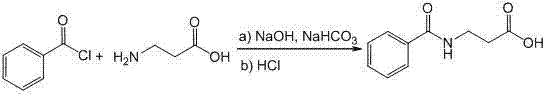
The pharmaceutical industry is constantly seeking robust methodologies to enhance the production efficiency of critical antibiotic adjuvants, and patent CN103588662A presents a groundbreaking approach to synthesizing Betamipron, also known as N-benzoyl-β-alanine. This compound plays a pivotal role in combination therapy with panipenem, effectively reducing nephrotoxicity by competitively inhibiting renal tubular secretion. The disclosed technology leverages continuous-flow microreactor systems to overcome the inherent limitations of traditional batch processing, offering a pathway to higher purity and improved process safety. By utilizing precise microfluidic control, the invention addresses the chronic issue of benzoyl chloride hydrolysis, which has long plagued the quality profile of this essential pharmaceutical intermediate. For R&D directors and procurement specialists alike, this patent represents a significant evolution in cost reduction in API manufacturing, shifting the paradigm from hazardous batch operations to streamlined, continuous processing.
Furthermore, the implementation of this microreactor technology aligns perfectly with the demands of modern regulatory environments that prioritize green chemistry and operator safety. The ability to maintain strict temperature control between -5°C and 5°C within a micro-channel environment ensures that the exothermic nature of the acylation reaction is managed with unprecedented precision. This level of control not only enhances the chemical yield but also drastically simplifies the downstream purification workflow, removing the dependency on toxic solvents like benzene. As a reliable pharmaceutical intermediate supplier, understanding these technological nuances is crucial for securing a supply chain that is both resilient and compliant with international safety standards. The transition to continuous flow is not merely a technical upgrade but a strategic imperative for sustaining the production of high-value carbapenem adjuvants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Betamipron have historically relied on batch reactors where β-alanine reacts with a significant excess of benzoyl chloride, typically ranging from 1.5 to 2 equivalents. This stoichiometric imbalance is necessary to drive the reaction forward but inevitably leads to substantial hydrolysis of the acid chloride, generating benzoic acid as a persistent and difficult-to-remove impurity. To achieve the requisite purity levels for pharmaceutical application, the crude product traditionally requires washing with benzene, a solvent known for its high toxicity and carcinogenic potential, followed by multiple rounds of recrystallization. These extensive purification steps not only degrade the overall process yield but also introduce significant environmental and occupational health hazards that are increasingly unacceptable in modern commercial scale-up of complex pharmaceutical intermediates. The operational complexity of managing large volumes of toxic solvents and the inefficiency of batch-wise heat dissipation create bottlenecks that limit production capacity and increase manufacturing costs.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a continuous-flow microreactor system that fundamentally alters the reaction dynamics through enhanced mass and heat transfer. By injecting an aqueous solution of β-alanine, sodium hydroxide, and sodium bicarbonate alongside benzoyl chloride into a micro-mixer, the reactants are brought into intimate contact within a confined space defined by micro-tubes with diameters ranging from 0.3 to 1.5mm. This configuration allows for rapid mixing and immediate heat removal, effectively suppressing the hydrolysis side reaction that plagues conventional methods. The result is a crude product with significantly lower benzoic acid content, which simplifies the purification process to a straightforward acidification and filtration step without the need for hazardous organic solvent washing. This technological leap facilitates the production of high-purity Betamipron while simultaneously improving operational safety and environmental compliance.
Mechanistic Insights into Microfluidic Acylation Kinetics
The core mechanism driving the success of this synthesis lies in the unique physical phenomena present within the microreactor environment, specifically the dominance of laminar flow and the high surface-area-to-volume ratio. In the micro-channels, diffusion becomes the primary mode of mixing, allowing for extremely precise control over the local concentration of reactants at the interface where the acylation occurs. This precise control prevents the localized accumulation of heat that typically accelerates the hydrolysis of benzoyl chloride in bulk solutions. By maintaining the reaction temperature strictly within the range of -5°C to 5°C, the kinetic energy available for the unwanted hydrolysis pathway is minimized, while the activation energy for the desired amide bond formation is still sufficiently met through efficient molecular collisions. This selective kinetic enhancement ensures that the majority of the benzoyl chloride is consumed in the formation of the betamipron sodium salt rather than being wasted as benzoic acid.
Moreover, the residence time within the micro-tube reactor, determined by the flow rates of 4-48ul/min for the aqueous phase and 0.35-4.14ul/min for the organic phase, is optimized to ensure complete conversion before the mixture exits the reactor. The subsequent acidification step to pH=2 using concentrated hydrochloric acid triggers the precipitation of the free acid form of Betamipron, which can be easily isolated. This mechanistic efficiency translates directly into a cleaner impurity profile, as the absence of benzene washing steps eliminates the risk of solvent residues in the final API intermediate. For quality control teams, this means a more consistent product with a predictable impurity spectrum, reducing the analytical burden and ensuring that every batch meets the stringent specifications required for combination antibiotic therapies.
How to Synthesize Betamipron Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this continuous flow technology in a production setting, emphasizing the critical parameters of flow rate, temperature, and reactor geometry. Operators must ensure that the aqueous feed containing the amino acid and bases is prepared at a concentration of 0.5~1M to maintain optimal solubility and reaction kinetics within the micro-channels. The synchronization of the two feed pumps is vital to maintain the stoichiometric balance and prevent channel clogging or pressure spikes that could disrupt the laminar flow regime. Detailed standardized synthetic steps see the guide below.
- Prepare an aqueous solution containing 0.5~1M β-alanine, sodium hydroxide, and sodium bicarbonate, and inject it into a micro-mixer at a controlled flow rate.
- Simultaneously inject benzoyl chloride through a connecting pipe into the micro-mixer, allowing the mixture to enter a micro-tube reactor maintained at -5°C to 5°C.
- Acidify the resulting betamipron sodium salt solution to pH=2 using concentrated hydrochloric acid to precipitate the solid product, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microreactor technology offers tangible benefits that extend beyond simple yield improvements, addressing critical pain points related to cost, safety, and scalability. The elimination of toxic benzene from the purification process removes the substantial costs associated with solvent procurement, recovery, and hazardous waste disposal, leading to a leaner and more cost-effective manufacturing operation. Furthermore, the continuous nature of the process allows for a smaller physical footprint compared to large batch reactors, enabling production facilities to maximize their existing infrastructure without requiring massive capital expenditure on new containment systems. This efficiency gain is particularly valuable in the context of reducing lead time for high-purity pharmaceutical intermediates, as the streamlined workflow reduces the total cycle time from raw material input to finished product packaging.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily by eliminating the need for expensive and hazardous solvent handling systems associated with benzene washing. By reducing the formation of benzoic acid impurities, the yield loss typically incurred during multiple recrystallization steps is significantly mitigated, meaning more saleable product is generated from the same amount of raw materials. Additionally, the continuous flow system operates with higher energy efficiency due to superior heat exchange, lowering the utility costs per kilogram of product produced. These factors combine to create a more competitive cost structure that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The robustness of the microreactor system ensures consistent product quality batch after batch, reducing the risk of supply disruptions caused by failed quality control tests. Since the process does not rely on complex, multi-step purification involving toxic solvents, the regulatory approval process for manufacturing sites is simplified, ensuring uninterrupted supply to global markets. The ability to scale production by numbering up microreactor units rather than scaling up vessel size provides flexibility to meet sudden increases in demand for panipenem combination therapies without long lead times for new equipment installation.
- Scalability and Environmental Compliance: This technology inherently aligns with green chemistry principles by minimizing waste generation and eliminating the use of carcinogenic solvents, making it easier to comply with increasingly strict environmental regulations worldwide. The modular nature of the microreactor setup allows for seamless capacity expansion, enabling manufacturers to ramp up production from pilot scale to commercial tonnage with minimal re-validation efforts. This scalability ensures that the supply chain remains agile and responsive to the dynamic needs of the pharmaceutical industry while maintaining a strong commitment to sustainability and operator safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous flow synthesis of Betamipron, providing clarity on how this patented method compares to established industry practices. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: How does the microreactor method improve the purity of Betamipron compared to traditional batch synthesis?
A: The microreactor method significantly reduces the hydrolysis of benzoyl chloride, which is the primary source of benzoic acid impurities in conventional methods. By precisely controlling mixing and temperature between -5°C and 5°C, the side reactions are minimized, eliminating the need for toxic benzene washing and multiple recrystallizations.
Q: What are the specific reaction conditions required for optimal yield in this continuous flow process?
A: Optimal results are achieved with a micro-tube diameter of 0.3~1.5mm and a length of 1~3m. The aqueous phase flow rate should be between 4-48ul/min, while benzoyl chloride is injected at 0.35-4.14ul/min, maintaining a water bath temperature strictly between -5°C and 5°C.
Q: Why is this synthesis method considered safer for large-scale pharmaceutical manufacturing?
A: This method eliminates the use of highly toxic benzene solvents required for purification in traditional routes. Additionally, the microreactor's enhanced heat transfer capabilities prevent thermal runaway risks associated with exothermic acylation reactions, ensuring operational safety during commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Betamipron Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of Betamipron in the global antibiotic market and possess the technical expertise to translate advanced patent technologies like CN103588662A into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of microreactor synthesis can be realized at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every shipment of Betamipron meets the highest standards required for pharmaceutical applications, providing our partners with absolute confidence in product quality and consistency.
We invite you to engage with our technical procurement team to discuss how we can tailor this continuous flow technology to your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this greener, more efficient synthesis route. We are ready to provide specific COA data and route feasibility assessments to support your R&D and procurement strategies, ensuring a seamless partnership that drives value and innovation in your antibiotic production lines.
