Advanced Continuous-Flow Synthesis of Betamipron: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
The pharmaceutical industry is constantly seeking robust methodologies to enhance the efficiency and sustainability of active pharmaceutical ingredient (API) intermediate production. Patent CN103588662A, published on February 19, 2014, introduces a groundbreaking approach for the synthesis of Betamipron, also known as N-benzoyl-β-alanine, utilizing continuous-flow microreactor technology. This innovation addresses critical limitations inherent in traditional batch processing, specifically targeting the reduction of hazardous solvent usage and the improvement of reaction selectivity. By leveraging the unique physical properties of microfluidic systems, such as enhanced heat and mass transfer, this method achieves superior control over the acylation reaction between β-alanine and benzoyl chloride. For R&D directors and process chemists, this patent represents a significant leap forward in process intensification, offering a pathway to produce high-purity Betamipron with reduced environmental impact. The transition from batch to continuous flow not only optimizes the reaction kinetics but also fundamentally alters the impurity profile, ensuring a cleaner crude product that requires less aggressive purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Betamipron has relied on conventional batch reactors where β-amino propionic acid reacts with a substantial excess of benzoyl chloride, typically ranging from 1.5 to 2 equivalents. This stoichiometric imbalance is necessary to drive the reaction to completion but inevitably leads to significant side reactions, most notably the hydrolysis of benzoyl chloride into benzoic acid. The presence of benzoic acid as a major by-product complicates the downstream purification process immensely, necessitating the use of highly toxic organic solvents like benzene for washing. Furthermore, achieving the requisite pharmaceutical grade purity often demands multiple recrystallization cycles, which drastically reduces the overall yield and increases operational costs. From a supply chain perspective, the reliance on hazardous solvents introduces severe safety risks and regulatory burdens, while the low purification yield creates inefficiencies in raw material utilization. These factors collectively hinder the scalability and economic viability of the traditional batch process, making it less attractive for modern, large-scale pharmaceutical manufacturing environments that prioritize safety and sustainability.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN103588662A utilizes a continuous-flow microreactor system to orchestrate the synthesis with unprecedented precision. By confining the reaction within micro-channels with diameters ranging from 0.3mm to 1.5mm, the system achieves an exceptionally high surface-area-to-volume ratio, facilitating rapid heat dissipation and uniform mixing. This environment allows the reaction to proceed at mild temperatures between -5°C and 5°C, effectively suppressing the hydrolysis of benzoyl chloride and minimizing the formation of benzoic acid impurities. The continuous nature of the process ensures that reactants are mixed instantaneously and consistently, eliminating the concentration gradients often found in batch reactors that lead to localized hot spots and side reactions. Consequently, the crude product obtained from this microfluidic process exhibits significantly higher purity, simplifying the purification workflow and eliminating the need for toxic benzene washes. This technological shift not only enhances the chemical efficiency of the synthesis but also aligns with global trends towards greener, safer, and more cost-effective pharmaceutical manufacturing practices.
Mechanistic Insights into Microfluidic Acylation Kinetics
The core of this technological advancement lies in the precise manipulation of reaction kinetics within the micro-reactor environment. The synthesis involves a Schotten-Baumann type acylation where the nucleophilic attack of the amino group in β-alanine on the carbonyl carbon of benzoyl chloride is the rate-determining step. In a conventional batch setting, the mixing time is often slower than the reaction time, leading to zones of high local concentration of benzoyl chloride which favor hydrolysis by water. However, in the microreactor, the laminar flow regime and short diffusion distances ensure that mixing occurs on a timescale much faster than the reaction itself. This phenomenon, known as mixing-controlled reaction, ensures that the concentration of benzoyl chloride remains uniformly low throughout the reaction channel, thereby kinetically favoring the desired acylation over the competing hydrolysis pathway. The ability to maintain the reaction temperature strictly within the -5°C to 5°C window further stabilizes the reactive acyl chloride species, preventing thermal degradation and ensuring high selectivity for the amide bond formation.
Furthermore, the impurity control mechanism is intrinsically linked to the residence time distribution within the micro-channels. Unlike batch reactors where a broad distribution of residence times can lead to over-reaction or degradation of sensitive intermediates, the plug-flow characteristics of the micro-tube reactor ensure that every fluid element experiences nearly identical reaction conditions. This uniformity prevents the accumulation of by-products and ensures consistent product quality from the start to the end of the production run. The result is a crude product with a markedly reduced content of benzoic acid, which directly translates to a simplified isolation procedure involving simple acidification to pH=2 followed by filtration. This mechanistic advantage underscores the value of microreactor technology for producing high-purity pharmaceutical intermediates where impurity profiles are critical for downstream drug safety and efficacy.
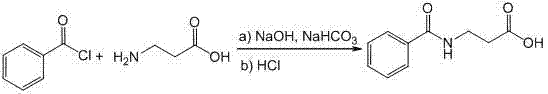
How to Synthesize Betamipron Efficiently
The implementation of this continuous flow synthesis route requires careful attention to the specific operational parameters defined in the patent to ensure reproducibility and optimal yield. The process begins with the preparation of an aqueous phase containing β-alanine, sodium hydroxide, and sodium bicarbonate, which serves as both the substrate source and the acid scavenger. This solution is pumped into the micro-mixer alongside a separate stream of benzoyl chloride, with flow rates precisely calibrated to maintain the desired stoichiometry and residence time. The reaction mixture then traverses a micro-tube reactor submerged in a temperature-controlled water bath, where the acylation takes place under isothermal conditions. Following the reaction, the effluent containing the sodium salt of Betamipron is collected and subjected to acidification to precipitate the final product. For a comprehensive understanding of the standardized operating procedures and specific equipment configurations required for this transformation, please refer to the detailed synthesis guide below.
- Prepare an aqueous solution containing 0.5~1M β-alanine, sodium hydroxide, and sodium bicarbonate, and inject it into a micro-mixer at a controlled flow rate of 4-48ul/min.
- Simultaneously inject benzoyl chloride through a connecting pipe at a flow rate of 0.35-4.14ul/min into the micro-mixer, allowing the mixture to enter a micro-tube reactor maintained at -5°C to 5°C.
- Acidify the resulting betamipron sodium salt solution with concentrated hydrochloric acid to pH=2 to precipitate the solid product, followed by filtration, washing, and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this microreactor-based synthesis offers compelling strategic advantages that extend beyond mere chemical efficiency. The elimination of toxic benzene from the purification process significantly reduces the costs associated with hazardous waste disposal and solvent recovery, leading to substantial operational expenditure savings. Moreover, the improved selectivity and yield mean that less raw material is wasted on by-product formation, optimizing the cost of goods sold (COGS) for this critical antibiotic adjuvant. The continuous nature of the process also enhances supply chain reliability by enabling flexible production scheduling and rapid response to demand fluctuations without the long turnaround times typical of batch cleaning and setup. This agility is crucial for maintaining the continuity of supply for essential medicines like carbapenem antibiotics, where Betamipron plays a vital role in reducing nephrotoxicity.
- Cost Reduction in Manufacturing: The transition to continuous flow technology inherently drives down manufacturing costs through several mechanisms. By minimizing the formation of benzoic acid impurities, the process eliminates the need for expensive and hazardous solvent washing steps, thereby reducing solvent procurement and disposal costs. Additionally, the higher reaction yield ensures that the expensive starting material, benzoyl chloride, is utilized more efficiently, directly lowering the raw material cost per kilogram of the final product. The simplified purification protocol also reduces labor hours and energy consumption associated with multiple recrystallization cycles, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Continuous manufacturing technologies offer superior scalability and consistency compared to traditional batch methods, which is a key factor in ensuring supply chain resilience. The modular nature of microreactor systems allows for capacity expansion through numbering-up rather than scaling-up, reducing the risk associated with large-scale process transfers. This capability ensures a stable and reliable supply of high-purity Betamipron, mitigating the risk of production delays that could impact the availability of life-saving antibiotic combinations. Furthermore, the reduced dependency on hazardous solvents simplifies logistics and storage requirements, enhancing overall supply chain safety and compliance.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process represents a significant advancement in green chemistry. The drastic reduction in hazardous waste generation and the elimination of carcinogenic solvents align perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturers and facilitates smoother audits and inspections. The ability to scale this process commercially while maintaining a small environmental footprint makes it an attractive option for companies aiming to meet sustainability goals and corporate social responsibility targets without compromising on production volume or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous flow synthesis of Betamipron. These insights are derived directly from the technical specifications and beneficial effects outlined in patent CN103588662A, providing clarity on the operational feasibility and advantages of this modern manufacturing approach. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing production frameworks or sourcing strategies.
Q: How does the microreactor method improve the purity of Betamipron compared to traditional batch synthesis?
A: The microreactor method significantly reduces the hydrolysis of benzoyl chloride, which is the primary source of benzoic acid impurities in conventional methods. By precisely controlling mixing and temperature between -5°C and 5°C, the process minimizes side reactions, eliminating the need for toxic benzene washing and multiple recrystallizations.
Q: What are the specific reaction conditions required for optimal yield in this continuous flow process?
A: Optimal results are achieved using a micro-tube reactor with a diameter of 0.3~1.5mm and a length of 1~3m. The aqueous phase flow rate should be maintained between 4-48ul/min, while the benzoyl chloride flow rate is kept between 0.35-4.14ul/min, with the reaction temperature strictly controlled within the range of -5°C to 5°C.
Q: Why is this continuous flow technology considered more environmentally friendly for pharmaceutical manufacturing?
A: This technology eliminates the use of highly toxic benzene solvents traditionally required for purification. Additionally, the enhanced selectivity reduces waste generation by minimizing by-product formation, aligning with green chemistry principles and simplifying downstream waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Betamipron Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthesis technologies like the continuous-flow microreactor method for producing high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that for R&D directors and procurement teams, consistency and reliability are paramount, and our state-of-the-art facilities are designed to deliver exactly that for complex molecules like Betamipron.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable pharmaceutical intermediate supplier dedicated to driving efficiency, safety, and quality in the global pharmaceutical market.
