Advanced Boric Acid Catalysis for Commercial Scale-up of Chiral Diethyl Tartrate
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for producing critical chiral intermediates, a demand directly addressed by the innovative methodology detailed in patent CN101100428A. This patent discloses a robust method for utilizing boric acid to catalyze the synthesis of chiral L- or D-diethyl tartrate, a compound of paramount importance in the realm of asymmetric synthesis and chiral resolution. Unlike traditional methods that rely on harsh mineral acids, this approach leverages the mild Lewis acidity of boric acid to drive the esterification of tartaric acid with ethanol, resulting in a process that is not only economically viable but also environmentally superior. For R&D directors and procurement specialists alike, understanding this technology is crucial, as it represents a significant leap forward in the reliable production of high-purity pharmaceutical intermediates. The ability to achieve high conversion rates while minimizing corrosive waste streams positions this technology as a cornerstone for modern green chemistry initiatives in the manufacturing of specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diethyl tartrate has heavily relied on the use of strong mineral acids such as concentrated sulfuric acid or hydrochloric acid as catalysts for the Fischer esterification reaction. While effective in driving the equilibrium towards the ester product, these conventional methods suffer from severe drawbacks that impact both operational safety and long-term cost efficiency. The highly corrosive nature of these strong acids necessitates the use of expensive glass-lined or Hastelloy reactors, significantly increasing capital expenditure for manufacturing facilities. Furthermore, the post-reaction workup is notoriously difficult, requiring extensive neutralization steps that generate large volumes of saline wastewater, thereby complicating environmental compliance and waste disposal logistics. The presence of residual acid can also lead to side reactions, such as the dehydration of the tartrate backbone, which compromises the optical purity and overall quality of the final chiral intermediate, creating significant challenges for downstream applications in drug synthesis.
The Novel Approach
In stark contrast to these legacy processes, the novel approach outlined in the patent utilizes boric acid as a mild, non-corrosive catalyst, fundamentally altering the economic and environmental profile of the synthesis. By employing boric acid in conjunction with carbon tetrachloride as an azeotropic solvent, the process effectively removes water generated during the reaction, driving the equilibrium towards the desired diethyl tartrate without the need for excessive acid loading. This method eliminates the severe corrosion issues associated with sulfuric acid, allowing for the use of standard stainless steel equipment and drastically reducing maintenance downtime and replacement costs. Moreover, the mild reaction conditions preserve the stereochemical integrity of the chiral center, ensuring that the resulting L- or D-diethyl tartrate meets the stringent purity specifications required for high-value applications in enantioselective catalysis and chiral chromatography, thus offering a superior value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Boric Acid-Catalyzed Esterification
The efficacy of this synthesis lies in the unique mechanistic role played by boric acid, which acts not merely as a proton source but as a Lewis acid that facilitates the formation of reactive intermediate species. In the reaction medium, boric acid interacts with ethanol to form triethyl borate intermediates, which are more electrophilic and capable of activating the carboxylic acid groups of the tartaric acid substrate. This activation lowers the energy barrier for the nucleophilic attack by the alcohol, accelerating the esterification rate significantly compared to uncatalyzed thermal reactions. The presence of carbon tetrachloride is critical in this mechanism, as it forms a low-boiling azeotrope with water, allowing for the continuous removal of the byproduct water via a Dean-Stark trap or similar separation apparatus. This continuous dehydration shifts the chemical equilibrium according to Le Chatelier's principle, ensuring high conversion rates without the need for a vast excess of reagents, thereby optimizing atom economy and reducing raw material consumption.
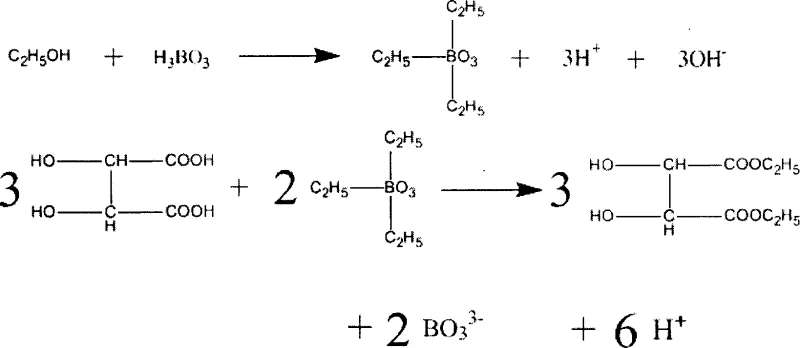
Furthermore, the mechanistic pathway ensures exceptional control over impurity profiles, a key concern for R&D directors overseeing process validation. The mild acidity of the boric acid system prevents the acid-catalyzed degradation or racemization of the chiral tartrate backbone, which is a common pitfall in strong acid catalysis. The reaction proceeds through a coordinated transition state where the boron center stabilizes the developing negative charge on the carbonyl oxygen, facilitating the elimination of water and the formation of the ester bond. This precise control over the reaction trajectory results in a cleaner crude product, simplifying the subsequent purification steps such as washing and vacuum distillation. The ability to recycle the boric acid catalyst from the reaction residue further underscores the efficiency of this mechanism, as the catalyst can be crystallized and returned to the process loop, minimizing waste generation and enhancing the overall sustainability of the manufacturing operation.
How to Synthesize Chiral Diethyl Tartrate Efficiently
The practical implementation of this boric acid-catalyzed route offers a streamlined workflow that is highly amenable to both laboratory-scale optimization and large-scale commercial production. The process begins with the precise mixing of chiral tartaric acid and absolute ethanol, followed by the introduction of the boric acid catalyst and the carbon tetrachloride solvent system. Operators must carefully control the reflux temperature, typically maintaining it around 70°C to 80°C, to ensure efficient azeotropic distillation of water while preventing thermal degradation of the sensitive chiral ester. The detailed standardized synthetic steps, including specific molar ratios, catalyst loading strategies, and purification protocols, are essential for reproducing the high yields reported in the patent literature. For a comprehensive guide on executing this synthesis with maximum efficiency and safety, please refer to the standardized operating procedure outlined below.
- Mix chiral tartaric acid with ethanol in a specific molar ratio (e.g., 1: 5) within a reaction vessel equipped with a reflux condenser and water separator.
- Add carbon tetrachloride as the solvent and introduce boric acid catalyst (approximately 1/15th of the acid mass) to initiate the esterification.
- Heat the mixture to reflux (around 70-80°C) to remove water azeotropically, then purify the crude product via washing, drying, and vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this boric acid-catalyzed technology translates into tangible strategic advantages that extend far beyond simple chemical yield. The shift away from corrosive mineral acids fundamentally changes the cost structure of the manufacturing process by reducing the requirement for specialized corrosion-resistant infrastructure, thereby lowering both capital investment and ongoing maintenance expenditures. Additionally, the capability to recover and reuse both the solvent and the catalyst creates a closed-loop system that significantly mitigates the volatility of raw material pricing and reduces the logistical burden of hazardous waste disposal. This process stability ensures a more predictable supply timeline, as the reduced complexity of the workup and purification stages allows for faster batch turnover and increased throughput capacity, directly addressing the critical need for reducing lead time for high-purity pharmaceutical intermediates in a competitive global market.
- Cost Reduction in Manufacturing: The elimination of strong mineral acids removes the necessity for expensive neutralization agents and the associated waste treatment costs, leading to substantial operational savings. By utilizing a catalyst that can be recovered and recycled directly from the process stream, the consumption of auxiliary chemicals is drastically minimized, contributing to a leaner and more cost-effective production model. The reduced corrosion rate extends the lifespan of reactor vessels and piping, deferring capital replacement costs and ensuring consistent production uptime without unexpected equipment failures. These factors combine to create a significantly lower cost of goods sold (COGS), providing a competitive edge in pricing strategies for bulk chemical procurement.
- Enhanced Supply Chain Reliability: The simplicity of the raw material portfolio, consisting primarily of readily available tartaric acid, ethanol, and boric acid, minimizes the risk of supply chain disruptions caused by scarce or regulated reagents. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, ensuring that production targets can be met consistently even during periods of high market demand. Furthermore, the reduced environmental footprint and zero-discharge potential of the process simplify regulatory compliance, preventing delays associated with environmental permitting and waste disposal audits. This reliability is crucial for maintaining continuous supply lines to downstream pharmaceutical manufacturers who depend on just-in-time delivery of critical chiral building blocks.
- Scalability and Environmental Compliance: The process is designed with inherent scalability, utilizing standard unit operations such as reflux, azeotropic distillation, and vacuum stripping that are easily transferred from pilot plants to multi-ton commercial reactors. The claim of zero discharge and the ability to recycle solvents align perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against tighter emission standards. By avoiding the generation of heavy saline wastewater, the facility reduces its environmental liability and enhances its corporate social responsibility profile. This alignment with green chemistry principles not only satisfies regulatory bodies but also appeals to end-clients who are prioritizing sustainable sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the boric acid-catalyzed synthesis of chiral diethyl tartrate, derived directly from the specific embodiments and claims of the referenced patent data. These insights are intended to clarify the operational parameters and benefits of this technology for potential partners and technical evaluators. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing portfolios or for qualifying new suppliers for critical chiral intermediates.
Q: Why is boric acid preferred over sulfuric acid for this esterification?
A: Boric acid is non-toxic and non-corrosive, significantly reducing equipment maintenance costs and eliminating the need for complex neutralization steps required by strong mineral acids.
Q: Can the solvent and catalyst be recovered in this process?
A: Yes, the patent specifies that both the carbon tetrachloride solvent and the boric acid catalyst can be recycled and reused, contributing to a zero-discharge process.
Q: What yields can be expected from this boric acid catalyzed route?
A: Under optimized conditions described in the patent, the process achieves an esterification rate of 96.68% and an isolated yield of approximately 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diethyl Tartrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation pharmaceuticals and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the boric acid method described in CN101100428A, we can offer our clients a reliable diethyl tartrate supplier partnership that balances technical excellence with commercial viability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for a new program, we are ready to provide the support you need. Contact us today to explore how our expertise in chiral chemistry can accelerate your development timelines and optimize your manufacturing costs.
