Advanced Asymmetric Synthesis of Dapoxetine Intermediates for Commercial Scale-Up
Advanced Asymmetric Synthesis of Dapoxetine Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and cost-efficient pathways for high-volume Active Pharmaceutical Ingredients (APIs), and the synthesis of Dapoxetine represents a prime example of this technological evolution. Patent CN110845369B discloses a groundbreaking methodology for producing Dapoxetine and its critical intermediate, (S)-3-(tert-butoxycarbonyl)amino-3-phenylpropanol, utilizing a novel supramolecular catalytic system. This approach fundamentally shifts the paradigm from wasteful racemic resolution strategies to direct asymmetric synthesis, leveraging inexpensive organocatalysts modified by polymeric structures to achieve superior stereocontrol. For R&D directors and procurement specialists, this technology offers a compelling value proposition by drastically simplifying the supply chain for high-purity pharmaceutical intermediates while ensuring rigorous compliance with environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of Dapoxetine has relied heavily on three primary strategies, each fraught with significant economic and operational inefficiencies that hinder optimal commercial scale-up. The first and most common conventional method involves the synthesis of racemic Dapoxetine followed by chiral resolution, typically using L-tartaric acid; however, this approach inherently wastes at least 50% of the synthesized material as the unwanted enantiomer, necessitating multiple recrystallization steps to achieve acceptable enantiomeric excess (ee) values. The second category relies on chiral pool starting materials, which, while effective, often involve prohibitively expensive raw materials that are difficult to source in bulk quantities, thereby creating bottlenecks in the supply chain and inflating the Cost of Goods Sold (COGS). Furthermore, existing asymmetric catalytic routes frequently employ costly transition metal complexes or sophisticated ligands that require stringent removal processes to meet residual metal specifications, adding complexity to the downstream purification workflow.
The Novel Approach
In stark contrast to these legacy processes, the methodology outlined in patent CN110845369B introduces a highly efficient asymmetric Mannich reaction that constructs the chiral center directly from achiral starting materials using a synergistic catalyst system. This novel route employs L-proline, an abundant and non-toxic amino acid, in conjunction with a polymeric supramolecular catalyst such as PEG1000, to create a chiral environment that directs the stereochemistry of the reaction with high fidelity. By eliminating the need for resolution steps and expensive chiral starting materials, this process significantly enhances atom economy and reduces the overall number of synthetic operations required to reach the target intermediate. The strategic integration of polyethylene glycol (PEG) derivatives not only improves the solubility and stability of the catalytic species but also facilitates easier product isolation, representing a substantial advancement in green chemistry principles for API manufacturing.
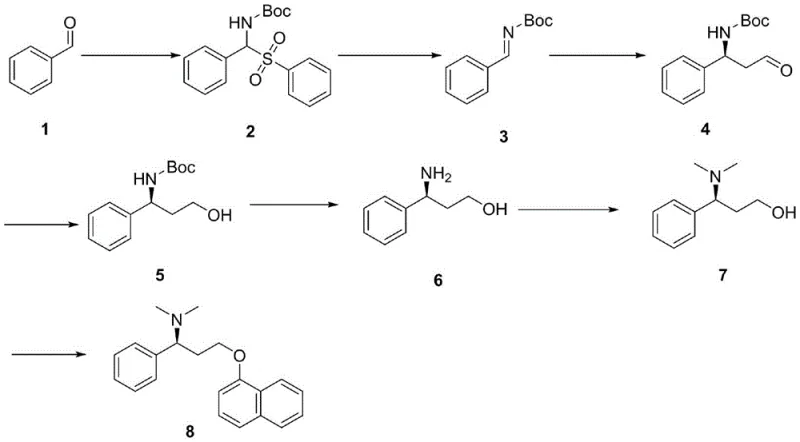
Mechanistic Insights into Polymer-Supported Asymmetric Mannich Reaction
The core innovation of this synthetic pathway lies in the intricate mechanism of the asymmetric Mannich reaction, where the interplay between the chiral organocatalyst and the polymeric additive creates a unique microenvironment for stereoselective bond formation. In this system, L-proline acts as the primary chiral inducer, forming an enamine intermediate with the aldehyde component, while the PEG polymer functions as a supramolecular scaffold that likely stabilizes the transition state through hydrogen bonding or phase-transfer effects. This dual-catalyst strategy ensures that the nucleophilic attack on the imine electrophile occurs with high facial selectivity, yielding the desired (S)-configured product with excellent enantiomeric purity without the need for precious metal catalysts. The use of acetaldehyde as the alkylating agent is particularly advantageous due to its low cost and high reactivity, although controlling its oligomerization requires precise temperature management and addition protocols as detailed in the experimental examples.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of diastereomeric byproducts that are notoriously difficult to separate in later stages of synthesis. The specific choice of solvent, such as acetonitrile, combined with the optimized molar ratios of catalyst to substrate, ensures that the reaction kinetics favor the formation of the target Mannich base over competing aldol condensation side reactions. Furthermore, the subsequent reduction step using sodium borohydride proceeds cleanly on the crude Mannich product, demonstrating the robustness of the intermediate against harsh reducing conditions. This tolerance allows for a streamlined workup procedure where simple extraction and crystallization techniques can achieve the necessary purity levels, thereby reducing the reliance on resource-intensive chromatographic separations that often plague complex pharmaceutical syntheses.
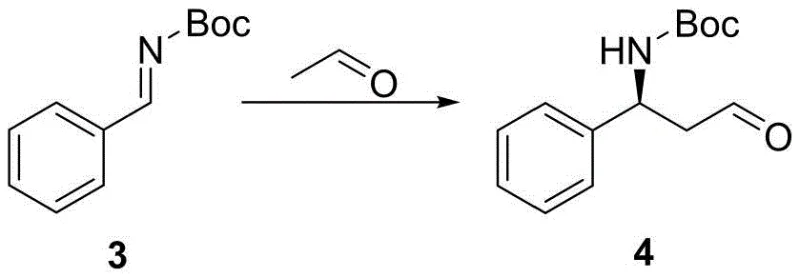
How to Synthesize (S)-3-(tert-butoxycarbonyl)amino-3-phenylpropanol Efficiently
The synthesis of this critical chiral intermediate serves as the foundation for the entire Dapoxetine production line, requiring precise adherence to the optimized reaction parameters to ensure consistent quality and yield. The process begins with the condensation of benzaldehyde to form a sulfone intermediate, followed by elimination to generate the reactive imine species which then undergoes the pivotal asymmetric transformation. Operators must pay close attention to the preparation of fresh acetaldehyde and the maintenance of low temperatures during the catalyst addition to prevent exothermic runaway and ensure maximum stereocontrol. For a comprehensive breakdown of the specific reagent quantities, reaction times, and purification protocols required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Perform condensation of benzaldehyde with sodium benzenesulfinate and tert-butyl carbamate to form the sulfone intermediate.
- Execute an elimination reaction under alkaline conditions to generate the chiral imine precursor.
- Conduct the key asymmetric Mannich reaction using acetaldehyde, L-proline, and a PEG-based supramolecular catalyst to establish chirality.
- Reduce the resulting aldehyde to the corresponding alcohol, followed by Boc deprotection and Eschweiler-Clark methylation.
- Finalize the synthesis via Mitsunobu reaction with 1-naphthol to yield the final Dapoxetine active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthetic route translates into tangible strategic benefits that extend far beyond simple laboratory yield improvements. By shifting away from resolution-based methods, manufacturers can effectively double their theoretical output from the same amount of starting raw materials, as no portion of the batch is discarded as the wrong enantiomer. This dramatic improvement in material efficiency directly correlates to a significant reduction in raw material procurement costs and waste disposal expenses, providing a competitive edge in pricing negotiations for the final API. Additionally, the reliance on commodity chemicals like benzaldehyde, acetaldehyde, and PEG ensures a stable and resilient supply chain that is less susceptible to the volatility often associated with specialized chiral reagents or imported catalysts.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps removes the inherent 50% yield loss associated with separating racemates, effectively doubling the material throughput without increasing reactor volume or processing time. Furthermore, the substitution of expensive transition metal catalysts with inexpensive, food-grade L-proline and commercially available polymers drastically lowers the catalyst cost per kilogram of product, while simultaneously removing the need for costly heavy metal scavenging and testing procedures required for regulatory compliance.
- Enhanced Supply Chain Reliability: The synthetic route relies exclusively on bulk commodity chemicals that are produced globally in massive quantities, ensuring that production schedules are not disrupted by the scarcity of niche chiral starting materials. The robustness of the supramolecular catalyst system also allows for more flexible sourcing of reagents, as the process tolerances are wider than those of sensitive metal-catalyzed reactions, thereby reducing the risk of batch failures due to minor variations in raw material quality.
- Scalability and Environmental Compliance: This organocatalytic approach aligns perfectly with modern green chemistry initiatives by avoiding toxic heavy metals and reducing the generation of hazardous waste streams associated with resolution agents. The simplified downstream processing, which favors crystallization and extraction over complex chromatography, facilitates easier scale-up from pilot plant to multi-ton commercial production, ensuring that environmental permits are easier to obtain and maintain while minimizing the facility's overall carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology, derived directly from the comparative data and beneficial effects reported in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source. These insights clarify why this specific catalytic system outperforms traditional methods in both economic and operational metrics.
Q: What is the primary advantage of the supramolecular catalyst system in this Dapoxetine synthesis?
A: The use of a polymer-supported supramolecular catalyst, specifically combining L-proline with PEG derivatives, significantly enhances enantioselectivity and yield compared to traditional small-molecule organocatalysts, while remaining cost-effective and easy to handle.
Q: How does this method improve upon traditional racemic resolution processes?
A: Unlike traditional methods that synthesize racemic mixtures and require wasteful resolution steps (losing up to 50% of material), this asymmetric synthesis builds chirality directly during the Mannich reaction, maximizing atom economy and reducing raw material costs.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process utilizes cheap, commercially available starting materials like benzaldehyde and acetaldehyde, avoids expensive transition metal catalysts, and operates under mild conditions, making it highly scalable and environmentally compliant for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dapoxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Dapoxetine depends on a partner who can seamlessly translate innovative patent chemistry into reliable, large-scale manufacturing reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is managed with precision and safety. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of (S)-3-(tert-butoxycarbonyl)amino-3-phenylpropanol meets the exacting standards required for global regulatory submissions and patient safety.
We invite you to engage with our technical procurement team to discuss how this advanced asymmetric synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your market competitiveness.
