Advanced Asymmetric Synthesis of Optically Pure 1,3-Alkylamine Intermediates for Commercial Dapoxetine Production
The pharmaceutical industry's relentless pursuit of efficient, scalable, and environmentally sustainable synthetic routes has brought significant attention to the production of optically pure amines, particularly those serving as key intermediates for central nervous system therapeutics. Patent CN101875666A introduces a groundbreaking methodology for the preparation of optically pure 1,3-alkylamine compounds, specifically targeting the synthesis of Dapoxetine and its analogues. This technology represents a paradigm shift from traditional resolution-based methods to direct asymmetric synthesis, leveraging the powerful stereocontrolling capabilities of chiral sulfinamide auxiliaries. By establishing a robust pathway to generate chiral centers with exceptional fidelity, this innovation addresses critical bottlenecks in the supply chain for selective serotonin reuptake inhibitors (SSRIs). 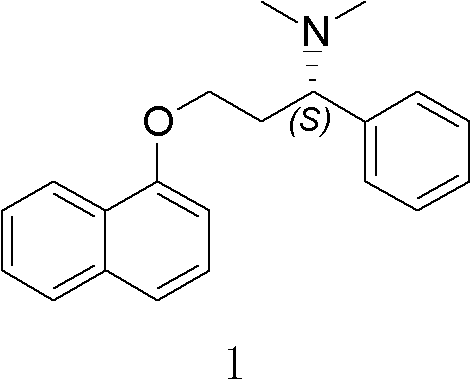
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure Dapoxetine has relied heavily on chiral resolution techniques, which are inherently inefficient and economically burdensome for large-scale manufacturing. Traditional approaches often involve the synthesis of racemic mixtures followed by separation using chiral selectors such as L-(+)-tartrate or D-(+)-di-p-toluoyl tartrate. These methods typically require multiple recrystallization steps to achieve acceptable enantiomeric excess (ee) values, resulting in a maximum theoretical yield of only 50% for the desired enantiomer while the other half is discarded as waste. Furthermore, alternative synthetic routes utilizing toxic reagents like sodium cyanide for chain elongation pose severe safety hazards and environmental compliance challenges, complicating the regulatory approval process for commercial facilities. The reliance on expensive, optically active starting materials that are difficult to source further exacerbates the cost structure, making these conventional pathways unsustainable for meeting the growing global demand for premature ejaculation treatments.
The Novel Approach
In stark contrast, the methodology disclosed in CN101875666A employs a highly efficient asymmetric addition strategy that constructs the chiral center directly with superior stereocontrol. By utilizing chiral N-tert-butylsulfinamides as auxiliaries, the process facilitates the nucleophilic addition of organometallic reagents to protected hydroxy-aldehyde imines with remarkable precision. This approach eliminates the inherent 50% yield loss associated with resolution, effectively doubling the material throughput from the same amount of starting inputs. The use of tert-butyldimethylsilyl (TBS) protection ensures compatibility with the reaction conditions while allowing for mild deprotection later in the sequence. This novel route not only streamlines the synthetic steps but also significantly reduces the generation of hazardous waste, aligning perfectly with modern green chemistry principles and offering a distinct competitive advantage in terms of both cost and environmental impact.
Mechanistic Insights into Chiral Sulfinamide-Mediated Asymmetric Addition
The core of this technological breakthrough lies in the unique ability of the chiral sulfinamide group to direct the stereochemical outcome of the nucleophilic attack. The process begins with the condensation of a protected 3-hydroxy aldehyde, such as 3-(tert-butyldimethylsilyloxy)propanal, with an enantiomerically pure sulfinamide to form a chiral N-sulfinyl imine. This intermediate adopts a specific conformation where the bulky tert-butyl group on the sulfur atom creates a steric shield, blocking one face of the imine double bond. 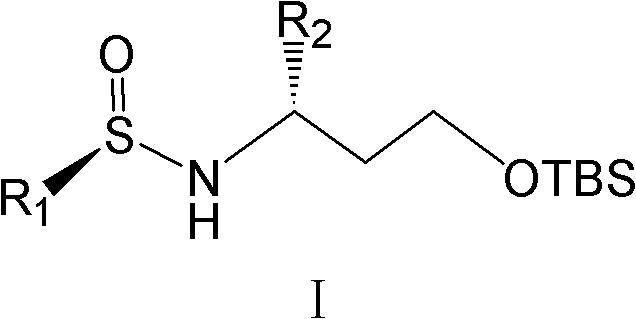 When an organometallic reagent, such as a Grignard reagent (e.g., phenylmagnesium bromide), approaches the imine, it is forced to attack from the less hindered face, dictated by the chiral environment of the sulfinyl group. This highly organized transition state ensures that the new carbon-carbon bond is formed with a specific spatial arrangement, resulting in the formation of a single diastereomer with high selectivity. The subsequent removal of the sulfinyl group under acidic conditions proceeds with retention of configuration, delivering the free amine with optical purity often exceeding 99% ee, thereby virtually eliminating the need for further chiral purification steps.
When an organometallic reagent, such as a Grignard reagent (e.g., phenylmagnesium bromide), approaches the imine, it is forced to attack from the less hindered face, dictated by the chiral environment of the sulfinyl group. This highly organized transition state ensures that the new carbon-carbon bond is formed with a specific spatial arrangement, resulting in the formation of a single diastereomer with high selectivity. The subsequent removal of the sulfinyl group under acidic conditions proceeds with retention of configuration, delivering the free amine with optical purity often exceeding 99% ee, thereby virtually eliminating the need for further chiral purification steps.
Impurity control in this system is intrinsically linked to the stability of the chiral auxiliary and the rigor of the reaction conditions. The patent specifies reaction temperatures ranging from -78°C to -25°C, which are critical for suppressing non-selective background reactions that could lead to racemization. At higher temperatures, the energy barrier for the non-preferred attack trajectory might be overcome, leading to the formation of the undesired diastereomer. Furthermore, the choice of solvent, such as tetrahydrofuran (THF) or toluene, plays a vital role in coordinating the metal cation of the Grignard reagent, further tightening the transition state geometry. By strictly adhering to these parameters, manufacturers can ensure that side products are minimized, resulting in a crude product profile that is exceptionally clean and easier to purify, thus reducing the load on downstream chromatography or crystallization units.
How to Synthesize Optically Pure 1,3-Alkylamine Efficiently
The synthesis of these high-value intermediates requires precise execution of the condensation and addition steps to maximize yield and optical purity. The process initiates with the preparation of the aldehyde precursor, followed by imine formation and the crucial stereoselective addition. Operators must ensure that all reagents are anhydrous and that the inert atmosphere is maintained throughout to prevent hydrolysis of the sensitive imine intermediate. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale implementation. For detailed standard operating procedures and specific stoichiometric ratios, please refer to the technical guide below.
- Condense 3-(tert-butyldimethylsilyloxy)propanal with (R)-tert-butylsulfinamide using a dehydrating agent like anhydrous copper sulfate to form the chiral sulfinimine intermediate.
- React the chiral sulfinimine with an organometallic reagent (such as phenylmagnesium bromide) at low temperatures (-78°C to -25°C) under inert atmosphere to achieve stereoselective addition.
- Remove the protecting groups (sulfinyl and silyl) under acidic conditions to yield the high-purity optically active amino alcohol, which serves as the key precursor for Dapoxetine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric synthesis route offers transformative benefits that extend far beyond simple chemical efficiency. By shifting away from resolution-based methods, companies can drastically reduce their raw material consumption, as the process does not discard half of the synthesized material. This fundamental improvement in atom economy translates directly into lower cost of goods sold (COGS) and a more predictable supply baseline, insulating the business from volatility in raw material pricing. Moreover, the elimination of toxic cyanide reagents simplifies waste disposal protocols and reduces the regulatory burden associated with handling hazardous substances, thereby accelerating the timeline for facility audits and production approvals.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substantial increase in overall yield. Unlike resolution methods that cap yield at 50%, this asymmetric route consistently delivers yields approaching 90% for the key intermediate, as demonstrated in the patent examples. This near-doubling of output per batch means that fixed costs such as labor, energy, and equipment depreciation are spread over a significantly larger volume of product. Additionally, the high optical purity achieved directly from the reaction minimizes the need for expensive chiral chromatography or multiple recrystallizations, further stripping away processing costs and solvent usage.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as simple aldehydes and commercially sourced sulfinamides, mitigates the risk of supply disruptions often associated with specialized chiral pool reagents. The robustness of the reaction conditions allows for flexible scheduling and scale-up without the fear of catastrophic yield drops due to minor parameter fluctuations. This stability ensures a continuous flow of intermediates to downstream API manufacturing sites, preventing bottlenecks that could delay final drug product launches or market replenishment.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations like low-temperature addition and aqueous workups that are easily replicated in multi-ton reactors. From an environmental perspective, the reduction in waste generation and the avoidance of heavy metals or cyanides align with stringent global environmental regulations. This compliance not only avoids potential fines but also enhances the corporate sustainability profile, a factor increasingly weighted by investors and partners in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these nuances is essential for technical teams evaluating the integration of this route into existing production portfolios.
Q: Why is asymmetric synthesis preferred over chiral resolution for Dapoxetine intermediates?
A: Traditional chiral resolution methods, such as those using tartrate salts, often suffer from a maximum theoretical yield of 50% for the desired enantiomer, leading to significant raw material waste and higher costs. The asymmetric synthesis method described in CN101875666A utilizes chiral sulfinamide auxiliaries to directly construct the chiral center with high stereoselectivity (ee > 99%), effectively doubling the potential yield compared to resolution techniques and eliminating the environmental burden of discarding the unwanted enantiomer.
Q: What are the critical reaction conditions for maintaining high optical purity?
A: Maintaining strict temperature control between -78°C and -25°C during the nucleophilic addition step is crucial for ensuring high diastereoselectivity. Additionally, the reaction must be conducted under an inert gas blanket to prevent moisture or oxygen from degrading the sensitive organometallic reagents or the chiral imine intermediate, which could otherwise lead to racemization and reduced ee values.
Q: Can this synthetic route be scaled for industrial production?
A: Yes, the route is designed for scalability. It avoids the use of highly toxic cyanide reagents often found in older synthetic pathways and utilizes robust protecting groups like TBS and tert-butyl sulfinyl that can be managed effectively in large-scale reactors. The high yields reported in the examples (up to 90% for the key intermediate) suggest a commercially viable process with simplified downstream purification requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dapoxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced asymmetric synthesis requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory patents are successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify optical purity and impurity profiles, guaranteeing that every batch of optically pure 1,3-alkylamine meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to leverage this cutting-edge technology for your Dapoxetine supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive down your manufacturing costs while securing a reliable, high-quality supply of critical pharmaceutical intermediates.
