Revolutionizing Azoxystrobin Production: A Deep Dive into Methyl-Bridge Catalyst Technology
The global demand for high-efficiency, broad-spectrum fungicides continues to drive innovation in agrochemical manufacturing, with azoxystrobin standing out as a critical molecule in crop protection. A significant technological breakthrough in this domain is documented in patent CN109529928B, which introduces a novel catalyst system designed to overcome the longstanding inefficiencies associated with traditional synthesis routes. This patent discloses a specialized catalytic system comprising azabicyclo compounds containing a methyl bridge or aza cage compounds, offering a transformative approach to producing azoxystrobin and its key intermediates. Unlike conventional methods that struggle with high energy consumption and difficult process control, this new system leverages unique steric and electronic properties of methyl-bridged nitrogen heterocycles to achieve exceptional conversion rates. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move towards more sustainable and cost-effective agrochemical intermediate manufacturing. The technology promises not only to streamline the synthetic pathway but also to enhance the purity profile of the final active ingredient, addressing critical quality parameters required by regulatory bodies worldwide.
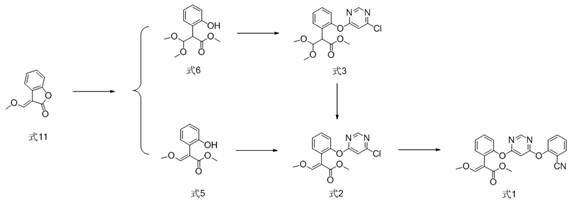
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of azoxystrobin has relied heavily on binary bridged ring compounds containing an ethylene bridge bond, most notably 1,4-diazabicyclo[2.2.2]octane (DABCO). As illustrated in the structural comparisons found in prior art, these ethylene-bridged catalysts often present significant operational challenges during scale-up. The primary limitation lies in the difficulty of controlling reaction conditions; traditional processes frequently require high catalyst loadings, sometimes ranging from 2mol% to 40mol%, to achieve acceptable yields. This excessive usage not only inflates raw material costs but also complicates downstream purification, as removing residual catalyst and associated byproducts becomes increasingly difficult. Furthermore, the reaction kinetics with these older catalysts can be sluggish, leading to prolonged reaction times that reduce overall plant throughput. The reliance on such catalysts often results in inconsistent batch-to-batch quality, with impurity profiles that may fail to meet the stringent specifications demanded by modern high-purity agrochemical standards. Consequently, manufacturers face a constant struggle to balance yield optimization with cost containment and environmental compliance.
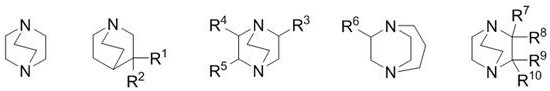
The Novel Approach
In stark contrast to the limitations of ethylene-bridged systems, the novel approach detailed in CN109529928B utilizes azabicyclo compounds featuring a methyl bridge or complex aza cage structures. This structural modification fundamentally alters the catalytic landscape, providing a more robust and efficient pathway for coupling reactions essential to azoxystrobin synthesis. The new catalysts, represented by general formulas 10A through 10E, exhibit superior nucleophilicity and steric accessibility, allowing them to facilitate the coupling of salicylonitrile with chloropyrimidine intermediates with remarkable efficiency. One of the most compelling advantages of this novel approach is the drastic reduction in catalyst loading; the patent demonstrates that effective catalysis can be achieved with as little as 0.001 mol of catalyst per mole of substrate, a fraction of what is required by traditional methods. This efficiency translates directly into simplified workup procedures and reduced waste generation. Moreover, the process operates effectively across a broad temperature range, from mild conditions for intermediate formation to elevated temperatures for final coupling, offering flexibility that is crucial for commercial scale-up of complex agrochemical intermediates. The result is a synthesis route that is not only faster but also inherently safer and more environmentally benign.
Mechanistic Insights into Methyl-Bridge Catalyzed Coupling
The efficacy of the methyl-bridge catalyst system stems from its unique ability to activate electrophilic centers without inducing unwanted side reactions. Mechanistically, the tertiary amine nitrogen atoms within the rigid bicyclic or caged framework act as potent nucleophiles, attacking the electron-deficient carbon atoms on the pyrimidine ring. This initial attack forms a reactive intermediate that facilitates the subsequent displacement of the chloride leaving group by the phenolic oxygen of the salicylonitrile derivative. The rigidity of the methyl-bridge structure prevents the catalyst from adopting conformations that might lead to non-productive binding or decomposition, thereby ensuring a high turnover number. This structural integrity is particularly important when dealing with sensitive functional groups present in the azoxystrobin backbone, such as the methoxy acrylate moiety, which is prone to isomerization or degradation under harsh conditions. By maintaining a controlled reaction environment, the catalyst minimizes the formation of geometric isomers and other structural impurities that often plague conventional syntheses. For R&D teams, this means a cleaner reaction profile that requires less intensive chromatographic purification, directly impacting the cost of goods sold.
Furthermore, the impurity control mechanism is enhanced by the specific solvation properties of the catalyst system. The patent highlights the use of diverse solvent systems, including toluene, DMF, and mixed solvents, which interact favorably with the methyl-bridge catalyst to stabilize the transition state. This stabilization lowers the activation energy required for the coupling reaction, allowing it to proceed rapidly even at lower temperatures where thermal degradation of reactants is minimized. The presence of the methyl bridge also influences the basicity of the nitrogen centers, optimizing the balance between nucleophilic attack and proton abstraction. This fine-tuned basicity prevents the hydrolysis of the ester groups in the acrylate side chain, a common side reaction in less optimized systems. Consequently, the final product exhibits a superior purity profile, with HPLC analysis in the patent examples showing content levels exceeding 98%. Such high purity is essential for meeting the rigorous quality standards of global agrochemical markets and ensures consistent biological performance in the field.
How to Synthesize Azoxystrobin Efficiently
The synthesis of azoxystrobin using this advanced catalyst system involves a series of well-defined steps that prioritize safety and efficiency. The process begins with the preparation of the key intermediates, where the methyl-bridge catalyst facilitates the coupling of 4,6-dichloropyrimidine with salicylonitrile or its salts. This step is critical as it establishes the core pyrimidine-ether linkage characteristic of the molecule. Following the formation of the intermediate, the final coupling with the methoxy acrylate moiety is performed under carefully controlled thermal conditions to preserve the stereochemistry of the double bond. The patent provides extensive experimental data demonstrating that this route is robust across various scales, making it an ideal candidate for technology transfer. Detailed standard operating procedures regarding reagent addition rates, temperature ramping, and quenching protocols are essential to replicate the high yields reported in the literature. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized protocol below.
- Prepare the reaction mixture by combining salicylonitrile or its salt with the chloropyrimidine intermediate in a suitable solvent such as toluene or DMF.
- Introduce the novel methyl-bridge azabicyclo catalyst (0.001 to 0.1 mol per mol of substrate) along with an acid-binding agent like potassium carbonate.
- Heat the mixture to 40-150°C depending on the specific intermediate stage, maintain until conversion is complete, then isolate the product via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this methyl-bridge catalyst technology offers tangible strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the significant reduction of raw material costs associated with the catalyst itself. By lowering the required catalyst loading from molar percentages typical of DABCO systems to trace amounts (0.001 mol), manufacturers can achieve substantial cost savings on a per-kilogram basis. This reduction is compounded by the elimination of expensive purification steps often needed to remove residual catalyst metals or organic bases, streamlining the entire production workflow. Additionally, the use of common, commercially available solvents like toluene and ethanol ensures that supply chain risks related to specialized reagents are minimized. The robustness of the reaction conditions also implies a lower risk of batch failures, enhancing supply continuity and reliability for downstream customers. These factors collectively contribute to a more resilient and cost-competitive supply chain for agrochemical intermediates.
- Cost Reduction in Manufacturing: The implementation of this catalyst system drives down manufacturing costs through multiple mechanisms, primarily by drastically reducing the quantity of catalyst required per batch. Since the catalyst is effective at concentrations as low as 0.001 mol, the direct material cost for this component becomes negligible compared to traditional methods. Furthermore, the high selectivity of the reaction reduces the formation of byproducts, which in turn lowers the cost of waste disposal and solvent recovery. The energy consumption is also optimized due to shorter reaction times and the ability to run reactions at moderate temperatures, leading to lower utility costs. These cumulative efficiencies result in a leaner manufacturing process that improves overall margin potential without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the simplicity and robustness of the new synthetic route. The catalysts described are stable organic compounds that do not require special storage conditions or inert atmospheres, simplifying logistics and inventory management. The tolerance of the reaction to a wide range of solvents allows procurement teams to source materials from multiple suppliers, mitigating the risk of shortages. Moreover, the high conversion rates ensure that production schedules are met consistently, reducing the likelihood of delays in fulfilling customer orders. This reliability is crucial for maintaining long-term contracts with major agrochemical companies that demand just-in-time delivery of high-quality intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is facilitated by the inherent safety and controllability of the reaction. The exothermic nature of the coupling is manageable within standard reactor configurations, reducing the need for specialized cooling equipment. From an environmental perspective, the reduction in catalyst waste and solvent usage aligns with green chemistry principles, helping manufacturers meet increasingly strict environmental regulations. The absence of heavy metals in the catalyst system eliminates the need for complex metal scavenging steps, further simplifying the effluent treatment process. This alignment with sustainability goals enhances the corporate image and ensures long-term operational viability in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. The focus is on practical aspects such as catalyst availability, reaction scope, and compatibility with current infrastructure.
Q: What distinguishes the methyl-bridge catalyst from traditional DABCO catalysts?
A: Unlike traditional ethylene-bridged catalysts like DABCO which often require high loadings and harsh conditions, the methyl-bridge azabicyclo compounds disclosed in CN109529928B offer superior steric properties that enhance nucleophilic activity, allowing for significantly lower catalyst dosages (down to 0.001 mol) while maintaining high conversion rates.
Q: Can this catalyst system be used for large-scale industrial production?
A: Yes, the patent explicitly states the system is suitable for large-scale industrial production due to its characteristics of easy process control, short reaction times, and low energy consumption, making it ideal for commercial scale-up of complex agrochemical intermediates.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction typically proceeds in solvents like toluene or mixed solvent systems at temperatures ranging from 40°C to 150°C. For intermediate preparation, milder temperatures between -20°C and 40°C are preferred, ensuring high selectivity and minimizing side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azoxystrobin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst technology described in CN109529928B and are fully equipped to leverage it for our clients' benefit. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this methyl-bridge catalyst system are realized in actual manufacturing output. Our facilities are designed to handle complex organic syntheses with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the highest international standards. We understand that the transition to a new catalytic system requires careful validation, and our technical team is dedicated to supporting this process through comprehensive route scouting and optimization services. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technologically advanced and compliant with global regulatory requirements.
We invite you to engage with our technical procurement team to discuss how this innovative catalyst system can be tailored to your specific production needs. Whether you require a Customized Cost-Saving Analysis to quantify the economic benefits or need specific COA data to validate the purity profile, we are ready to provide the necessary support. Our commitment to transparency and technical excellence ensures that you receive accurate route feasibility assessments that align with your strategic goals. Contact us today to explore how we can collaborate to bring high-quality, cost-efficient azoxystrobin intermediates to the global market, securing your position as a leader in the agrochemical industry.
