Advanced Rivaroxaban Intermediate Synthesis for Scalable Pharmaceutical Production
Introduction to Next-Generation Rivaroxaban Synthesis
The global demand for effective anticoagulants has placed rivaroxaban at the forefront of cardiovascular therapeutics, necessitating robust and economically viable manufacturing processes for its key intermediates. Patent CN102250077B introduces a groundbreaking synthetic methodology that addresses the critical bottlenecks of traditional production, specifically focusing on the construction of the chiral oxazolidinone core. This innovation eliminates the reliance on extreme reaction conditions such as cryogenic temperatures or strictly anhydrous environments, which have historically plagued the scalability of this high-value pharmaceutical intermediate. By streamlining the synthetic pathway into fewer, more efficient steps, this technology offers a compelling solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their production capabilities. The strategic implementation of this route not only enhances atom economy but also aligns with modern green chemistry principles, making it an ideal candidate for cost reduction in API manufacturing initiatives across the global supply chain.
Furthermore, the versatility of this synthetic approach allows for seamless integration into existing facility infrastructures without requiring specialized high-pressure or low-temperature reactors. The patent details a comprehensive protocol that ensures high stereoselectivity, a paramount factor in the efficacy and safety profile of the final anticoagulant drug. For procurement and technical teams evaluating long-term sourcing strategies, understanding the mechanistic advantages of this route is essential for securing a stable supply of high-purity rivaroxaban intermediate. The following analysis dissects the technical nuances of this process, highlighting how it overcomes the limitations of prior art to deliver a commercially superior manufacturing solution.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of rivaroxaban and its precursors has been hindered by the use of highly hazardous and operationally complex reagents. Traditional routes, such as those disclosed in earlier patents like WO2005/068456, often necessitate the use of phosgene, a toxic gas that poses severe safety risks and requires specialized containment infrastructure. Additionally, other methods rely on organolithium reagents like butyllithium, which demand stringent moisture-free and oxygen-free conditions, typically at cryogenic temperatures below -70°C. These苛刻 conditions not only escalate energy consumption and capital expenditure but also introduce significant variability in batch-to-batch consistency. The multi-step nature of these legacy processes, often involving protection and de-protection sequences, leads to cumulative yield losses and generates substantial chemical waste, thereby complicating the commercial scale-up of complex pharmaceutical intermediates. Such inefficiencies create vulnerabilities in the supply chain, leading to potential delays and increased costs for downstream API manufacturers.
The Novel Approach
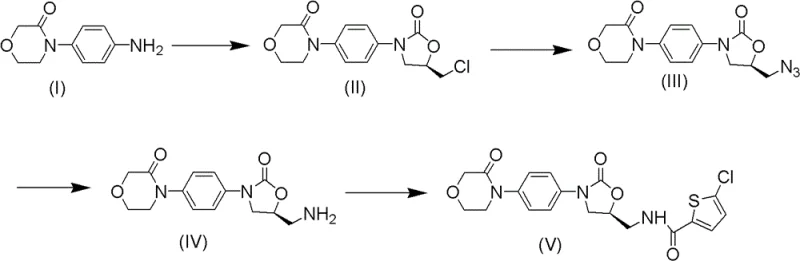
In stark contrast, the methodology described in CN102250077B presents a streamlined, two-step strategy to construct the rivaroxaban nuclear structure with exceptional efficiency. This novel approach utilizes 4-morpholinone aniline and (R)-epichlorohydrin as starting materials, reacting them under reflux conditions to form the chiral backbone without the need for cryogenic cooling. The subsequent cyclization employs carbonyldiimidazole (CDI), a safe and effective reagent that replaces toxic phosgene derivatives, facilitating the formation of the oxazolidinone ring under mild thermal conditions. This shift in chemistry drastically simplifies the operational workflow, allowing for reactions to proceed in common solvents like isopropanol or dichloromethane at atmospheric pressure. By removing the dependency on dangerous reagents and extreme physical parameters, this route significantly lowers the barrier to entry for reducing lead time for high-purity anticoagulants production. The result is a robust process that maintains high stereochemical integrity while offering a safer, more environmentally friendly alternative to established industrial practices.
Mechanistic Insights into CDI-Mediated Cyclization and Azide Substitution
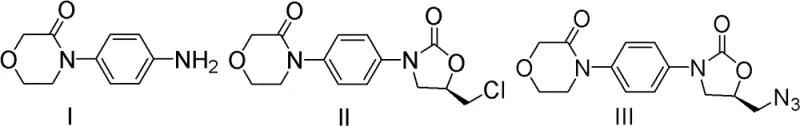
The core of this technological advancement lies in the precise mechanistic execution of the ring-closing and functionalization steps. The reaction begins with the nucleophilic attack of the aniline nitrogen on the less hindered carbon of (R)-epichlorohydrin, establishing the critical chiral center that defines the biological activity of the final drug. Following this, the addition of CDI activates the hydroxyl group, promoting an intramolecular cyclization that forms the five-membered oxazolidinone ring. This mechanism is particularly advantageous because CDI acts as a dehydrating coupling agent that generates imidazole as a benign byproduct, avoiding the formation of corrosive acids or salts that could degrade the sensitive morpholine moiety. The reaction kinetics are optimized by selecting solvents such as tetrahydrofuran or 2-methyltetrahydrofuran, which provide the necessary polarity to dissolve intermediates while maintaining thermal stability during the 5-24 hour reflux period. This careful control of reaction parameters ensures that the intermediate is formed with minimal racemization, preserving the optical purity required for regulatory compliance.
Subsequent functionalization involves a nucleophilic substitution where the chloromethyl group is displaced by an azide ion using sodium azide in polar aprotic solvents like DMF or DMSO. This step is conducted at moderate temperatures ranging from 40°C to 100°C, which provides sufficient activation energy for the substitution without triggering decomposition of the azide functionality. The choice of solvent is critical here; DMF and DMSO effectively solvate the sodium cation, increasing the nucleophilicity of the azide anion and driving the reaction to completion within 6-15 hours. This high-yield transformation is pivotal as it installs the nitrogen handle required for the final amide bond formation in the API. By avoiding harsh acidic or basic workups, the process minimizes the generation of impurities, resulting in a crude product that requires less intensive purification. This mechanistic elegance translates directly to process robustness, ensuring that the high-purity rivaroxaban intermediate can be produced consistently even at large scales.
How to Synthesize Rivaroxaban Intermediate Efficiently
The practical implementation of this synthesis route is designed for straightforward execution in standard chemical processing equipment. The process initiates by dissolving 4-morpholinone aniline in an organic solvent such as isopropanol and adding (R)-epichlorohydrin, followed by heating to reflux for approximately 16 hours to ensure complete ring-opening. After concentrating the reaction mixture, the residue is redissolved in dichloromethane, and CDI is added to effect cyclization, yielding the chloromethyl oxazolidinone intermediate after standard aqueous workup and silica gel chromatography. The second phase involves reacting this intermediate with sodium azide in DMF at 80°C for 12 hours, followed by extraction and concentration to isolate the azide derivative. Detailed standardized synthesis steps see the guide below.
- React 4-morpholinone aniline with (R)-epichlorohydrin under reflux, followed by cyclization with carbonyldiimidazole (CDI) to form the chloromethyl oxazolidinone intermediate.
- Perform nucleophilic substitution using sodium azide in polar aprotic solvents like DMF or DMSO at moderate temperatures (40-100°C) to introduce the azide group.
- Execute catalytic hydrogenation to reduce the azide to an amine, followed by acylation with 5-chlorothiophene-2-carbonyl chloride to finalize the rivaroxaban structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents like phosgene and butyllithium removes the need for specialized storage facilities and complex safety protocols, which directly correlates to a reduction in operational overhead and insurance costs. Furthermore, the use of readily available, commodity-grade solvents and reagents ensures that the supply chain is resilient against market fluctuations that often affect specialty chemicals. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients. By simplifying the process flow, manufacturers can achieve higher throughput rates, effectively reducing lead time for high-purity anticoagulants and enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the substitution of expensive and dangerous reagents with cost-effective alternatives like CDI and sodium azide. The avoidance of cryogenic cooling systems significantly lowers energy consumption, while the simplified workup procedures reduce solvent usage and waste disposal costs. Additionally, the high atom economy of the cyclization step minimizes raw material waste, contributing to substantial cost savings in the overall production budget. These efficiencies allow for a more competitive pricing structure without compromising on quality, making it an attractive option for cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous raw materials mitigates the risk of supply disruptions caused by regulatory restrictions on toxic substances. Since the process does not require exotic catalysts or custom-synthesized building blocks, sourcing becomes more predictable and less susceptible to geopolitical or logistical bottlenecks. This reliability ensures that production can be scaled up rapidly to meet surges in demand, providing a secure foundation for long-term supply agreements. Consequently, partners can depend on a consistent flow of intermediates, safeguarding their own downstream manufacturing operations against volatility.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns perfectly with increasingly stringent global regulations regarding chemical emissions and waste management. The absence of heavy metals and toxic gases simplifies the permitting process for new production lines and reduces the burden on effluent treatment plants. The mild reaction conditions also extend the lifespan of reactor vessels and ancillary equipment by reducing corrosion and thermal stress. This durability supports the commercial scale-up of complex pharmaceutical intermediates, enabling facilities to transition from pilot batches to multi-ton production with confidence and regulatory ease.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rivaroxaban intermediate synthesis. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their specific manufacturing contexts.
Q: How does this synthesis method improve safety compared to traditional routes?
A: Unlike conventional methods that rely on hazardous reagents like phosgene or butyllithium and require strict anhydrous/oxygen-free conditions, this patented process utilizes mild reaction conditions and safer reagents like CDI and sodium azide, significantly reducing operational risks and environmental impact.
Q: What ensures the stereoselectivity of the final rivaroxaban product?
A: The process initiates with chiral (R)-epichlorohydrin, which dictates the stereochemistry early in the synthesis. The subsequent ring-closing and substitution steps proceed with high retention of configuration, ensuring the final API meets the rigorous enantiomeric purity standards required for clinical efficacy.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It avoids cryogenic temperatures and expensive catalysts, uses common organic solvents, and features a 'one-pot' capability for the initial steps, which simplifies equipment requirements and enhances throughput for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivaroxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102250077B are fully realized in practical application. We are committed to delivering intermediates that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch supports the high safety standards required for anticoagulant therapies. Our infrastructure is designed to handle the specific solvent systems and thermal profiles of this novel route, providing a seamless bridge between innovative chemistry and commercial reality.
We invite global partners to collaborate with us to leverage these technological advancements for their supply chains. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this superior synthesis method. We encourage you to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Together, we can optimize the production of life-saving medications while driving down costs and enhancing supply security.
