Scalable Synthesis of 4-(4-Aminophenyl)morpholine-3-one for Rivaroxaban Production
Introduction to Advanced Rivaroxaban Intermediate Synthesis
The pharmaceutical landscape for anticoagulant therapies has shifted dramatically towards direct oral anticoagulants, with Rivaroxaban standing as a cornerstone treatment for venous thrombosis and pulmonary embolism. Central to the efficient manufacturing of this blockbuster drug is the availability of high-quality key intermediates, specifically 4-(4-aminophenyl)morpholine-3-one. A recent technological breakthrough detailed in patent CN115260117A introduces a robust, environmentally friendly synthesis method that addresses long-standing inefficiencies in the supply chain. This novel approach utilizes diethylene glycol as a primary feedstock, bypassing the complex and hazardous steps associated with traditional phthalimide or ethanolamine-based routes. By streamlining the oxidative cyclization and condensation processes, this technology offers a compelling value proposition for manufacturers seeking to optimize cost structures while maintaining rigorous quality standards required for API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-(4-aminophenyl)morpholine-3-one has been plagued by low yields, hazardous reagents, and difficult purification challenges. Early attempts utilizing ethanolamine and chloroacetyl chloride often failed to achieve the desired ring closure efficiently, leading to significant material loss. Furthermore, alternative pathways relying on p-bromonitrobenzene encountered similar bottlenecks where the critical ring-closing step did not proceed to the target product effectively. Perhaps most concerning for industrial safety and scalability was the phthalimide-based route, which, while offering cheap starting materials, relied on sodium chlorite for a critical oxidation step. As illustrated in the reaction scheme below, this oxidation process is notoriously difficult to control thermally.
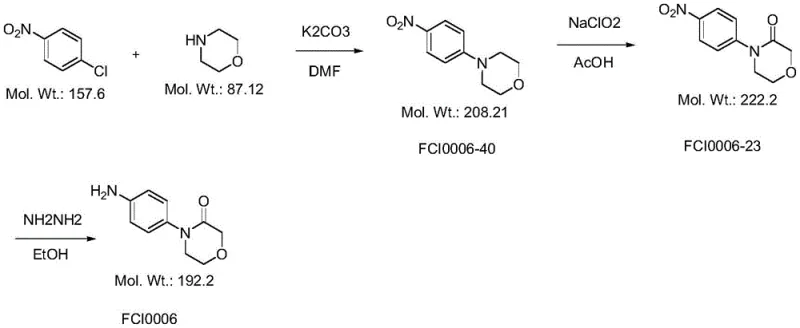
In practical testing of the phthalimide method, temperatures exceeding 30°C triggered the release of large amounts of yellow-green smoke, indicating unstable reaction conditions and potential safety hazards. Conversely, maintaining temperatures below 20°C resulted in unacceptably slow reaction kinetics. These thermal instabilities create significant operational risks for large-scale reactors, necessitating expensive cooling infrastructure and rigorous monitoring systems that drive up operational expenditures. Additionally, the reliance on heavy metal catalysts in some variations of these older routes introduces the risk of metal residues in the final product, complicating the regulatory approval process for the final API.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally reimagines the synthetic pathway by employing diethylene glycol as the foundational building block. This strategy eliminates the need for hazardous halogenated precursors and unstable oxidation reagents. Instead, the process initiates with a controlled oxidative cyclization to form a morpholine-3-one core, followed by a straightforward condensation with p-nitroaniline. This sequence not only simplifies the operational workflow but also significantly enhances the overall atom economy of the synthesis. By avoiding the generation of toxic byproducts and minimizing the use of exotic reagents, the new route aligns perfectly with modern green chemistry principles. The simplicity of the post-treatment procedures, involving standard extraction and crystallization techniques, further reduces the time and solvent consumption typically associated with intermediate manufacturing.
Mechanistic Insights into Oxidative Cyclization and Condensation
The core of this technological advancement lies in the efficient oxidative cyclization of diethylene glycol. In the first step, the linear diol undergoes a transformation mediated by a catalytic system, preferably involving TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) and a co-oxidant such as trichloroisocyanuric acid. This catalytic cycle selectively oxidizes the hydroxyl groups to facilitate intramolecular ring closure, yielding the morpholine-3-one derivative (Compound 3) with high selectivity. The use of TEMPO allows the reaction to proceed under mild conditions, typically between 0°C and room temperature, preventing the degradation of the sensitive ether linkage. This precise control over the oxidation state is crucial for minimizing side reactions that could lead to polymeric impurities or over-oxidized species, thereby ensuring a clean crude profile before the subsequent condensation step.
Following the formation of the morpholine core, the synthesis proceeds via an acid-catalyzed condensation with p-nitroaniline. This step is critical for establishing the aryl-amine bond that defines the final molecular architecture. The reaction is conducted in high-boiling solvents such as DMSO or decalin at temperatures ranging from 130°C to 180°C, driven by strong organic acids like p-toluenesulfonic acid. The mechanism involves the nucleophilic attack of the aniline nitrogen on the carbonyl carbon of the morpholine intermediate, followed by dehydration to stabilize the amide linkage. The final reduction of the nitro group to the amine is achieved using a hydrazine-based system, which is highly chemoselective and avoids the use of high-pressure hydrogenation equipment. The complete reaction pathway is depicted below, highlighting the streamlined progression from simple diol to complex heterocycle.
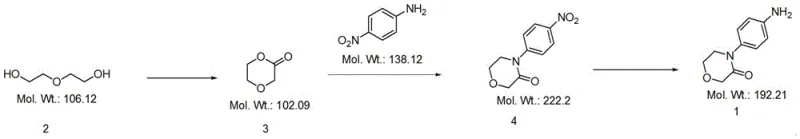
How to Synthesize 4-(4-Aminophenyl)morpholine-3-one Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and temperature gradients, particularly during the oxidative and condensation phases. The patent outlines a reproducible three-step sequence that balances reaction kinetics with safety profiles, making it ideal for pilot plant and commercial scale operations. Operators must ensure precise addition rates for the oxidant in the first step to manage exotherms, while the condensation step benefits from extended reaction times to maximize conversion. For a comprehensive understanding of the specific operational parameters, including solvent volumes, addition rates, and workup procedures, please refer to the standardized synthesis guide provided below.
- Perform oxidative cyclization on diethylene glycol using an oxidant like TEMPO/trichloroisocyanuric acid to obtain Compound 3.
- React Compound 3 with p-nitroaniline in the presence of an acid catalyst (e.g., p-toluenesulfonic acid) at elevated temperatures to form Compound 4.
- Reduce Compound 4 using a hydrazine hydrate and iron chloride system under reflux to yield the final target product 1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this diethylene glycol-based route represents a strategic opportunity to de-risk the supply of Rivaroxaban intermediates. The shift away from hazardous oxidants like sodium chlorite eliminates the need for specialized containment systems and reduces the regulatory burden associated with handling dangerous goods. Furthermore, the use of commodity chemicals such as diethylene glycol and p-nitroaniline ensures a stable and diversified supply base, insulating manufacturers from the volatility often seen with specialized halogenated starting materials. The simplified purification process also translates to reduced solvent consumption and lower waste disposal costs, contributing to a more sustainable and cost-efficient manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts and the use of readily available bulk chemicals significantly lowers the raw material cost per kilogram. By avoiding complex purification steps required to remove metal residues, the downstream processing costs are drastically reduced, leading to substantial overall savings in the cost of goods sold. The high yield reported in the condensation step further maximizes the utility of every unit of input material, enhancing the economic efficiency of the entire production line.
- Enhanced Supply Chain Reliability: Sourcing diethylene glycol and p-nitroaniline is far more reliable than procuring specialized brominated intermediates or phthalimide derivatives, which may be subject to supply constraints from fewer vendors. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by batch failures or safety incidents. This reliability ensures consistent delivery timelines for API manufacturers, allowing for better inventory planning and reduced safety stock requirements across the global supply network.
- Scalability and Environmental Compliance: The absence of toxic gas evolution and the manageable thermal profile of the new route make it inherently safer for scale-up to multi-ton quantities. Waste streams are simpler to treat, primarily consisting of aqueous salts and organic solvents that can be recovered or neutralized using standard protocols. This alignment with environmental regulations facilitates smoother permitting processes for new manufacturing facilities and supports corporate sustainability goals by minimizing the ecological impact of pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on yield expectations, safety profiles, and scalability factors. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the diethylene glycol route superior to the phthalimide method?
A: The phthalimide route utilizes sodium chlorite for oxidation, which presents significant safety hazards due to exothermic heat release and difficult temperature control above 30°C. The new diethylene glycol route avoids these thermal risks and uses cheaper, more accessible starting materials.
Q: What is the purity profile of the final intermediate?
A: The optimized reduction step using hydrazine hydrate and iron chloride yields the target product with a purity of approximately 99.5%, significantly reducing the burden on downstream purification processes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method features simple reaction steps, high atom economy, and avoids the use of heavy metal catalysts that leave difficult-to-remove residues, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Aminophenyl)morpholine-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your final API production depends heavily on the quality and consistency of your intermediates. Our technical team has extensively analyzed this novel diethylene glycol pathway and possesses the expertise to implement it with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of 4-(4-aminophenyl)morpholine-3-one meets the exacting standards required for cardiovascular drug manufacturing.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security for your Rivaroxaban production program.
