Advanced Manufacturing Strategy for Etoricoxib: Technical Breakthroughs and Commercial Scalability
The pharmaceutical landscape for non-steroidal anti-inflammatory drugs (NSAIDs) continues to evolve, with Etoricoxib standing out as a potent selective cyclooxygenase-2 (COX-2) inhibitor. The technical disclosure within patent CN103204803A presents a pivotal advancement in the synthetic methodology for this critical active pharmaceutical ingredient. This patent outlines a robust pathway that addresses historical inefficiencies in constructing the complex bipyridine core, specifically focusing on the transformation of key sulfide intermediates into the final sulfone structure. By leveraging acid-catalyzed condensation reactions followed by controlled oxidation, this approach offers a compelling alternative to traditional palladium-catalyzed couplings or hazardous phosphorus-based cyclizations. For industry stakeholders, understanding the nuances of this synthesis is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials consistently. The strategic shift towards safer reagents and more manageable reaction conditions underscores a broader industry trend towards sustainable and scalable chemical manufacturing.
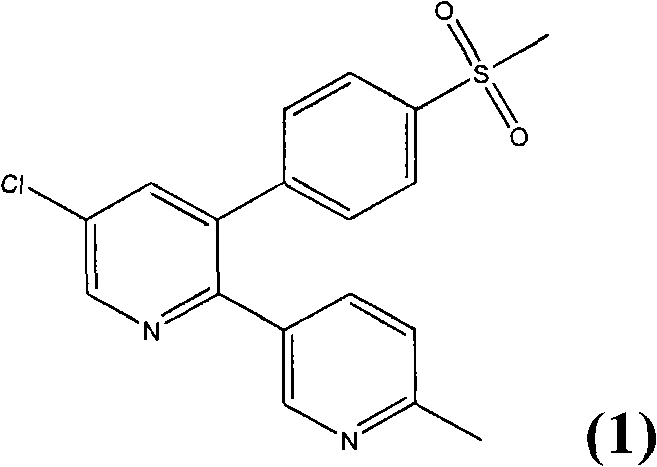
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Etoricoxib and its precursors has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up. Early methodologies, such as those described in EP 0 912 518 B1, relied heavily on palladium-catalyzed cross-coupling reactions between aryl halides and boron or stannane species. While chemically effective on a small scale, these routes are notoriously expensive due to the high cost of precious metal catalysts and the complexity of removing trace metal impurities to meet stringent regulatory standards. Furthermore, alternative routes disclosed in documents like EP 1 071 745 B1 utilized highly hazardous reagents such as phosphoryl chloride and hexafluorophosphate salts to facilitate ring closure. These substances pose severe occupational health risks and generate toxic waste streams that complicate environmental compliance and increase disposal costs. The reliance on such dangerous chemicals not only elevates the operational risk profile but also necessitates specialized equipment and rigorous safety protocols, thereby inflating the overall capital expenditure required for production facilities.
The Novel Approach
In stark contrast, the methodology detailed in CN103204803A introduces a streamlined process that circumvents these critical bottlenecks by utilizing readily available and economically favorable starting materials. The core innovation lies in the acid-catalyzed condensation of methylthio benzyl-pyridyl ketone with 2-chloroacrolein or its acetoxy derivatives. This reaction pathway avoids the need for expensive transition metals and eliminates the use of highly toxic phosphorus reagents, resulting in a significantly safer operational environment. The process capitalizes on the inherent reactivity of chloroacrolein derivatives under acidic conditions to construct the pyridine ring with high efficiency. By shifting the synthetic logic towards simple condensation and oxidation steps, the method drastically simplifies the workflow, reducing the number of unit operations required. This simplification translates directly into enhanced process robustness, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing while maintaining the high quality required for downstream drug formulation.
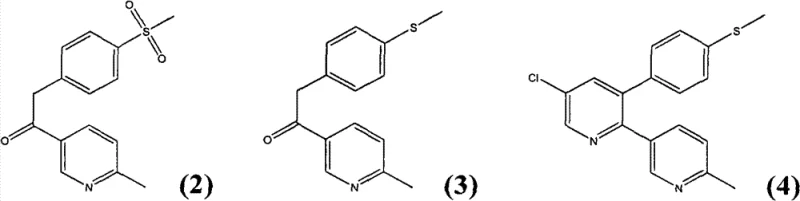
Mechanistic Insights into Acid-Catalyzed Cyclization and Oxidation
The chemical elegance of this synthesis is rooted in the precise control of reaction conditions to favor the formation of the desired bipyridine scaffold. The initial step involves the condensation of the ketone precursor with the chloroacrolein derivative in the presence of an acid and an ammonium salt. This environment promotes a cyclization reaction that effectively closes the pyridine ring, yielding the 5-chloro-3-(4-methylthio phenyl)-6'-methyl-[2,3']dipyridyl intermediate. The choice of acid, ranging from acetic to propionic acid, and the use of ammonium salts like ammonium acetate are critical for driving the equilibrium towards product formation without degrading the sensitive functional groups. Following the cyclization, the intermediate exists as a sulfide, which possesses distinct physicochemical properties compared to the final sulfone target. Specifically, the sulfide intermediate demonstrates superior solubility in organic solvents such as ethyl acetate, hexane, and tert-butyl methyl ether. This solubility profile is a crucial mechanistic advantage, as it allows for effective purification via recrystallization or extraction before the final oxidation step, ensuring that impurities are removed early in the process.
The final transformation involves the oxidation of the methylthio group to the methylsulfonyl group, a step that must be carefully managed to avoid over-oxidation or degradation of the pyridine rings. The patent specifies the use of hydrogen peroxide or peracetic acid in the presence of catalysts such as sodium molybdate or ammonium tungstate. This catalytic system ensures a high-yield conversion while maintaining selectivity for the sulfur atom. The reaction is typically conducted in alcohol solvents like methanol or ethanol at moderate temperatures, further enhancing the safety and scalability of the process. The ability to isolate the intermediate sulfide in high purity before oxidation is a key differentiator, as it prevents the carryover of byproducts into the final API. This two-stage approach—cyclization followed by oxidation—provides a modular framework that allows for rigorous quality control at the intermediate stage, ultimately delivering a high-purity pharmaceutical intermediate that meets the exacting standards of global regulatory bodies.
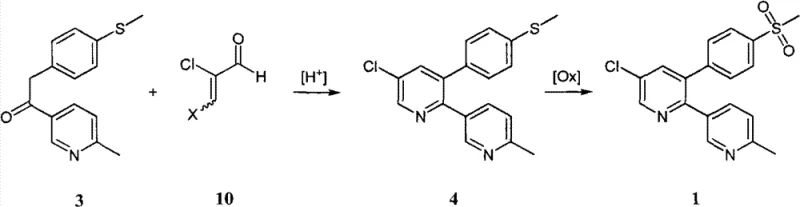
How to Synthesize Etoricoxib Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters that govern yield and purity. The process begins with the preparation of the reaction mixture containing the ketone and aldehyde precursors in a suitable acid solvent, followed by heating to facilitate ring closure. Once the cyclization is complete, the workup involves neutralization and extraction to isolate the sulfide intermediate, which can be further purified by recrystallization from hydrocarbon solvents. The final oxidation step is then performed on the purified intermediate using aqueous hydrogen peroxide and a molybdate catalyst. Detailed standard operating procedures for temperature control, reagent addition rates, and purification steps are critical for reproducibility. For a comprehensive breakdown of the specific experimental conditions and stoichiometry required for optimal results, please refer to the technical guide below.
- Condense methylthio benzyl-pyridyl ketone with 2-chloroacrolein or its acetoxy derivative under acidic conditions with an ammonium salt to form the sulfide intermediate.
- Isolate the 5-chloro-3-(4-methylthio phenyl)-6-methyl-[2,3]dipyridyl intermediate using organic solvent extraction and recrystallization.
- Oxidize the sulfide intermediate to the final sulfone product using hydrogen peroxide and a molybdate or tungstate catalyst in an alcohol solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement and supply chain management teams seeking to optimize their sourcing strategies. The elimination of palladium catalysts and hazardous phosphorus reagents results in a substantial reduction in raw material costs and waste disposal expenses. Furthermore, the use of commodity chemicals like 2-chloroacrolein and acetic acid ensures a stable and reliable supply chain, mitigating the risks associated with sourcing specialized or controlled substances. The improved solubility of the intermediate also simplifies downstream processing, reducing solvent consumption and energy requirements during purification. These factors collectively contribute to a more resilient and cost-effective manufacturing model that can withstand market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The removal of expensive precious metal catalysts and the substitution of hazardous reagents with common acids and oxidants lead to significant savings in material costs. Additionally, the simplified purification process reduces solvent usage and waste treatment expenses, further enhancing the economic viability of the production line. This efficiency allows for competitive pricing without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials such as chloroacrolein derivatives and simple ammonium salts, the risk of supply disruptions is minimized. This accessibility ensures consistent production schedules and reliable delivery timelines, which are critical for maintaining inventory levels and meeting customer demand in the fast-paced pharmaceutical market.
- Scalability and Environmental Compliance: The avoidance of toxic reagents simplifies environmental compliance and reduces the regulatory burden associated with waste management. The process is inherently safer and more amenable to scale-up, allowing for seamless transition from pilot batches to commercial production volumes without the need for specialized containment equipment or extensive safety modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Etoricoxib via this patented method. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on the feasibility and advantages of this approach. Understanding these details is crucial for technical teams evaluating the potential for technology transfer or process optimization within their own facilities.
Q: How does this synthesis method improve safety compared to prior art?
A: This method eliminates the need for hazardous reagents such as phosphoryl chloride and hexafluorophosphate salts used in previous routes, significantly reducing occupational health risks and environmental waste.
Q: What are the solubility advantages of the intermediate compound?
A: The sulfide intermediate exhibits superior solubility in common organic solvents like ethyl acetate and hexane compared to the final sulfone, allowing for effective purification before the final oxidation step.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of readily available raw materials like 2-chloroacrolein and the avoidance of expensive palladium catalysts make this route highly scalable and economically viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etoricoxib Supplier
The technical potential of this synthesis route is immense, offering a pathway to high-quality Etoricoxib that aligns with modern manufacturing standards. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against global pharmacopoeia standards. We understand the critical nature of API intermediates in the drug development lifecycle and are dedicated to providing materials that facilitate seamless downstream processing.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this route. We encourage you to contact us for specific COA data and route feasibility assessments to ensure that our capabilities align perfectly with your production goals. Let us partner with you to drive efficiency and innovation in your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
