Scalable Manufacturing of Sarpogrelate Hydrochloride via Novel Purification Technology
Introduction to Advanced Sarpogrelate Hydrochloride Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic viability, particularly for complex cardiovascular agents like Sarpogrelate Hydrochloride. Patent CN101239920A introduces a transformative methodology that addresses the longstanding challenge of removing triphenylphosphine oxide (TPPO) generated during the initial Wittig reaction. Traditionally, the presence of TPPO has necessitated expensive and labor-intensive silica gel chromatography, creating a significant bottleneck for commercial scale-up. This new technical disclosure outlines a streamlined pathway where the crude Wittig mixture undergoes direct catalytic hydrogenation, bypassing intermediate purification entirely. By leveraging specific solubility characteristics during the subsequent amination and esterification stages, manufacturers can achieve purity levels exceeding 99% without chromatographic intervention. 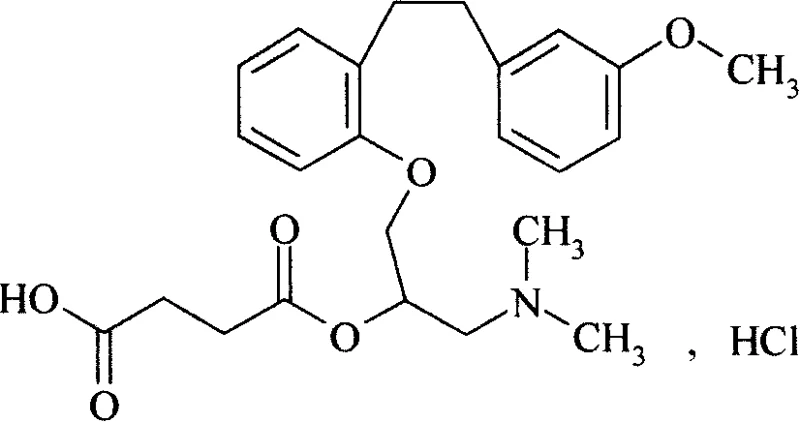
This strategic shift not only simplifies the operational workflow but also drastically reduces the environmental footprint associated with large-scale solvent usage in column chromatography. For R&D directors and process chemists, understanding the nuances of this hydrogenation-first approach is critical for optimizing yield and minimizing impurity profiles. The patent details specific catalytic systems, including Palladium on Carbon (Pd-C) and Raney Nickel, operating under moderate pressures of 0.1 to 1 MPa. These conditions ensure complete reduction of the styryl double bond while maintaining the integrity of the methoxy and phenolic functionalities. The ability to carry forward the TPPO impurity through multiple synthetic steps, only to discard it efficiently at the final salt formation stage, represents a significant leap forward in process chemistry efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sarpogrelate intermediates has been plagued by the difficulties associated with purifying 2-((3-methoxyl)styryl)phenol. As documented in prior art such as J. Med. Chem. 1990, the Wittig reaction inherently produces stoichiometric amounts of triphenylphosphine oxide. Conventional wisdom dictated that this byproduct must be removed immediately via silica gel column chromatography before proceeding to hydrogenation. While effective on a gram scale in a laboratory setting, this technique is economically disastrous for industrial manufacturing. The consumption of vast quantities of silica gel and elution solvents drives up operational costs exponentially. Furthermore, the throughput of column chromatography is inherently low, creating a severe bottleneck that limits the total production capacity of the facility. Attempting to scale this method to multi-kilogram or metric-ton batches often results in inconsistent purity and prolonged cycle times, making it unsuitable for reliable supply chain management.
The Novel Approach
The innovative strategy presented in the patent data fundamentally reimagines the purification sequence by deferring the removal of triphenylphosphine oxide until the final stages of synthesis. Instead of isolating the pure alkene intermediate, the crude mixture containing substantial amounts of TPPO is subjected directly to catalytic hydrogenation. This bold move eliminates an entire unit operation from the front end of the process. Following hydrogenation, the resulting phenol intermediate reacts with epichlorohydrin and dimethylamine to form the key amino-alcohol intermediate. 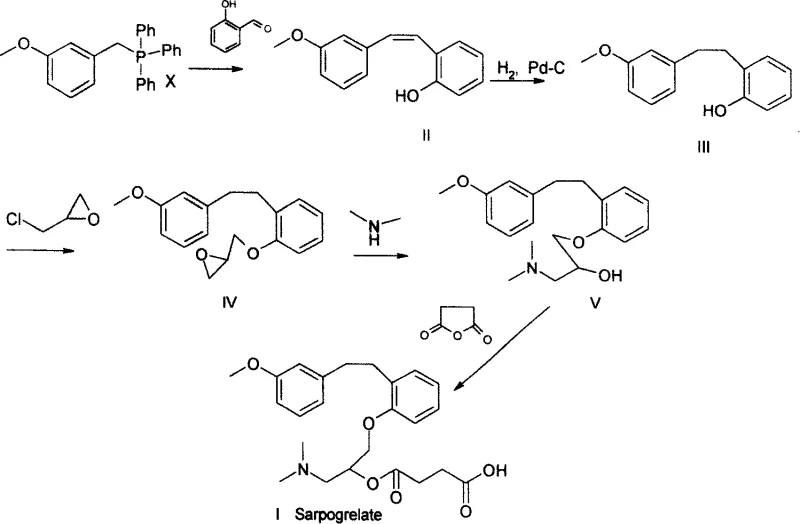
At this juncture, the process exploits the distinct physicochemical properties of the amine intermediate versus the neutral TPPO. Through careful pH control and solvent selection, the amine can be extracted into an aqueous acidic phase, leaving the lipophilic TPPO behind in the organic layer. Alternatively, the TPPO remains dissolved in the mother liquor during the final crystallization of the Sarpogrelate hydrochloride salt. This approach transforms a difficult separation problem into a straightforward liquid-liquid extraction or crystallization event, both of which are highly scalable unit operations. The result is a continuous, fluid manufacturing process that avoids the stop-start nature of batch chromatography, ensuring consistent quality and significantly improved throughput for commercial production.
Mechanistic Insights into Catalytic Hydrogenation and Selective Separation
The core of this technological advancement lies in the robustness of the catalytic hydrogenation step and the subsequent exploitation of solubility differentials. The patent specifies the use of heterogeneous catalysts such as 5% Pd-C, Raney Nickel, or Platinum Oxide, which facilitate the reduction of the carbon-carbon double bond in the styryl moiety. The reaction is typically conducted in alcoholic solvents like ethanol or methanol at temperatures ranging from 0 to 100°C and hydrogen pressures between 0.1 and 1 MPa. Under these conditions, the catalyst selectively reduces the alkene without affecting the aromatic rings or the ether linkages. Crucially, the presence of triphenylphosphine oxide does not poison the catalyst to a degree that inhibits conversion, allowing for high yields (reported up to 96-98%) even in the presence of the impurity. This tolerance is vital for maintaining process efficiency and avoiding the need for pre-purification.
Following the formation of the amino-alcohol intermediate, the separation mechanism relies on the basicity of the dimethylamine group. By treating the reaction mixture with an inorganic acid such as hydrochloric acid, the amine is converted into its water-soluble ammonium salt. In contrast, triphenylphosphine oxide remains neutral and retains high solubility in organic solvents like ethyl acetate or toluene. This allows for a clean phase separation where the aqueous layer contains the desired product precursor, and the organic layer retains the phosphine oxide waste. Subsequent basification and extraction recover the free amine, which is then esterified with succinic anhydride. The final purification involves recrystallization of the hydrochloride salt from solvents like methanol, ethanol, or acetonitrile. These solvents are chosen because Sarpogrelate Hydrochloride has moderate solubility in them at reflux but low solubility at room temperature, whereas TPPO remains soluble, ensuring the final API intermediate meets stringent purity specifications of greater than 99%.
How to Synthesize Sarpogrelate Hydrochloride Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity carryover. The process begins with the hydrogenation of the crude Wittig product, followed by nucleophilic substitution with epichlorohydrin and amination. The critical control point is the pH adjustment during the extraction phase to ensure quantitative transfer of the amine into the aqueous layer. Detailed standard operating procedures regarding catalyst loading, hydrogen pressure, and solvent ratios are essential for reproducibility. For a comprehensive breakdown of the specific experimental conditions, reagent quantities, and workup procedures described in the patent, please refer to the standardized synthesis guide below.
- Hydrogenate the crude Wittig reaction mixture containing triphenylphosphine oxide directly using Pd-C or Raney Nickel catalyst without prior purification.
- React the resulting phenol intermediate with epichlorohydrin and dimethylamine to form the amino-alcohol intermediate while retaining impurities in the organic phase.
- Perform esterification with succinic anhydride followed by salt formation and recrystallization using methanol or ethanol to isolate high-purity Sarpogrelate Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the elimination of silica gel chromatography represents a monumental shift in cost structure and operational reliability. Traditional chromatographic processes are not only expensive due to material costs but are also prone to variability and downtime. By replacing this with catalytic hydrogenation and crystallization, the manufacturing process becomes significantly more robust and predictable. The removal of the chromatography step drastically simplifies the equipment requirements, allowing production to take place in standard stainless steel reactors rather than specialized glass columns. This transition facilitates a smoother scale-up from pilot plant to commercial manufacturing, reducing the lead time for process validation and regulatory approval. Furthermore, the reduction in solvent volume associated with column elution translates to lower waste disposal costs and a reduced environmental burden, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this protocol is the complete avoidance of silica gel and the massive volumes of elution solvents typically required for purification. Chromatography is notoriously inefficient in terms of solvent usage, often requiring hundreds of liters of solvent per kilogram of product. By shifting to a crystallization-based purification strategy, the process significantly lowers the raw material input for solvents and consumables. Additionally, the ability to recycle mother liquors in crystallization processes further enhances material efficiency. The use of common, commodity-grade catalysts like Pd-C instead of specialized chromatographic media also contributes to a leaner bill of materials. These factors combine to create a substantially lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for cardiovascular intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex purification steps that have low throughput and high failure rates. The novel hydrogenation and extraction method described herein utilizes unit operations that are standard in the fine chemical industry. Hydrogenation reactors and centrifuges for crystal isolation are widely available and easy to maintain, reducing the risk of equipment-related production delays. The robustness of the chemistry, which tolerates the presence of triphenylphosphine oxide throughout the early stages, means that variations in the quality of the starting Wittig mixture do not necessarily derail the entire batch. This tolerance builds resilience into the supply chain, ensuring that delivery schedules can be met consistently even when facing minor fluctuations in raw material quality. It allows for larger batch sizes to be processed in single runs, improving overall facility utilization rates.
- Scalability and Environmental Compliance: Scaling a chromatographic process from grams to tons is a non-linear challenge that often requires disproportionate increases in floor space and manpower. In contrast, the proposed method scales linearly; increasing the reactor size directly increases output without changing the fundamental physics of the separation. This makes it ideal for meeting the growing global demand for Sarpogrelate. From an environmental perspective, the reduction in hazardous waste generation is significant. Silica gel waste is difficult to dispose of, and the mixed solvent streams from chromatography require complex distillation for recovery. The new process generates primarily aqueous waste streams and organic solvents that are easier to recover and reuse. This alignment with environmental, social, and governance (ESG) goals is increasingly important for pharmaceutical buyers who are under pressure to reduce the carbon footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Sarpogrelate Hydrochloride using this advanced methodology. These insights are derived directly from the experimental data and claims within the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding supplier qualification and process adoption. The answers reflect the practical realities of implementing this chemistry in a GMP-compliant environment.
Q: How does this process handle Triphenylphosphine Oxide (TPPO) impurities?
A: Unlike traditional methods requiring silica gel chromatography, this novel approach leverages solubility differences. TPPO is retained in the organic mother liquor during the acid-base extraction of the amine intermediate or during the final salt crystallization, eliminating the need for column purification.
Q: What are the optimal recrystallization solvents for Sarpogrelate Hydrochloride?
A: The patent data indicates that alcohols such as methanol and ethanol, or nitriles like acetonitrile, either alone or in mixtures, provide superior recrystallization results compared to acetone, yielding purity levels exceeding 99%.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, by removing the bottleneck of chromatographic purification and utilizing standard catalytic hydrogenation and batch crystallization techniques, the process is highly scalable and economically viable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sarpogrelate Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team has extensively analyzed the pathways described in CN101239920A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that achieving the reported purity levels of greater than 99% requires not just the right recipe, but rigorous process control and state-of-the-art analytical capabilities. Our facilities are equipped with high-pressure hydrogenation reactors and advanced crystallization units capable of executing this specific purification logic with precision. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of Sarpogrelate Hydrochloride meets the exacting standards required for pharmaceutical applications, free from residual triphenylphosphine oxide.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and scalable technology. By optimizing the supply chain for this critical cardiovascular intermediate, we can help you reduce lead times and secure a stable source of high-quality material. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patent technology can add value to your portfolio. Let us be your strategic partner in delivering reliable, high-purity Sarpogrelate Hydrochloride to the market.
