Advanced Manufacturing Strategy for Sarpogrelate Hydrochloride: Eliminating Chromatographic Bottlenecks
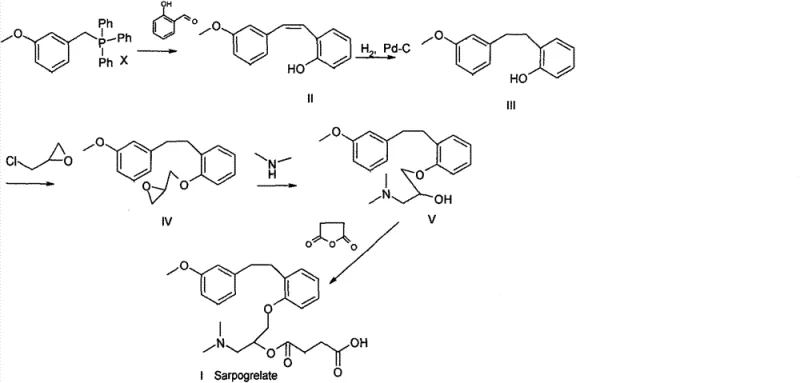
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic viability, particularly for cardiovascular agents like Sarpogrelate Hydrochloride. Patent CN101239920B, filed in 2013, presents a transformative approach to the manufacturing of this serotonin receptor antagonist, specifically addressing the notorious purification challenges associated with Wittig reaction byproducts. Traditionally, the synthesis of the key intermediate involves a Wittig olefination that generates stoichiometric amounts of triphenylphosphine oxide (TPPO), a byproduct that is notoriously difficult to separate from the desired phenolic intermediate using standard crystallization techniques. This patent discloses a novel methodology wherein the crude reaction mixture, containing significant levels of TPPO ranging from 10% to 90%, is subjected directly to catalytic hydrogenation without prior chromatographic purification. This strategic deviation from conventional protocols eliminates the need for expensive and labor-intensive silica gel column chromatography, thereby streamlining the production workflow for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Sarpogrelate precursors has been hindered by the reliance on rigorous purification steps immediately following the Wittig reaction. Standard literature procedures, such as those cited in the Journal of Medicinal Chemistry, mandate the use of silicagel column chromatography to isolate the pure 2-((3-methoxyl)styryl)phenol intermediate before any subsequent transformations can occur. This dependency creates a severe bottleneck in cost reduction in API manufacturing because column chromatography is inherently batch-limited, consumes vast quantities of organic solvents, and suffers from significant product adsorption losses on the silica matrix. Furthermore, the scalability of column chromatography is poor; transitioning from gram-scale laboratory synthesis to multi-kilogram or ton-scale commercial production requires disproportionately large equipment footprints and generates substantial hazardous waste, making it economically unfeasible for high-volume generic drug production.
The Novel Approach
In stark contrast, the methodology outlined in CN101239920B circumvents these inefficiencies by embracing the crude nature of the Wittig product. The inventors discovered that the presence of triphenylphosphine oxide does not inhibit the subsequent catalytic hydrogenation step, allowing the double bond reduction to proceed with high conversion rates even in the contaminated mixture. By deferring the purification of TPPO to later stages—specifically leveraging the differential solubility of the amine intermediates versus the phosphine oxide during acid-base extraction and final salt crystallization—the process achieves a drastic simplification of the workflow. This approach not only reduces the number of unit operations but also minimizes solvent consumption and waste generation, aligning perfectly with the goals of green chemistry and enhanced supply chain reliability for complex pharmaceutical intermediates.
Mechanistic Insights into Catalytic Hydrogenation and Impurity Management
The core chemical innovation lies in the tolerance of the hydrogenation catalyst to phosphine oxide contaminants. Typically, phosphorus compounds are known catalyst poisons for noble metals like palladium and platinum; however, the specific conditions employed here—using solvents such as ethanol, methanol, or ethyl acetate with catalysts like 5% Pd-C or Raney Nickel at pressures between 0.1 MPa and 1 MPa—facilitate the selective reduction of the carbon-carbon double bond. The reaction proceeds efficiently at temperatures ranging from 0°C to 100°C, converting the styryl moiety into the corresponding phenylethyl structure without requiring the removal of the phosphine oxide first. This mechanistic resilience allows manufacturers to bypass the most fragile step of the synthesis, ensuring that the commercial scale-up of complex pharmaceutical intermediates remains robust against variations in upstream Wittig reaction quality.
Following hydrogenation, the management of impurities shifts from physical separation (chromatography) to physicochemical partitioning. The subsequent reaction with epichlorohydrin and dimethylamine generates a tertiary amine intermediate which possesses distinct basic properties compared to the neutral triphenylphosphine oxide. By manipulating the pH of the reaction medium, the amine can be selectively protonated and extracted into an aqueous phase, leaving the lipophilic TPPO in the organic layer, or vice versa depending on the specific solvent system employed. This exploitation of pKa differences and solubility profiles ensures that the final esterification with succinic anhydride occurs with a significantly purer substrate, ultimately yielding Sarpogrelate Hydrochloride with purity exceeding 98% and often reaching 99.5% after a single recrystallization step.
How to Synthesize Sarpogrelate Hydrochloride Efficiently
The synthesis protocol detailed in the patent offers a clear pathway for R&D teams to replicate this high-efficiency route. The process begins with the hydrogenation of the crude Wittig mixture, followed by nucleophilic substitution with epichlorohydrin under basic conditions, and concludes with ring opening by dimethylamine and final esterification. This sequence is designed to maximize yield while minimizing purification overhead, making it an ideal candidate for technology transfer. For detailed operational parameters regarding temperature controls, catalyst loading ratios, and specific solvent volumes required for each stage, please refer to the standardized synthesis steps provided in the technical guide below.
- Hydrogenate the crude 2-((3-methoxyl)styryl)phenol mixture containing triphenylphosphine oxide using a metal catalyst like Pd-C or Raney Nickel without prior purification.
- React the resulting intermediate with epichlorohydrin and subsequently with dimethylamine to form the amino-alcohol intermediate, utilizing solubility differences to separate impurities.
- Perform esterification with succinic anhydride followed by salt formation with hydrochloric acid and recrystallization using alcohol-based solvents to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the elimination of chromatographic purification represents a paradigm shift in cost structure and operational agility. By removing the requirement for silica gel and the associated large volumes of elution solvents, the variable costs per kilogram of product are significantly reduced. This process intensification means that production cycles are shorter, as the time-consuming loading and elution phases of column chromatography are entirely excised from the critical path. Consequently, this leads to substantial cost savings in raw material procurement and waste disposal, allowing for more competitive pricing strategies in the global marketplace for cardiovascular therapeutics.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the removal of the silica gel purification step, which is traditionally one of the most expensive unit operations in fine chemical synthesis due to the high cost of chromatography media and the energy required for solvent recovery. By processing the crude mixture directly through hydrogenation and relying on crystallization for final purity, the manufacturer avoids the capital expenditure associated with large-scale chromatography columns and the operational expenditure of disposing of tons of silica waste. This structural change in the process flow results in a leaner manufacturing model that is less sensitive to fluctuations in solvent prices and more resilient to regulatory pressures regarding hazardous waste generation.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the number of potential failure points in the production line. Chromatography is often a source of batch-to-batch variability and yield loss due to tailing or incomplete separation; by replacing this with robust catalytic hydrogenation and crystallization steps, the process becomes more predictable and easier to control. This stability translates to improved on-time delivery performance for clients, as the risk of batch rejection or reprocessing is minimized. Furthermore, the use of common industrial catalysts like Pd-C and readily available solvents ensures that raw material sourcing remains stable and unaffected by niche supply constraints.
- Scalability and Environmental Compliance: The transition from batch chromatography to continuous or large-batch hydrogenation and crystallization facilitates seamless scale-up from pilot plant to commercial production. The reduced solvent intensity of this method aligns with increasingly stringent environmental regulations, lowering the facility's overall environmental footprint. The ability to handle crude intermediates without extensive workup allows for larger batch sizes in existing reactor vessels, effectively increasing the throughput capacity of the manufacturing plant without the need for significant infrastructure expansion, thereby supporting long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on critical process parameters and quality control measures. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement specialists assessing the quality assurance protocols of their suppliers.
Q: How is triphenylphosphine oxide removed without column chromatography?
A: The process utilizes the significant difference in solubility between the intermediate amine salts and triphenylphosphine oxide in specific organic solvents and aqueous phases, allowing separation during extraction and final crystallization.
Q: Does the presence of triphenylphosphine oxide affect the hydrogenation catalyst?
A: Experimental data indicates that catalysts such as Palladium on Carbon (Pd-C) and Raney Nickel remain effective for the hydrogenation of the styryl double bond even in the presence of substantial amounts of triphenylphosphine oxide.
Q: What are the optimal recrystallization solvents for the final product?
A: Lower alcohols such as methanol and ethanol, either alone or in mixtures with esters like ethyl acetate or ketones, provide superior purification compared to acetone-water systems, yielding purity greater than 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sarpogrelate Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in patent literature are faithfully reproduced in our manufacturing facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Sarpogrelate Hydrochloride meets the highest international standards for residual impurities and assay content.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a solid foundation of quality and reliability.
