Optimizing 2,2-Diisopropylpropionitrile Production for High-Purity Cooling Agent Manufacturing
The chemical landscape for producing high-value cooling agent intermediates has evolved significantly with the disclosure of patent CN102093257B, which details a robust method for preparing 2,2-diisopropylpropionitrile. This compound serves as a critical precursor for WS-23, a widely utilized cooling agent found in applications ranging from tobacco products and cosmetics to pharmaceuticals and food additives. The traditional manufacturing pathways have long been plagued by severe operational constraints, including the necessity for cryogenic conditions and the handling of hazardous gaseous reagents that complicate scale-up efforts. This new technical approach fundamentally shifts the paradigm by utilizing isopropyl sulfonic acid esters derived from readily available isopropanol and sulfonyl chlorides. By replacing volatile alkyl halides with stable sulfonate esters, the process mitigates significant safety risks while enhancing the overall economic feasibility of large-scale production. For R&D directors and procurement specialists alike, understanding this shift is vital for securing a reliable supply chain for high-purity flavor and fragrance intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,2-diisopropylpropionitrile relied heavily on methodologies that imposed extreme burdens on industrial infrastructure and operational safety. As illustrated in the prior art reaction schemes, conventional routes often employed lithium diisopropylamide in tetrahydrofuran or sodium amide in liquefied ammonia systems. These methods necessitate the use of liquefied ammonia, which requires specialized low-temperature reactors capable of maintaining temperatures well below -30°C to keep the ammonia in a liquid state. Furthermore, the reliance on gaseous monobromethane or expensive methyl iodide introduces significant handling complexities, as these reagents are difficult to meter accurately in large-scale solid-liquid or gas-liquid systems. The extended reaction times, often exceeding 30 hours in some documented variations, coupled with the need for rigorous refrigeration units, result in substantial energy consumption and prolonged production cycles. These factors collectively render traditional methods economically inefficient and logistically challenging for modern continuous manufacturing environments.
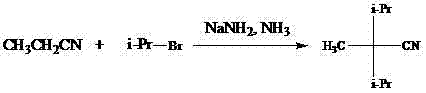
The Novel Approach
In stark contrast to the cumbersome legacy techniques, the novel approach outlined in the patent data introduces a streamlined two-step sequence that operates under significantly milder and more controllable conditions. The core innovation lies in the in situ generation or separate preparation of isopropyl sulfonate esters, which act as superior leaving groups compared to simple halides in this specific nucleophilic substitution context. This modification allows the reaction to proceed effectively at temperatures ranging from 0°C to 120°C, completely eliminating the dependency on cryogenic liquefied ammonia systems. The use of sulfonyl chlorides, such as methanesulfonyl chloride or benzenesulfonyl chloride, provides a versatile platform where the R-group can be tuned to optimize reaction kinetics and purification profiles. By shifting to a liquid-phase reaction system using common organic solvents like pyridine, ethyl acetate, or toluene, the process becomes inherently safer and more amenable to standard stainless-steel reactor setups. This transition not only simplifies the engineering requirements but also drastically reduces the production cycle time, enabling faster turnover and improved responsiveness to market demand fluctuations.
Mechanistic Insights into Sulfonate Ester Activation and Alkylation
The mechanistic elegance of this synthesis lies in the enhanced electrophilicity of the sulfonate ester intermediate, which facilitates a smoother nucleophilic attack by the propionitrile anion. In the first stage, isopropanol reacts with a sulfonyl chloride in the presence of a base like pyridine to form the isopropyl sulfonate ester. This esterification step is highly efficient, often yielding crude products with sufficient purity to proceed directly to the next stage without extensive purification, thereby saving time and solvent resources. The sulfonate group (-OSO2R) is an exceptional leaving group, far superior to hydroxyl groups and often more manageable than halides in terms of side-reaction profiles. When this activated ester encounters the carbanion generated from propionitrile under the influence of a strong base, the nucleophilic substitution occurs rapidly. The steric bulk of the isopropyl group is effectively managed by the high reactivity of the sulfonate leaving group, ensuring that the desired dialkylation at the alpha-position of the nitrile proceeds with high selectivity. This mechanism minimizes the formation of elimination by-products that typically plague reactions involving secondary alkyl halides under basic conditions.
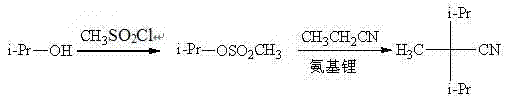
Furthermore, the choice of strong base plays a pivotal role in controlling the impurity profile and ensuring high conversion rates. The patent specifies the use of metal amides, hydrides, or alkoxides, such as lithium amide, sodium hydride, or potassium ethoxide, in a molar ratio of approximately 2:1 relative to the propionitrile. This excess base ensures complete deprotonation of the propionitrile to form the reactive nucleophile while neutralizing any acidic by-products generated during the substitution. The reaction medium, whether it be tetrahydrofuran, toluene, or ethanol, is selected based on its ability to dissolve both the organic reactants and the resulting salts, facilitating homogeneous reaction conditions. Post-reaction workup involves simple aqueous extraction to remove inorganic salts, followed by solvent recovery and vacuum distillation. The distinct boiling point differences between the solvents and the final product allow for efficient separation, yielding 2,2-diisopropylpropionitrile with a purity content reaching 98%. This high level of purity is critical for downstream applications in the flavor and fragrance industry, where trace impurities can significantly impact the sensory profile of the final cooling agent.
How to Synthesize 2,2-Diisopropylpropionitrile Efficiently
Implementing this synthesis route in a commercial setting requires careful attention to the sequential addition of reagents and temperature control to maximize yield and safety. The process begins with the formation of the sulfonate ester, where sulfonyl chloride is dissolved in an organic solvent and isopropanol is added dropwise to manage the exotherm. Following the removal of the solvent under reduced pressure, the crude ester is carried forward without further purification, demonstrating the robustness of the method against minor impurities. In the subsequent alkylation step, the propionitrile is mixed with the chosen strong base under an inert atmosphere to prevent moisture interference. The crude sulfonate ester is then introduced slowly to the reaction mixture, maintaining the temperature within the specified range to prevent thermal runaway while ensuring complete conversion. Detailed standardized operating procedures regarding specific flow rates, agitation speeds, and distillation cuts are essential for reproducibility.
- React isopropanol with sulfonyl chloride in an organic solvent like pyridine to form isopropyl sulfonate ester.
- Mix propionitrile with a strong base such as lithium amide or sodium ethoxide in a solvent like THF or ethanol.
- Add the isopropyl sulfonate crude product to the propionitrile mixture, maintain temperature between 0°C to 120°C, and isolate the product via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this sulfonate-based methodology offers profound advantages in terms of cost structure and supply chain resilience. The elimination of liquefied ammonia removes the need for specialized pressure vessels and cryogenic refrigeration units, which represents a significant reduction in capital expenditure for manufacturing facilities. Moreover, the raw materials involved, such as isopropanol and various sulfonyl chlorides, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague more exotic reagents. The simplified operational workflow translates directly into lower utility costs, as the energy demand for maintaining ultra-low temperatures is completely eradicated. This efficiency gain allows manufacturers to offer more competitive pricing structures without compromising on quality margins. Additionally, the shorter reaction cycles mean that production assets can be turned over more frequently, increasing overall plant capacity and allowing suppliers to respond more agilely to sudden spikes in demand from the flavor and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with cost-effective alternatives. By avoiding the use of lithium diisopropylamide or large quantities of liquefied ammonia, the direct material costs are significantly lowered. Furthermore, the ability to use crude sulfonate intermediates without intermediate purification steps reduces solvent consumption and waste disposal costs. The recovery and recycling of organic solvents like pyridine and ethyl acetate further enhance the economic viability of the process. These cumulative savings allow for a more favorable cost of goods sold, providing a buffer against market volatility and enabling long-term price stability for downstream customers seeking reliable flavor intermediate suppliers.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely available feedstocks that are produced by multiple vendors globally. Unlike specialized alkylating agents that may have limited suppliers, sulfonyl chlorides and isopropanol are standard industrial chemicals with robust logistics networks. The simplified processing requirements also mean that production is less susceptible to disruptions caused by equipment failure or maintenance issues associated with complex cryogenic systems. This reliability ensures consistent delivery schedules, which is paramount for clients managing just-in-time inventory systems. The reduced lead time for production batches means that safety stocks can be optimized, freeing up working capital for both the manufacturer and the purchaser while ensuring uninterrupted availability of high-purity intermediates.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the absence of extreme operating conditions. The reaction can be safely conducted in standard glass-lined or stainless-steel reactors, facilitating easy technology transfer between sites. From an environmental standpoint, the process generates less hazardous waste compared to methods involving heavy metals or volatile halogenated gases. The efficient solvent recovery systems minimize volatile organic compound emissions, aligning with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden and potential liability for manufacturers, making the supply chain more sustainable and resilient against future legislative changes regarding chemical safety and waste management practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific nitrile intermediate. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions about integrating this material into their broader supply chains. The answers reflect the consensus on best practices for handling, storage, and quality assurance associated with this synthesis route.
Q: Why is the sulfonate route superior to traditional alkyl halide methods for this intermediate?
A: The sulfonate route eliminates the need for hazardous liquefied ammonia and ultra-low temperature conditions required by traditional sodium amide methods, significantly simplifying industrial equipment requirements and improving safety profiles.
Q: What purity levels can be achieved with this novel synthesis method?
A: The patent data indicates that the final product can achieve a purity content of approximately 98% after vacuum distillation, with effective removal of inorganic impurities and solvent residues through standard extraction and washing protocols.
Q: Does this process require expensive transition metal catalysts?
A: No, the process utilizes common strong bases like lithium amide, sodium hydride, or metal alkoxides, avoiding the cost and contamination risks associated with precious transition metal catalysts often found in cross-coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Diisopropylpropionitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the global fine chemical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102093257B are fully realized in practical manufacturing scenarios. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,2-diisopropylpropionitrile meets the exacting standards required for WS-23 production. Our facility is equipped to handle the specific solvent systems and base reagents required for this sulfonate chemistry safely and efficiently. By leveraging our expertise in process optimization, we deliver a product that not only meets chemical specifications but also supports your broader goals of cost efficiency and supply security.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities align with your project timelines. Whether you are looking to secure a long-term supply agreement or need support for a new product development initiative, our commitment to quality and transparency makes us the ideal partner. Contact us today to discuss how our advanced production methods can enhance your supply chain resilience and drive value for your organization.
