Revolutionizing 2,2-Diisopropylpropionitrile Production: A Scalable Sulfonate Coupling Strategy for Global Supply Chains
Introduction: A Paradigm Shift in Nitrile Intermediate Synthesis
The global demand for high-purity cooling agents, specifically WS-23, has placed immense pressure on the supply chains of its key precursors, necessitating a re-evaluation of traditional manufacturing methodologies. Patent CN102093257A introduces a transformative synthetic route for 2,2-diisopropylpropionitrile, a critical intermediate, effectively addressing the longstanding bottlenecks of high raw material costs and hazardous operating conditions associated with legacy processes. This technology leverages a robust sulfonate coupling strategy that replaces the reliance on cryogenic liquid ammonia systems and scarce alkylating agents with readily available commodity chemicals like isopropanol and propionitrile. By shifting the reaction paradigm to a sulfonate-based nucleophilic substitution, the patent outlines a pathway that not only enhances safety profiles but also significantly streamlines the production cycle, offering a compelling value proposition for large-scale fine chemical manufacturers seeking reliable agrochemical intermediate supplier partnerships. The strategic implementation of this method allows for operation at ambient to moderately elevated temperatures, removing the energy-intensive burden of maintaining ultra-low reaction environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,2-diisopropylpropionitrile has been plagued by severe economic and operational inefficiencies, primarily stemming from two dominant prior art methods that are ill-suited for modern industrial scale-up. The first conventional approach involves the alkylation of 2,2-diisopropylacetonitrile using methyl bromide or methyl iodide in the presence of strong bases like lithium diisopropylamide; however, the starting nitrile itself is expensive, difficult to source, and complex to synthesize, creating a prohibitive cost barrier. Furthermore, the use of gaseous methyl bromide introduces significant handling complexities and safety hazards in a plant setting, while methyl iodide carries an exorbitant price tag that erodes profit margins. The second traditional method utilizes a condensation reaction between propionitrile and isopropyl bromide under sodium amide and liquid ammonia conditions, which mandates cryogenic temperatures to maintain the ammonia in a liquid state. This requirement for extreme refrigeration not only escalates energy consumption but also extends the production cycle to upwards of 30 hours, severely limiting throughput and creating a fragile supply chain vulnerable to equipment failure.
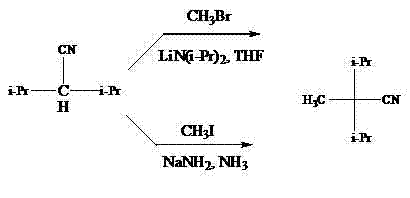
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel methodology disclosed in the patent employs a two-step sequence that begins with the conversion of isopropanol into an isopropyl sulfonate ester, followed by a coupling reaction with propionitrile. This innovative route circumvents the need for cryogenic ammonia entirely, allowing the reaction to proceed in common organic solvents such as tetrahydrofuran, toluene, or ethanol at temperatures ranging from 0°C to 120°C. By utilizing sulfonyl chlorides—such as methanesulfonyl chloride, benzenesulfonyl chloride, or trifluoromethanesulfonyl chloride—as activating agents, the process generates a highly reactive electrophile that couples efficiently with the propionitrile anion. This shift eliminates the dependency on gaseous alkylating agents and scarce precursors, replacing them with stable, easy-to-handle liquids that facilitate continuous processing and simplified logistics. The result is a streamlined workflow that drastically reduces the production cycle time and enhances the overall safety profile of the manufacturing facility, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
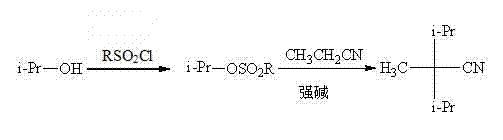
Mechanistic Insights into Sulfonate-Mediated Nucleophilic Substitution
The core chemical innovation of this process lies in the generation of a superior leaving group via sulfonate esterification, which facilitates a clean SN2-type nucleophilic substitution under relatively mild basic conditions. In the first stage, isopropanol reacts with a sulfonyl chloride in the presence of a base like pyridine to form an isopropyl sulfonate (e.g., isopropyl mesylate or tosylate); this transformation activates the secondary carbon of the isopropyl group, rendering it susceptible to nucleophilic attack despite the inherent steric hindrance. In the subsequent step, a strong base such as lithium amide, sodium hydride, or sodium ethoxide deprotonates the alpha-carbon of propionitrile to generate a resonance-stabilized carbanion. This nucleophile then attacks the electrophilic carbon of the isopropyl sulfonate, displacing the sulfonate anion—a much better leaving group than a halide in many contexts—to form the quaternary carbon center of 2,2-diisopropylpropionitrile. The choice of base and solvent is critical; for instance, using lithium amide in THF at 0°C ensures high selectivity and minimizes side reactions, while the use of alkoxides in ethanol or toluene at higher temperatures provides flexibility for different reactor configurations.
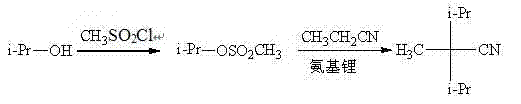
From an impurity control perspective, this mechanism offers distinct advantages over the chaotic environment of liquid ammonia reactions. The use of defined sulfonate esters prevents the formation of poly-alkylated byproducts that often plague direct alkylation methods, as the stoichiometry can be tightly controlled. Furthermore, the boiling point differences between the sulfonate intermediates, the solvents, and the final nitrile product allow for efficient separation via vacuum distillation, ensuring that inorganic salts and unreacted starting materials are effectively removed. The patent data indicates that collecting the fraction at 74-76°C under 2mmHg yields a product with 98% purity, demonstrating the robustness of the purification protocol. This high level of chemical fidelity is essential for downstream applications in the flavor and fragrance industry, where trace impurities can alter sensory profiles, thereby validating the process as a reliable source for high-purity OLED material precursors or similar high-value fine chemicals.
How to Synthesize 2,2-Diisopropylpropionitrile Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to a precise sequence of operations that maximizes yield while maintaining safety standards. The process begins with the preparation of the sulfonate ester, where strict temperature control during the addition of isopropanol to the sulfonyl chloride solution is vital to prevent exothermic runaway. Following the isolation of the crude sulfonate, which often requires no further purification, the focus shifts to the coupling reaction where the choice of base dictates the thermal profile; lithium amide allows for low-temperature operation, whereas metal alkoxides permit reflux conditions. This flexibility enables producers to adapt the protocol to their existing infrastructure, whether they possess cryogenic capabilities or prefer ambient pressure reactors, ensuring a versatile approach to commercial scale-up of complex pharmaceutical intermediates.
- React isopropanol with a sulfonyl chloride (e.g., methanesulfonyl chloride) in an organic solvent like pyridine to form the corresponding isopropyl sulfonate ester.
- Generate the propionitrile anion by treating propionitrile with a strong base such as lithium amide, sodium hydride, or metal alkoxides in solvents like THF or toluene.
- Couple the isopropyl sulfonate with the propionitrile anion at temperatures ranging from 0°C to 120°C, followed by aqueous workup and vacuum distillation to isolate the pure nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sulfonate-based methodology represents a strategic opportunity to de-risk the supply of critical cooling agent intermediates while achieving substantial cost savings. The primary economic driver is the replacement of exotic and expensive starting materials with commodity chemicals; isopropanol and propionitrile are produced on a massive global scale, ensuring price stability and consistent availability compared to the volatile market for 2,2-diisopropylacetonitrile or methyl iodide. Moreover, the elimination of liquid ammonia removes the need for specialized pressure vessels and extensive refrigeration units, leading to a drastic simplification of the capital equipment required for production. This reduction in infrastructure complexity translates directly into lower fixed costs and a more agile manufacturing footprint that can be rapidly deployed or scaled according to market demand without the long lead times associated with installing cryogenic plants.
- Cost Reduction in Manufacturing: The shift to sulfonate chemistry fundamentally alters the cost structure by removing the most expensive line items from the bill of materials. By avoiding the use of gaseous methyl bromide and scarce lithium bases, the variable cost per kilogram of the final product is significantly lowered, allowing for more competitive pricing in the global marketplace. Additionally, the ability to recycle solvents like pyridine and ethyl acetate further enhances the economic efficiency of the process, reducing waste disposal costs and raw material consumption. This leaner operational model ensures that manufacturers can maintain healthy margins even during periods of raw material price fluctuation, providing a buffer against market volatility.
- Enhanced Supply Chain Reliability: Relying on widely available feedstocks like isopropanol mitigates the risk of supply disruptions that often occur with niche reagents. The simplified reaction conditions also mean that production is less susceptible to delays caused by equipment maintenance or utility failures, as the process does not depend on the continuous operation of energy-intensive cooling systems. This reliability is crucial for meeting the just-in-time delivery requirements of multinational clients, ensuring that reducing lead time for high-purity specialty chemicals becomes a tangible reality rather than just a goal. The robustness of the process allows for consistent batch-to-batch quality, fostering long-term trust with downstream customers.
- Scalability and Environmental Compliance: The absence of cryogenic ammonia and gaseous alkylating agents significantly improves the environmental and safety profile of the facility, easing the regulatory burden associated with hazardous material handling. The process generates less hazardous waste and operates at safer pressures, facilitating easier permitting and expansion of production capacity. This alignment with green chemistry principles not only reduces compliance costs but also enhances the brand reputation of the manufacturer as a sustainable partner. The straightforward workup and distillation steps are easily adaptable to large-scale reactors, ensuring that the transition from pilot plant to full commercial production is smooth and predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, derived directly from the detailed specifications and experimental data provided in the patent documentation. These insights are intended to clarify the operational benefits and chemical rationale behind the sulfonate coupling strategy for stakeholders evaluating this technology for potential licensing or procurement. Understanding these nuances is essential for making informed decisions about integrating this process into existing manufacturing portfolios.
Q: Why is the sulfonate route superior to the traditional liquid ammonia method for 2,2-diisopropylpropionitrile?
A: The traditional method relies on liquid ammonia and sodium amide at cryogenic temperatures, requiring specialized refrigeration and posing significant safety risks. The sulfonate route operates at much milder temperatures (0°C to 120°C) using common organic solvents, drastically simplifying equipment requirements and improving operational safety.
Q: What are the key cost drivers reduced by this new manufacturing process?
A: The primary cost reduction comes from replacing expensive and scarce starting materials like 2,2-diisopropylacetonitrile or gaseous methyl bromide with commodity chemicals like isopropanol and propionitrile. Additionally, the elimination of cryogenic cooling systems significantly lowers energy consumption and capital expenditure.
Q: Can this process be scaled for industrial production of WS-23 intermediates?
A: Yes, the process is specifically designed for industrial suitability. It avoids the handling of gaseous reagents and ultra-low temperature conditions, utilizes recyclable solvents, and achieves high purity (98%) through standard distillation, making it highly scalable for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Diisopropylpropionitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value intermediates like 2,2-diisopropylpropionitrile, and we are uniquely positioned to leverage this patented technology for our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for pharmaceutical and flavor applications. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal profiles required by this sulfonate method, delivering consistent quality and reliability.
We invite forward-thinking partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this advanced process. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can drive value and efficiency in your production of WS-23 and related cooling agents.
