Optimizing Indiplon Manufacturing: A Safer, Cost-Effective Synthetic Route for Global Supply Chains
Optimizing Indiplon Manufacturing: A Safer, Cost-Effective Synthetic Route for Global Supply Chains
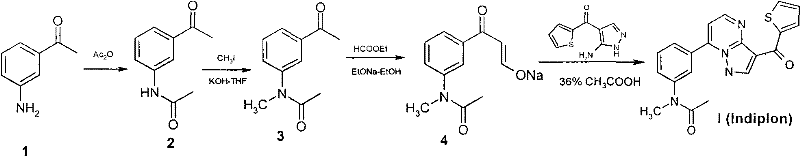
The pharmaceutical industry is constantly seeking robust manufacturing processes that balance high purity with operational safety and cost efficiency. Patent CN101591338B introduces a transformative synthetic methodology for Indiplon, a potent non-benzodiazepine hypnotic agent targeting GABA-A receptors. This innovation addresses critical bottlenecks in the legacy production of this complex pyrazolopyrimidine derivative by fundamentally altering the reagent profile and reaction media. By shifting from hazardous strong bases and expensive enamines to stable alkali metal hydroxides and aqueous condensation systems, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to secure long-term contracts with major drug developers. The technical breakthrough lies not just in the chemical transformation but in the holistic redesign of the process flow to minimize environmental impact and maximize throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Indiplon and its analogues has relied heavily on methodologies described in earlier patents such as US4626538 and US4900836, which present significant challenges for modern commercial scale-up of complex pharmaceutical intermediates. The traditional route typically employs sodium hydride (NaH) for the critical N-methylation step, a reagent known for its pyrophoric nature and the generation of flammable hydrogen gas, posing severe safety risks in large-scale reactors. Furthermore, the formation of the enamine intermediate traditionally utilizes N,N-dimethylformamide dimethyl acetal (DMF-DMA), a costly specialty reagent that complicates waste stream management due to its toxicity and potential for generating carcinogenic byproducts. These legacy processes often require stringent anhydrous conditions and complex workup procedures involving organic solvents, leading to elevated operational expenditures and extended production cycles that strain supply chain continuity.
The Novel Approach
The methodology disclosed in CN101591338B represents a paradigm shift by substituting these hazardous and expensive components with benign, commodity-grade alternatives. Instead of sodium hydride, the process utilizes powdered potassium hydroxide (KOH) for the methylation of 3-acetamidoacetophenone, eliminating the risk of hydrogen evolution and allowing for safer handling in standard stainless steel equipment. Additionally, the expensive DMF-DMA is replaced by ethyl formate in the presence of sodium ethoxide to generate the key beta-keto enolate intermediate, drastically reducing raw material costs. Perhaps most significantly, the final condensation step is conducted in an aqueous medium rather than organic solvents, leveraging the poor solubility of the product to drive the reaction equilibrium forward and simplify isolation through simple filtration. This approach not only enhances cost reduction in API manufacturing but also aligns with green chemistry principles by minimizing volatile organic compound (VOC) emissions.
Mechanistic Insights into KOH-Mediated Methylation and Aqueous Condensation
The core chemical innovation of this process revolves around the efficient N-methylation of the acetamide nitrogen and the subsequent construction of the pyrazolopyrimidine core. In the methylation step, powdered potassium hydroxide acts as a heterogeneous base to deprotonate the amide nitrogen of 3-acetamidoacetophenone, generating a nucleophilic amidate anion in situ. This anion then undergoes an SN2 reaction with methyl iodide to form N-methyl-N-(3-acetylphenyl)acetamide. The use of KOH is mechanistically superior in an industrial context because it avoids the passivation issues often seen with sodium hydride and provides a more controlled reaction profile, reducing the formation of O-methylated impurities that can be difficult to separate. The resulting N-methylated ketone is then subjected to a Claisen-type condensation with ethyl formate, where sodium ethoxide facilitates the formation of the resonance-stabilized enolate anion, a crucial precursor for the ring-closing step.
The final cyclization mechanism involves the condensation of this lipophilic enolate with (3-amino-1H-pyrazol-4-yl)-2-thiophenemethanone under acidic aqueous conditions. The introduction of water as the reaction medium serves a dual purpose: it acts as a heat sink to manage the exothermicity of the acid-base neutralization and functions as an anti-solvent. As the Indiplon molecule forms, its low solubility in the acidic aqueous phase causes it to precipitate out of the solution immediately. This precipitation effectively removes the product from the equilibrium, driving the reaction to completion according to Le Chatelier's principle and preventing retro-aldol type decomposition. This mechanism ensures high-purity Indiplon with minimal side products, as the harsh conditions that typically degrade sensitive heterocyclic structures in organic solvents are mitigated by the buffering capacity of the aqueous system.
How to Synthesize Indiplon Efficiently
The synthesis of Indiplon via this optimized route involves a logical sequence of four distinct chemical transformations that can be executed with standard processing equipment. The process begins with the protection of the amine functionality followed by methylation, enolate formation, and finally, heterocyclic ring closure. Each step has been engineered to maximize yield while minimizing the need for chromatographic purification, relying instead on crystallization and filtration. For R&D teams looking to implement this technology, the detailed standardized operating procedures regarding stoichiometry, temperature profiles, and addition rates are critical for reproducibility. The following guide outlines the strategic workflow derived from the patent claims to ensure successful technology transfer.
- Acetylate 3-aminoacetophenone with acetic anhydride to form 3-acetamidoacetophenone.
- Methylate the acetamide using powdered potassium hydroxide and methyl iodide in THF or toluene.
- Perform Claisen condensation with ethyl formate and sodium ethoxide to generate the key enolate intermediate.
- Condense the enolate with (3-amino-1H-pyrazol-4-yl)-2-thiophenemethanone in water/acetic acid to yield Indiplon.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple unit cost savings. By eliminating the dependency on sodium hydride, facilities can reduce their insurance premiums and safety compliance overheads associated with handling pyrophoric materials. The substitution of DMF-DMA with ethyl formate represents a shift from a specialty chemical with volatile pricing to a bulk commodity, stabilizing the cost of goods sold (COGS) against market fluctuations. Furthermore, the ability to perform the final reaction in water significantly reduces the volume of organic waste solvent requiring disposal, lowering environmental compliance costs and accelerating batch turnover times. These factors collectively enhance the resilience of the supply chain against regulatory changes and raw material shortages.
- Cost Reduction in Manufacturing: The replacement of expensive reagents like DMF-DMA and sodium hydride with commodity chemicals such as ethyl formate and potassium hydroxide results in a substantial decrease in direct material costs. Additionally, the simplified workup procedures, which rely on filtration rather than complex extraction and distillation sequences, reduce energy consumption and labor hours per batch. The elimination of hazardous waste streams further lowers the total cost of ownership by minimizing disposal fees and environmental levies, creating a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing stability is improved as the key reagents (KOH, ethyl formate, methyl iodide) are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the aqueous condensation step ensures consistent batch-to-batch quality, reducing the rate of failed batches that can disrupt delivery schedules. This reliability allows for more accurate demand forecasting and inventory planning, ensuring that downstream API manufacturers receive their intermediates on time without unexpected delays caused by process upsets or reagent shortages.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of non-hazardous bases and aqueous media, which simplifies heat transfer and mixing dynamics in large reactors. The reduction in VOC emissions aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing site against tighter emission caps. The solid product isolation method facilitates easy drying and packaging, streamlining the logistics of moving large quantities of material. This scalability ensures that the supply can grow in tandem with clinical demand without requiring disproportionate capital investment in specialized safety infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Indiplon synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide clarity on feasibility and performance. Understanding these nuances is essential for technical teams evaluating the fit of this process within their existing manufacturing portfolios. The responses highlight the specific advantages in safety, purity, and operational simplicity that distinguish this method from prior art.
Q: How does this new Indiplon synthesis improve safety compared to traditional methods?
A: The process replaces sodium hydride, which releases hazardous hydrogen gas, with stable powdered potassium hydroxide, significantly reducing explosion risks during industrial scale-up.
Q: What are the cost advantages of using ethyl formate over DMF-DMA?
A: Ethyl formate is a commodity chemical with significantly lower procurement costs compared to the specialized and expensive dimethylformamide dimethyl acetal (DMF-DMA) used in legacy routes.
Q: Does the aqueous workup affect the purity of the final Indiplon API intermediate?
A: No, the use of water as a solvent for the final condensation facilitates the precipitation of the product, simplifying filtration and washing while maintaining high purity specifications suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indiplon Supplier
The synthetic route detailed in CN101591338B exemplifies the kind of process innovation that drives value in the modern pharmaceutical supply chain. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such patented methodologies into commercial reality, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the necessary corrosion-resistant reactors and filtration systems to handle the specific requirements of this aqueous-organic hybrid process, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to continuous process improvement, we are positioned to be your strategic partner in securing a stable supply of high-quality Indiplon intermediates.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project timelines and budget constraints. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this greener, safer route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your supply chain is built on a foundation of scientific excellence and operational reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
