Advanced Manufacturing Strategy for Indiplon: A Cost-Effective and Scalable Synthetic Route
The pharmaceutical landscape for treating insomnia has shifted significantly towards non-benzodiazepine agents that offer improved safety profiles and reduced side effects. Among these, Indiplon stands out as a potent pyrazolopyrimidine derivative with selective binding affinity for GABA-A receptors. However, the commercial viability of such advanced therapeutics often hinges on the efficiency and cost-effectiveness of their synthetic routes. Patent CN101870700B introduces a transformative methodology for synthesizing Indiplon, addressing critical bottlenecks in traditional manufacturing processes. This innovation focuses on optimizing the construction of the key heterocyclic scaffold by substituting high-cost reagents with economically viable alternatives while maintaining high purity standards. For stakeholders in the fine chemical sector, understanding this technological leap is essential for securing a competitive edge in the supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
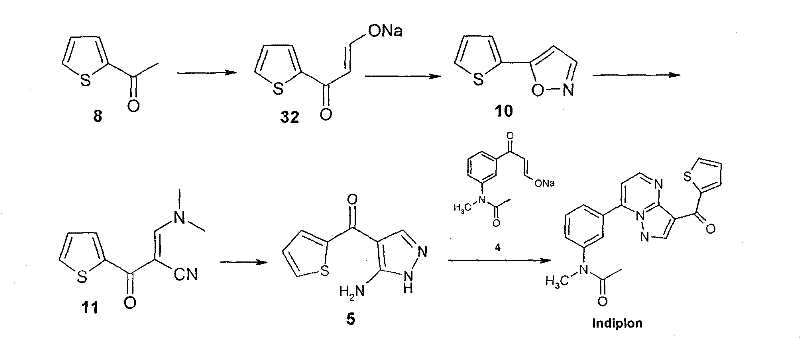
Historically, the synthesis of Indiplon and its precursors has been plagued by reliance on expensive and hazardous reagents that complicate scale-up efforts. Traditional pathways frequently utilize N,N-dimethylformamide dimethyl acetal (DMF-DMA) for enamine formation, a reagent that imposes a substantial financial burden on the overall cost of goods. Furthermore, methylation steps often employ sodium hydride as a base, which presents significant safety risks due to the evolution of hydrogen gas, requiring specialized equipment and rigorous safety protocols. The final condensation reactions typically occur in glacial acetic acid, necessitating complex neutralization steps with sodium bicarbonate that generate excessive carbon dioxide gas, creating operational hazards and complicating waste management. These factors collectively result in a process that is not only costly but also environmentally taxing and difficult to manage on a multi-ton scale.
The Novel Approach
The methodology disclosed in the patent offers a strategic departure from these legacy constraints by re-engineering the synthetic sequence for maximum efficiency. A pivotal improvement involves the replacement of expensive DMF-DMA with ethyl formate during the initial formation of the beta-keto enolate from acetylthiophene. This substitution drastically lowers raw material costs without compromising reaction yields. Additionally, the process utilizes alkali metal alkoxides or powdered potassium hydroxide instead of sodium hydride, effectively mitigating the safety risks associated with hydrogen gas release. The final coupling step is conducted in an aqueous medium with controlled acidity, allowing the product to precipitate directly from the reaction mixture. This simplifies isolation to mere filtration and washing, eliminating the need for extensive solvent extraction and reducing the environmental footprint associated with organic waste disposal.
Mechanistic Insights into Enolate-Mediated Heterocycle Formation
The core of this synthetic advancement lies in the precise manipulation of enolate chemistry to construct the pyrazolopyrimidine core. The process initiates with a Claisen-type condensation where acetylthiophene reacts with ethyl formate in the presence of a base to form a stable alkali metal beta-ketothienylpropionaldehyde enolate. This intermediate is crucial as it serves as the nucleophilic partner for subsequent cyclization. Unlike traditional enamines which can be unstable or difficult to purify, this enolate salt is water-soluble, stable against moisture absorption, and easy to store, providing a robust platform for downstream processing. The subsequent reaction with hydroxylamine hydrochloride under acidic conditions facilitates the formation of the isoxazole ring, a key structural motif that directs the regioselectivity of the final pyrazole fusion. This mechanistic pathway ensures high fidelity in bond formation, minimizing the generation of regioisomeric impurities that are common in less controlled heterocyclic syntheses.
Impurity control is further enhanced by the specific choice of reaction media and workup procedures. In the final condensation step, the use of water as a solvent combined with acetic acid creates a unique solubility profile where the target Indiplon molecule precipitates out of solution as it forms. This phenomenon, known as reactive crystallization, drives the equilibrium towards product formation according to Le Chatelier's principle, thereby maximizing conversion rates. Moreover, the direct precipitation allows for the exclusion of soluble byproducts and unreacted starting materials, which remain in the aqueous phase. This intrinsic purification mechanism reduces the reliance on chromatographic separation or multiple recrystallization steps, ensuring that the final API intermediate meets stringent purity specifications required for pharmaceutical applications while streamlining the manufacturing workflow.
How to Synthesize Indiplon Efficiently
Implementing this optimized synthetic route requires careful attention to reaction parameters such as temperature control and stoichiometric ratios to ensure reproducibility and high yield. The process is divided into distinct stages, beginning with the preparation of the thiophene-based enolate and concluding with the final condensation to form the pyrazolopyrimidine skeleton. Each step has been refined to operate under mild conditions, typically ranging from 0°C to 100°C, which reduces energy consumption and equipment stress. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for technical teams aiming to adopt this cost-effective manufacturing strategy.
- React acetylthiophene with alkali metal alkoxide and ethyl formate to form alkali metal beta-ketothienylpropionaldehyde enolate.
- Convert the enolate to 5-(2-thiophene)isoxazole using hydroxylamine hydrochloride under acidic conditions.
- React the isoxazole with DMF-DMA to generate alpha-[(dimethylamino)methylene]-beta-keto-2-thiophene acrylonitrile.
- Cyclize the acrylonitrile with aminoguanidine sulfate to obtain the key intermediate (3-amino-1H-pyrazol-4-yl)-2-thiophenemethanone.
- Condense the aminopyrazole intermediate with the phenyl enolate salt in an aqueous acidic medium to finalize Indiplon.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple unit cost reductions. By fundamentally altering the reagent profile, the process eliminates dependency on volatile and expensive specialty chemicals, thereby stabilizing the supply chain against market fluctuations. The shift towards safer reagents like ethyl formate and potassium hydroxide reduces the regulatory burden and insurance costs associated with handling hazardous materials like sodium hydride. Furthermore, the simplification of post-treatment procedures means faster batch turnover times and reduced labor requirements, contributing to overall operational efficiency. These factors collectively enhance the reliability of supply for high-purity pharmaceutical intermediates, ensuring consistent delivery schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The substitution of high-cost reagents such as DMF-DMA with commodity chemicals like ethyl formate results in a significant decrease in raw material expenditure. Additionally, the elimination of sodium hydride removes the need for expensive safety infrastructure and hazardous waste disposal protocols. The streamlined workup process, which relies on filtration rather than complex extractions, further reduces solvent consumption and utility costs, leading to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this route, including acetylthiophene and ethyl formate, are widely available commodity chemicals with stable global supply chains. This reduces the risk of production delays caused by shortages of specialized reagents. The stability of the intermediate enolate salts also allows for potential stockpiling or batch scheduling flexibility, ensuring continuous production capabilities even during periods of high demand or logistical disruptions in the supply of complex fine chemicals.
- Scalability and Environmental Compliance: The use of water as a primary solvent in the final condensation step aligns with green chemistry principles by minimizing the generation of organic volatile compounds (VOCs). The process avoids the massive CO2 evolution associated with bicarbonate neutralization in acetic acid, simplifying venting requirements and improving workplace safety. These environmental advantages facilitate easier regulatory approval for new manufacturing sites and support corporate sustainability goals, making the process highly scalable for commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Indiplon synthesis method. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on process feasibility and performance metrics. Understanding these details is crucial for technical decision-makers evaluating the transition from legacy processes to this optimized pathway.
Q: How does this new synthesis route reduce manufacturing costs for Indiplon?
A: The process replaces expensive reagents like N,N-dimethylformamide dimethyl acetal (DMF-DMA) with significantly cheaper ethyl formate for the initial enolate formation. Additionally, it substitutes hazardous sodium hydride with powdered potassium hydroxide or alkoxides, eliminating safety costs associated with hydrogen gas management.
Q: What are the environmental benefits of this Indiplon production method?
A: The final condensation step utilizes water as the primary solvent rather than large volumes of organic solvents like glacial acetic acid. This minimizes organic waste generation and simplifies the neutralization process, which traditionally produces large amounts of carbon dioxide gas, thereby enhancing the overall environmental profile of the manufacturing process.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It features mild reaction conditions ranging from 0°C to 100°C, simple post-treatment procedures involving filtration and washing, and the use of stable, easy-to-store intermediates, making it highly robust for commercial scale-up operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indiplon Supplier
The technical innovations described in patent CN101870700B represent a significant leap forward in the manufacturing of CNS-active pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory-scale breakthroughs into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this cost-effective route are fully realized at an industrial level. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Indiplon intermediate meets the exacting standards required for global pharmaceutical registration and patient safety.
We invite procurement leaders and R&D directors to explore how our optimized manufacturing capabilities can enhance your supply chain resilience. By leveraging this advanced synthetic route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to process innovation can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
