Optimizing Iopromide Production: A Novel Synthetic Route for High-Purity Contrast Agents
Optimizing Iopromide Production: A Novel Synthetic Route for High-Purity Contrast Agents
The pharmaceutical industry continuously seeks robust manufacturing processes for critical diagnostic agents, and the synthesis of Iopromide, a non-ionic iodinated contrast medium, represents a significant area of chemical engineering focus. Patent CN102964269A introduces a transformative preparation method that addresses longstanding purity and yield challenges associated with traditional synthetic pathways. By leveraging 5-amino-3-(2,3-dihydroxypropylamino carbonyl)-2,4,6-triiodoisophthalic acid (Formula II) or its methoxyacetamido derivative (Formula II-1) as key starting materials, this innovation establishes a streamlined route that bypasses the formation of persistent impurities. The core of this technological breakthrough lies in a strategic sequence of protection, chlorination, amidation, and hydrolysis, which collectively ensure the production of high-purity intermediates and final active pharmaceutical ingredients. For global procurement teams and R&D directors, understanding this shift from complex resin-based purifications to efficient crystallization-driven processes is essential for securing reliable supply chains and optimizing cost structures in contrast media manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Iopromide, such as those disclosed in US Patent No. 4364921, have been plagued by significant chemoselectivity issues that compromise both yield and product quality. Specifically, the direct reaction of triiodoisophthalic acid dichloride with amines often results in the formation of bis-amide byproducts, commonly referred to as 'bismers,' due to the nucleophilic attack of excess amine reagents on the activated acid chloride species. These structural analogs are notoriously difficult to separate from the target molecule, often requiring extensive and costly purification steps. Furthermore, alternative methods developed to mitigate this issue, such as Method 2 and Method 3 in the prior art, introduced their own set of operational inefficiencies by relying heavily on resin column chromatography. While these resin-based approaches successfully avoided bismer formation, they imposed severe limitations on scalability, necessitating large solvent volumes and complex post-treatment procedures that are ill-suited for multi-ton commercial production. The reliance on such labor-intensive purification techniques inevitably drives up manufacturing costs and extends lead times, creating vulnerabilities in the supply chain for this critical diagnostic agent.
The Novel Approach
The methodology outlined in CN102964269A fundamentally reengineers the synthesis trajectory by introducing a protective group strategy that preemptively blocks side reactions. Instead of struggling to remove impurities after they form, this novel approach utilizes highly purified starting materials where the reactive amino and hydroxyl functionalities are temporarily masked with methoxyacetyl groups. This protection step generates a stable intermediate (Formula III) that can be readily purified via simple recrystallization, ensuring a high degree of homogeneity before the molecule undergoes further transformation. Subsequent conversion to the acyl chloride (Formula IV) and reaction with methylaminopropanediol proceeds with high specificity, effectively eliminating the pathway for bismer generation. The final hydrolysis step cleanly removes the protecting groups to yield Iopromide with exceptional purity profiles. This shift from remedial purification to preventative synthesis design represents a paradigm shift in process chemistry, offering a route that is not only chemically superior but also inherently more scalable and economically viable for industrial applications.
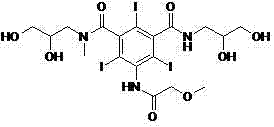
Mechanistic Insights into Protective Group Strategy and Amidation
The chemical elegance of this new process is rooted in the precise manipulation of functional group reactivity through the use of methoxyacetyl protecting groups. In the initial stage, the reaction of the starting triiodoisophthalic acid derivative with methoxyacetic anhydride or methoxyacetyl chloride selectively acylates the primary amine and the vicinal diol moieties. This dual protection serves a dual purpose: it enhances the solubility and crystallinity of the intermediate, facilitating easy purification, and it sterically hinders the hydroxyl groups from participating in unwanted esterification or etherification side reactions during the subsequent activation of the carboxylic acid. When the protected acid is treated with a chlorinating agent such as thionyl chloride, the resulting acyl chloride is stabilized against intramolecular cyclization or degradation, a common pitfall in polyfunctional iodinated aromatics. The mechanistic control extends to the amidation step, where the protected acyl chloride reacts with methylaminopropanediol. Because the competing nucleophilic sites on the aromatic core are blocked, the incoming amine attacks exclusively at the activated carbonyl carbon, ensuring the formation of the desired diamide structure with high regioselectivity. This level of control is critical for maintaining the integrity of the iodine substituents, which are susceptible to dehalogenation under harsh conditions.
Furthermore, the impurity control mechanism is intrinsically linked to the quality of the starting materials and the efficiency of the protection step. By employing starting materials like Formula II, which can be obtained in high purity through recrystallization, the process minimizes the introduction of foreign contaminants at the very beginning of the synthesis. The patent data highlights that the intermediate Formula III can be isolated with HPLC purity exceeding 98%, creating a 'clean' foundation for the remainder of the synthesis. This contrasts sharply with older methods where impurities generated in early steps would propagate through the entire sequence, accumulating in the final product and necessitating aggressive downstream processing. The final hydrolysis using sodium hydroxide is designed to be orthogonal, cleaving the methoxyacetyl esters and amides without affecting the stable aromatic amide bonds or the carbon-iodine bonds. This selective deprotection ensures that the final Iopromide molecule retains its structural fidelity while shedding the temporary masking groups, resulting in a product that meets stringent pharmacopeial standards for color, clarity, and related substances without the need for complex chromatographic separation.
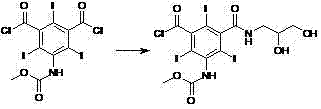
How to Synthesize Iopromide Efficiently
The synthesis of Iopromide via this novel pathway involves a logical progression of four distinct chemical transformations that prioritize yield and purity at every stage. The process begins with the protection of the multifunctional starting material, followed by activation of the carboxylic acid, coupling with the side-chain amine, and final deprotection. Each step has been optimized to utilize standard reagents and manageable reaction conditions, making the technology accessible for implementation in existing fine chemical facilities. The detailed operational parameters, including specific molar ratios, temperature profiles, and workup procedures, are critical for replicating the high success rates reported in the patent examples. For process chemists looking to implement this route, attention to the crystallization conditions of the protected intermediates is paramount, as this serves as the primary purification checkpoint. The following guide outlines the standardized workflow derived from the patent embodiments, providing a clear roadmap for laboratory and pilot-scale execution.
- Protect the amino and hydroxyl groups of the starting triiodoisophthalic acid derivative using methoxyacetyl groups to form the protected intermediate (Formula III).
- Convert the carboxylic acid group of the protected intermediate into an acyl chloride (Formula IV) using a chlorinating agent like thionyl chloride.
- React the acyl chloride with methylaminopropanediol to form the diamide precursor (Formula V) under controlled temperature conditions.
- Perform alkaline hydrolysis using sodium hydroxide to remove protecting groups and purify the final Iopromide product via ion exchange and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced supply reliability. By eliminating the need for resin column chromatography—a process that is notoriously solvent-intensive and difficult to scale—the new method significantly lowers the environmental footprint and waste disposal costs associated with production. The ability to rely on crystallization for purification means that the process can be scaled from kilogram to multi-ton quantities with minimal re-engineering, ensuring that supply can flexibly meet market demand fluctuations. Moreover, the higher yields reported in the patent embodiments imply a more efficient utilization of raw materials, particularly the expensive iodinated precursors, leading to substantial cost savings in the cost of goods sold (COGS). This efficiency creates a buffer against raw material price volatility, allowing for more stable pricing models for downstream pharmaceutical customers.
- Cost Reduction in Manufacturing: The elimination of resin-based purification steps removes a major cost center from the production budget. Resin columns require significant capital investment for equipment, regular replacement of consumable resins, and large volumes of organic solvents for elution and regeneration. By replacing this with crystallization and filtration, the process drastically reduces solvent consumption and waste treatment costs. Additionally, the improved yield at each step means that less starting material is required to produce a unit of final product, optimizing the consumption of high-value iodinated intermediates. This cumulative effect results in a leaner manufacturing process with a lower break-even point, enabling competitive pricing strategies in the global contrast media market.
- Enhanced Supply Chain Reliability: The robustness of the new synthetic route contributes significantly to supply chain resilience. Traditional methods prone to variable yields and difficult purifications often result in batch failures or extended cycle times, creating bottlenecks that disrupt delivery schedules. In contrast, the predictable nature of the protection-chlorination-amidation sequence ensures consistent batch-to-batch quality and turnaround times. The use of stable, isolable intermediates allows for the strategic stocking of key precursors, decoupling the production of the final API from the immediate availability of all raw materials. This flexibility is crucial for maintaining continuity of supply in the face of logistical disruptions or raw material shortages, ensuring that pharmaceutical partners can rely on a steady flow of high-quality Iopromide intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the new process aligns perfectly with modern green chemistry principles. The reduction in solvent usage and the avoidance of hazardous resin waste streams simplify the permitting process and reduce the regulatory burden on manufacturing sites. The process is inherently safer and more controllable, with exothermic reactions managed through controlled addition and temperature regulation. This ease of control facilitates seamless scale-up from pilot plants to commercial reactors, minimizing the risk of thermal runaways or safety incidents. For multinational corporations with strict ESG (Environmental, Social, and Governance) mandates, sourcing from a supplier utilizing this cleaner, more efficient technology demonstrates a commitment to sustainable manufacturing practices and responsible chemical stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel Iopromide synthesis method. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, providing clarity on how this technology outperforms legacy processes. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value of this supply partnership.
Q: How does this new method address the bis-amide impurity problem found in older patents?
A: Unlike conventional methods that generate bis-amide byproducts (bismers) due to excessive amine usage, this novel route utilizes highly purified starting materials (Formula II or II-1) and employs a strategic protection step. By protecting the hydroxyl and amino groups prior to the critical amidation step, the formation of these difficult-to-remove impurities is effectively suppressed at the source.
Q: What are the scalability advantages of avoiding resin column chromatography?
A: Previous synthetic routes often relied on resin column processing for purification, which presents significant bottlenecks for industrial scale-up due to solvent volume requirements and column capacity limits. The new method replaces these complex purification steps with straightforward crystallization and hydrolysis protocols, significantly enhancing throughput and reducing operational complexity for commercial manufacturing.
Q: What purity levels can be achieved with this preparation method?
A: The patent data indicates that by utilizing the specific protection and hydrolysis sequence described, the final Iopromide product can achieve HPLC purity levels exceeding 99%. This high purity is critical for contrast media applications to ensure patient safety and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iopromide Supplier
The technological advancements detailed in patent CN102964269A underscore the potential for producing Iopromide with unprecedented efficiency and purity, setting a new benchmark for the industry. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in large-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Iopromide intermediate meets the highest global pharmacopeial standards. Our commitment to quality assurance ensures that the complex challenges of iodinated contrast media synthesis are managed with precision, delivering products that support the safety and efficacy of diagnostic imaging procedures worldwide.
We invite pharmaceutical manufacturers and contract research organizations to collaborate with us to leverage this advanced synthetic technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and samples for your validation processes. Together, we can optimize your production of high-purity Iopromide, reducing lead times and securing a competitive advantage in the dynamic market for radiopharmaceutical intermediates.
