Scalable Chiral Resolution of NLG919 Precursors for Commercial Pharmaceutical Manufacturing
Introduction to Advanced Chiral Resolution Technologies
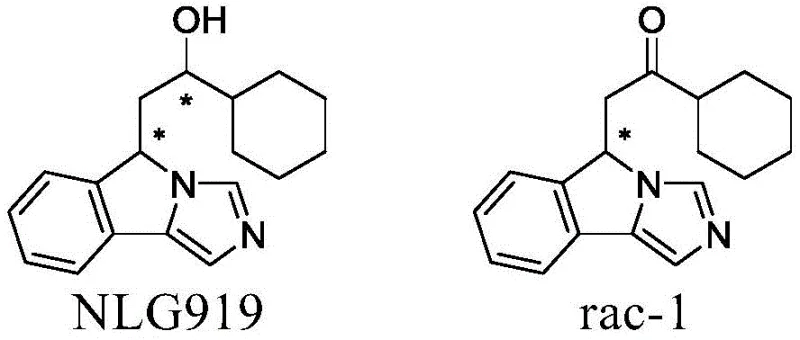
The pharmaceutical industry's relentless pursuit of potent oncology therapeutics has placed Indoleamine-2,3-dioxygenase 1 (IDO1) inhibitors at the forefront of immunotherapy research. Specifically, the compound NLG919 has demonstrated significant potential in reversing IDO1-mediated immune tolerance, thereby enhancing the immune system's ability to recognize and attack tumor cells. However, the synthesis of NLG919 presents a formidable stereochemical challenge due to the presence of two chiral centers, resulting in four possible stereoisomers with distinct pharmacological profiles. Addressing this complexity, Chinese Patent CN108239091B, published in August 2021, introduces a groundbreaking chemical resolution methodology for the key synthetic precursor, rac-1 (1-cyclohexyl-2-(5H-imidazo[5,1-a]isoindole)ethyl-1-one). This patent delineates a robust process utilizing chiral tartaric acid derivatives to efficiently separate enantiomers, offering a viable pathway for producing high-purity intermediates essential for next-generation cancer treatments.
The significance of this technological advancement cannot be overstated for stakeholders involved in the commercial scale-up of complex pharmaceutical intermediates. Traditional approaches to obtaining single enantiomers often rely on asymmetric synthesis or preparative chiral chromatography, both of which carry substantial economic and operational burdens. The method described in CN108239091B circumvents these hurdles by leveraging classical diastereomeric salt formation, a technique renowned for its scalability and cost-effectiveness in industrial settings. By providing a clear route to isolate both (R)-1 and (S)-1 configurations with high optical purity, this innovation empowers manufacturers to supply the precise stereoisomers required for rigorous Structure-Activity Relationship (SAR) studies and clinical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advent of this resolution technology, the production of NLG919 stereoisomers was fraught with inefficiencies that hindered large-scale adoption. Existing literature, such as Patent WO 2012/142237, describes an asymmetric synthesis route involving seven distinct reaction steps. This multi-step sequence not only consumes excessive time but also necessitates harsh reaction conditions that can compromise yield and safety. Furthermore, the reliance on specialized chiral catalysts drives up raw material costs significantly, making the process economically unviable for bulk manufacturing. Alternatively, some protocols employ chiral chromatography to separate the isomers. While effective on a laboratory scale, chiral chromatography is notoriously expensive due to the high cost of chiral stationary phases and the limited loading capacity of the columns. These factors collectively result in low total yields and prohibitive production costs, creating a bottleneck for the reliable NLG919 intermediate supplier seeking to meet global demand.
The Novel Approach
In stark contrast to these cumbersome methodologies, the novel approach detailed in the patent utilizes a direct chemical resolution strategy that dramatically simplifies the workflow. By reacting the racemic precursor rac-1 with specific chiral resolving agents, such as L-di-p-methylbenzoyl tartaric acid or its D-enantiomer, the process forms diastereomeric salts that exhibit distinct solubility properties. This fundamental physicochemical difference allows for the selective crystallization of one enantiomer over the other from a mixed solvent system. The elegance of this method lies in its operational simplicity; it avoids the need for exotic catalysts or complex equipment. Moreover, the resolving agent can be recovered from the mother liquor or aqueous layers through simple acidification and extraction, facilitating a circular economy within the manufacturing process. This shift from synthesis-based chirality introduction to separation-based resolution represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, offering a streamlined path to high-value chiral building blocks.
Mechanistic Insights into Diastereomeric Salt Formation
The core of this technology rests on the principles of stereochemistry and solubility equilibria. The precursor molecule, rac-1, contains a basic nitrogen atom within its imidazoisoindole ring system, which serves as the anchor point for interaction with the acidic resolving agent. When rac-1 is mixed with a chiral acid like D-di-p-methylbenzoyl tartaric acid in a suitable solvent mixture, an acid-base reaction occurs, generating two diastereomeric salts: one comprising the (R)-enantiomer of the base and the chiral acid, and the other comprising the (S)-enantiomer. Unlike enantiomers, which possess identical physical properties in an achiral environment, diastereomers have different melting points, solubilities, and crystallization behaviors. The patent optimizes the solvent system—typically a blend of a non-polar solvent like petroleum ether or n-hexane and a polar solvent like dichloromethane or ethanol—to maximize the solubility difference between these two salts.
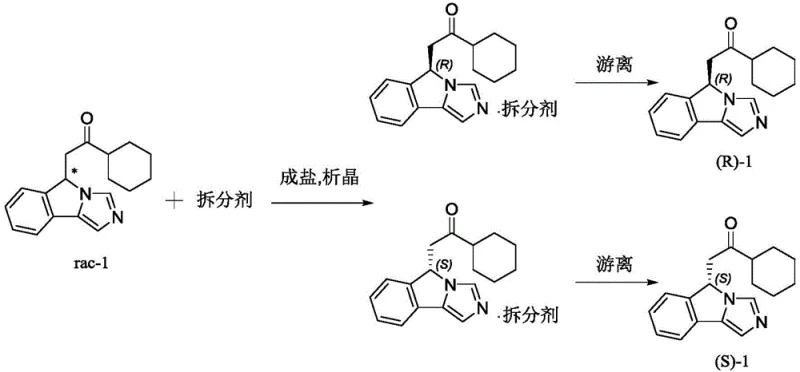
Under controlled temperature conditions, preferably between 20°C and 35°C, the less soluble diastereomeric salt selectively precipitates out of the solution as high-quality crystals. This crystallization step is critical for impurity control, as the crystal lattice formation inherently rejects the unwanted enantiomer and other organic impurities, leading to a significant upgrade in optical purity. Following filtration, the pure salt is treated with an inorganic base, such as sodium carbonate or potassium carbonate, to liberate the free base form of the enantiomer. The absolute configuration of the resulting product is rigorously confirmed using Electronic Circular Dichroism (ECD) spectroscopy, comparing experimental data with theoretical calculations derived from Time-Dependent Density Functional Theory (TD-DFT). This mechanistic understanding ensures that the high-purity pharmaceutical intermediates produced meet the stringent quality standards required for downstream drug synthesis.
How to Synthesize 1-Cyclohexyl-2-(5H-imidazo[5,1-a]isoindole)ethyl-1-one Efficiently
Implementing this resolution protocol requires precise control over stoichiometry, solvent composition, and crystallization parameters to achieve optimal yields and enantiomeric excess (ee). The patent outlines a versatile procedure where the ratio of the resolving agent to the racemic substrate can be adjusted between 1:0.5 and 1:3, allowing manufacturers to tune the process for either maximum yield or maximum purity depending on their specific production goals. The choice of solvent is equally critical, with mixed systems providing the necessary polarity gradient to induce selective precipitation. For R&D teams looking to replicate or scale this process, the following standardized steps provide a foundational framework for execution. Detailed operational parameters regarding temperature ramps and stirring rates should be optimized based on specific reactor geometries.
- Dissolve rac-1 and a chiral resolving agent (such as L-di-p-methylbenzoyl tartaric acid) in a mixed solvent system comprising a non-polar solvent like petroleum ether and a polar solvent like dichloromethane.
- Allow the solution to stand for crystallization at temperatures between 20-35°C to form diastereomeric salt crystals, then filter and wash the crystals with the resolution solvent.
- Suspend the salt complex in water, adjust pH to 9-10 with an inorganic base like sodium carbonate, extract with organic solvent, and dry to obtain the optically pure enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this resolution-based manufacturing route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike asymmetric synthesis which depends on scarce and expensive chiral ligands or transition metal catalysts, this method utilizes chiral tartaric acid derivatives which are commodity chemicals available in bulk quantities from multiple global sources. This diversification of supply mitigates the risk of bottlenecks and price volatility, ensuring a steady flow of materials for continuous production. Furthermore, the ability to recover and recycle the resolving agent significantly reduces the net consumption of chiral auxiliaries, directly impacting the cost of goods sold (COGS) without compromising product quality.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the avoidance of multi-step asymmetric synthesis sequences lead to substantial cost savings. By replacing complex catalytic cycles with a straightforward salt formation and crystallization process, manufacturers can reduce energy consumption, labor hours, and waste disposal costs. The recovery of the resolving agent further enhances economic efficiency, turning what is typically a consumable expense into a reusable asset. This lean manufacturing approach aligns perfectly with initiatives aimed at cost reduction in pharmaceutical intermediates manufacturing, allowing companies to offer competitive pricing while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The robustness of the chemical resolution process translates directly into supply chain resilience. Because the reaction conditions are mild and the reagents are stable, the risk of batch failure due to sensitive catalyst deactivation is minimized. The use of common organic solvents like dichloromethane and petroleum ether ensures that solvent supply is never a constraint. Additionally, the scalability of crystallization processes is well-understood in the chemical industry, meaning that scaling from pilot plant to commercial production involves predictable engineering challenges rather than unknown chemical risks. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that clinical and commercial timelines are met without delay.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a cleaner profile compared to traditional routes. The absence of heavy metals eliminates the need for costly and complex metal scavenging steps, reducing the burden on wastewater treatment facilities. The solvent systems employed are standard and can be efficiently distilled and recycled, minimizing volatile organic compound (VOC) emissions. The high selectivity of the crystallization process reduces the generation of mother liquor waste containing mixed isomers, simplifying downstream purification. These factors collectively support sustainable manufacturing practices, making the process attractive for facilities operating under strict environmental regulations while facilitating the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the applicability of this technology, we have compiled answers to common inquiries regarding the resolution process and product specifications. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for integrating this intermediate into your broader synthesis strategy.
Q: What are the advantages of this chemical resolution method over asymmetric synthesis?
A: Unlike asymmetric synthesis which often requires expensive chiral catalysts and involves multiple steps with harsh conditions, this chemical resolution method utilizes readily available chiral acids, operates under mild conditions, and allows for the recovery and reuse of the resolving agent, significantly simplifying the process.
Q: How is the optical purity of the resolved enantiomers verified?
A: The optical purity is determined using chiral High-Performance Liquid Chromatography (HPLC) combined with Circular Dichroism (CD) detection. Additionally, the absolute configuration is assigned by comparing experimental Electronic Circular Dichroism (ECD) spectra with theoretical calculations.
Q: Can the chiral resolving agent be recovered after the process?
A: Yes, the process includes a specific step where the aqueous layer remaining after extraction is acidified to pH 2-3. This allows the chiral resolving agent to precipitate or be extracted, enabling its recovery and reuse, which contributes to cost efficiency and waste reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NLG919 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of life-saving oncology therapies. Our technical team has thoroughly analyzed the resolution methodology described in CN108239091B and is fully prepared to leverage this knowledge for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC and ECD analysis, to guarantee that every batch of NLG919 precursor meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain for IDO1 inhibitor production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to a reliable source of chiral building blocks backed by deep technical expertise and a commitment to excellence. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can accelerate your drug development timeline.
