Advanced Chiral Resolution of NLG919 Precursors for Commercial Scale-Up
Introduction to Advanced Chiral Resolution Technologies
The pharmaceutical industry's relentless pursuit of potent immuno-oncology therapies has placed significant focus on Indoleamine-2,3-dioxygenase 1 (IDO1) inhibitors, with NLG919 standing out as a prime candidate currently in clinical development. Central to the efficient production of this therapeutic agent is the availability of high-purity chiral precursors, specifically the enantiomers of 1-cyclohexyl-2-(5H-imidazo[5,1-a]isoindole)ethyl-1-one, known technically as rac-1. Patent CN108239091B introduces a groundbreaking chemical resolution methodology that addresses the critical bottlenecks associated with synthesizing these complex heterocyclic intermediates. By leveraging specific tartaric acid derivatives, this technology enables the separation of racemic mixtures into optically pure (R)-1 and (S)-1 isomers with exceptional efficiency. This advancement is not merely a laboratory curiosity but represents a robust industrial solution designed to support the rigorous demands of modern drug supply chains.
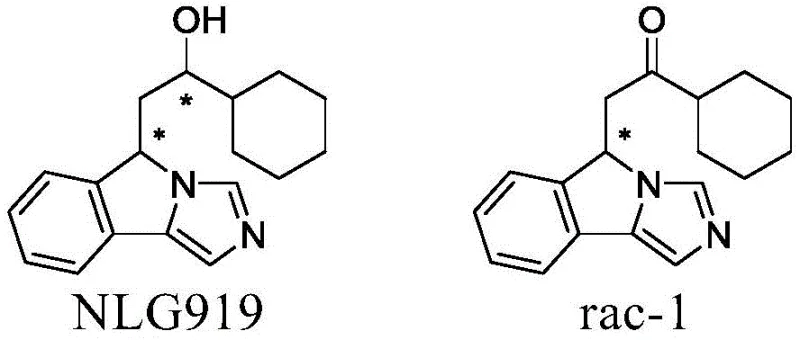
Understanding the structural complexity of these molecules is paramount for R&D teams evaluating process feasibility. As illustrated in the structural comparison, NLG919 contains two chiral centers, leading to four possible stereoisomers, yet biological activity is heavily dependent on specific configurations, particularly the S,S and R,S forms. The precursor rac-1 retains one of these critical chiral centers, making its stereochemical integrity vital for downstream synthesis. The ability to access single enantiomers of rac-1 allows researchers to systematically explore structure-activity relationships and optimize pharmacokinetic profiles without the confounding variables introduced by racemic mixtures. This patent provides the foundational chemistry required to secure a reliable supply of these high-value building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral intermediates for IDO1 inhibitors has relied heavily on asymmetric synthesis or preparative chiral chromatography, both of which present substantial hurdles for commercial manufacturing. Asymmetric synthesis routes, such as those reported in earlier literature, often involve multi-step sequences exceeding seven distinct chemical transformations, each introducing potential yield losses and impurity profiles that complicate purification. Furthermore, these routes frequently necessitate the use of expensive chiral catalysts and harsh reaction conditions that are difficult to control on a large scale. Alternatively, while chiral chromatography can effectively separate enantiomers, it is economically prohibitive for bulk production due to the high cost of chiral stationary phases, limited loading capacities, and significant solvent consumption. These traditional approaches result in extended lead times and inflated production costs, creating vulnerabilities in the supply chain for critical oncology drugs.
The Novel Approach
In stark contrast, the methodology disclosed in CN108239091B utilizes a classical yet highly optimized chemical resolution strategy that bypasses the complexities of asymmetric catalysis. By employing readily available chiral tartaric acid derivatives, specifically D-di-p-methylbenzoyl tartaric acid or its L-enantiomer, the process converts the racemic ketone into diastereomeric salts. These salts exhibit distinct solubility differences in specific mixed solvent systems, allowing for selective crystallization of the desired isomer. This approach drastically simplifies the operational workflow, reducing the number of unit operations and eliminating the need for specialized chromatographic equipment. The result is a streamlined process that offers superior scalability, lower capital expenditure, and a significantly reduced environmental footprint compared to legacy methods, positioning it as the preferred route for cost reduction in API manufacturing.
Mechanistic Insights into Diastereomeric Salt Crystallization
The core of this technological breakthrough lies in the precise molecular recognition between the racemic ketone substrate and the chiral resolving agent. When rac-1 is introduced to a solution containing L-di-p-methylbenzoyl tartaric acid, the carboxylic acid groups of the resolver interact with the basic nitrogen atoms or through hydrogen bonding networks with the ketone functionality, although the primary interaction is likely driven by the formation of stable salt complexes or co-crystals depending on the specific protonation state. The steric bulk of the p-toluoyl groups on the tartaric acid backbone creates a highly differentiated chiral environment. This environment favors the lattice energy of one diastereomeric pair over the other, leading to the preferential precipitation of the less soluble complex, typically the (S)-1 salt in the case of the L-resolver. This thermodynamic driving force is the key to achieving high enantiomeric excess without the need for kinetic control.
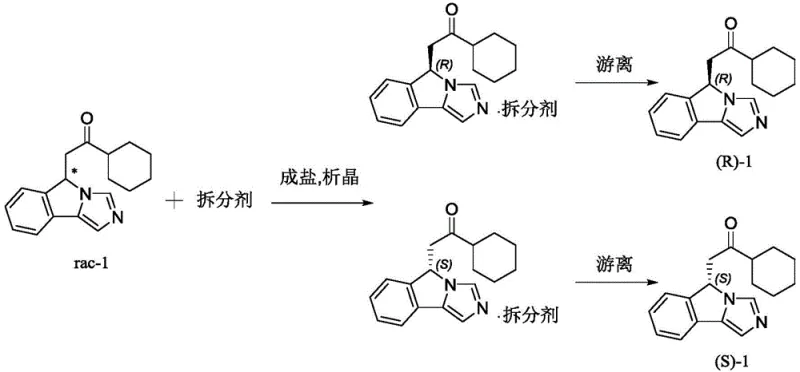
Following the isolation of the crystalline salt, the process employs a straightforward liberation step to recover the free base. By suspending the salt in water and adjusting the pH to an alkaline range of 9-10 using inorganic bases like sodium carbonate, the ionic bond between the drug intermediate and the resolving agent is broken. The free chiral ketone partitions into an organic solvent such as dichloromethane, while the resolving agent remains in the aqueous phase as a salt. Crucially, the patent details a recovery mechanism where the aqueous layer is subsequently acidified to pH 2-3, precipitating the tartaric acid derivative for reuse. This closed-loop system minimizes waste generation and ensures that the cost of the chiral auxiliary is amortized over multiple batches, a critical factor for maintaining margin integrity in competitive pharmaceutical markets.
How to Synthesize 1-Cyclohexyl-2-(5H-imidazo[5,1-a]isoindole)ethyl-1-one Efficiently
Implementing this resolution protocol requires careful attention to solvent selection and temperature control to maximize yield and optical purity. The patent specifies the use of mixed solvent systems, such as dichloromethane combined with petroleum ether or n-hexane, to fine-tune the solubility profile of the diastereomeric salts. The crystallization temperature is maintained between -10°C and 39°C, with a preference for ambient conditions around 20-35°C to facilitate ease of operation. Detailed standardized synthetic steps, including specific mass-to-volume ratios and stirring times, are essential for reproducibility and are outlined in the technical guide below for process engineers.
- Perform salt formation by reacting rac-1 with a chiral resolving agent like L-di-p-methylbenzoyl tartaric acid in a mixed solvent system.
- Induce crystallization at controlled temperatures between -10°C and 39°C to isolate the diastereomeric salt complex.
- Liberate the free base by adjusting pH to 9-10 with inorganic base, followed by extraction and solvent removal to obtain pure (S)-1 or (R)-1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this resolution-based manufacturing route offers transformative benefits regarding cost stability and sourcing security. The elimination of precious metal catalysts and expensive chiral columns removes significant volatility from the raw material cost structure. Instead, the process relies on commodity chemicals like tartaric acid derivatives and common organic solvents, which are widely available from multiple global suppliers, thereby mitigating the risk of single-source dependency. This shift not only lowers the direct cost of goods sold but also simplifies the logistics of raw material management, ensuring a more resilient supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The economic advantage of this method is driven primarily by the recyclability of the resolving agent and the simplicity of the equipment required. Unlike asymmetric synthesis which consumes stoichiometric or catalytic amounts of high-value chiral ligands that are often lost, this process allows for the quantitative recovery of the tartaric acid derivative. Furthermore, the reliance on crystallization rather than chromatography means that existing standard chemical plant infrastructure can be utilized without the need for specialized, high-cost purification units. This results in substantial capital savings and lower operating expenses per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is significantly improved due to the robustness of the chemical resolution process. The reagents used are stable, shelf-stable, and do not require the stringent storage conditions often necessary for sensitive organometallic catalysts. The process tolerance is high, meaning that minor variations in feedstock quality do not necessarily lead to batch failures, a common issue with more fragile catalytic systems. This reliability translates to predictable production schedules and the ability to commit to long-term supply agreements with pharmaceutical partners, reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is inherently green and scalable. Crystallization is a unit operation that scales linearly from grams to tons with minimal re-optimization, unlike chromatographic separations which face severe throughput limitations. Additionally, the ability to recover and reuse the resolving agent drastically reduces the volume of chemical waste generated. The solvent systems employed are standard and can be efficiently distilled and recycled, aligning with modern sustainability goals and regulatory requirements for waste minimization in fine chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical application of the method for industrial partners seeking to optimize their production of IDO1 inhibitor precursors.
Q: What resolving agents are most effective for separating rac-1 enantiomers?
A: The patent identifies D-di-p-methylbenzoyl tartaric acid and L-di-p-methylbenzoyl tartaric acid, along with their hydrates, as highly effective agents for forming separable diastereomeric salts.
Q: How does this resolution method compare to chiral chromatography?
A: Unlike chiral chromatography which suffers from high column costs and limited loading capacity, this chemical resolution utilizes inexpensive tartaric acid derivatives and standard crystallization equipment, making it far more suitable for industrial tonnage production.
Q: Can the chiral resolving agent be recovered after the process?
A: Yes, the process includes a recovery step where the aqueous layer is acidified to pH 2-3, allowing the resolving agent to be extracted and reused, significantly lowering material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Cyclohexyl-2-(5H-imidazo[5,1-a]isoindole)ethyl-1-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation oncology therapeutics depends on the availability of high-quality chiral intermediates. Our technical team has thoroughly analyzed the resolution pathways described in CN108239091B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and advanced analytical capabilities to ensure that every batch of 1-cyclohexyl-2-(5H-imidazo[5,1-a]isoindole)ethyl-1-one meets stringent purity specifications, including precise control over enantiomeric excess and residual solvent levels.
We invite global pharmaceutical partners to collaborate with us to leverage this efficient manufacturing route for your IDO1 inhibitor programs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your development timelines and enhance your project economics.
