Optimizing Gliclazide Intermediate Production: A Technical Breakdown of p-Toluenesulfonylurea Synthesis
Optimizing Gliclazide Intermediate Production: A Technical Breakdown of p-Toluenesulfonylurea Synthesis
The global demand for second-generation sulfonylurea hypoglycemic agents, particularly Gliclazide B.P. 2000, has surged in response to the escalating prevalence of diabetes, necessitating robust and scalable synthetic routes for key intermediates. Patent CN102557997A introduces a transformative preparation method for p-toluenesulfonylurea, a critical precursor in this therapeutic class, addressing long-standing inefficiencies in yield and safety. By shifting from hazardous chlorobenzene systems to a safer toluene-based protocol utilizing inexpensive inorganic bases, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing footprint. The disclosed process achieves exceptional purity metrics exceeding 99.7% via HPLC analysis, demonstrating a significant leap forward in quality control for complex organic synthesis.
This technical insight report dissects the mechanistic advantages and commercial implications of this novel route, specifically tailored for R&D directors and procurement strategists evaluating cost reduction in API manufacturing. The methodology eliminates the reliance on potassium carbonate and high-pressure conditions, replacing them with a streamlined, atmospheric pressure reaction that utilizes sodium hydroxide or potassium hydroxide. Such modifications not only enhance operational safety by mitigating explosion hazards associated with volatile halogenated solvents but also simplify the downstream purification workflow. As we analyze the specific reaction parameters and stoichiometric ratios defined in the patent, it becomes evident that this approach represents a mature, industrial-grade solution capable of supporting commercial scale-up of complex pharmaceutical intermediates with minimal environmental liability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of p-toluenesulfonylurea has been plagued by significant safety and efficiency bottlenecks, primarily relying on chlorobenzene as the reaction medium and potassium carbonate as the base. Literature references, such as those cited in the patent background from the Indian Chemical Engineer, indicate that traditional processes often operate under high-temperature and high-pressure conditions, creating substantial equipment stress and elevated danger coefficients for plant operators. The use of chlorobenzene, a halogenated aromatic hydrocarbon, presents severe environmental and health challenges, including high toxicity and potential carcinogenicity, which complicates waste disposal and regulatory compliance. Furthermore, conventional post-treatment procedures typically necessitate the consumption of large volumes of alcoholic solvents for purification, driving up raw material costs and generating significant volatile organic compound (VOC) emissions. Yields in these legacy processes have historically fluctuated between 70% and 80%, indicating substantial material loss and inconsistent batch-to-batch reproducibility that undermines supply chain stability.
The Novel Approach
In stark contrast, the methodology outlined in CN102557997A employs a "one-pot" two-step strategy that fundamentally reengineers the reaction landscape for improved safety and economics. By substituting chlorobenzene with toluene or xylene, the process utilizes solvents that are not only less toxic but also easier to recover and recycle, thereby aligning with modern green chemistry principles. The replacement of potassium carbonate with sodium hydroxide or potassium hydroxide leverages widely available, low-cost inorganic bases that react efficiently at moderate temperatures ranging from 80°C to 120°C without the need for pressurized vessels. This shift allows for a dramatic reduction in energy consumption and equipment complexity, as the reaction proceeds smoothly under atmospheric pressure with shortened reaction periods. The resulting crude product exhibits superior quality, enabling purification via hot water in many instances rather than expensive alcohols, which directly translates to lower operational expenditures and a reduced environmental footprint for high-purity pharmaceutical intermediates.
Mechanistic Insights into Base-Promoted Sulfonylurea Formation
The core of this synthetic advancement lies in the efficient generation of the reactive sodium or potassium salt of 4-methylbenzenesulfonamide, which serves as the nucleophile in the subsequent condensation with urea. In the initial step, the sulfonamide reacts with the inorganic base in the organic solvent phase, where the base deprotonates the sulfonamide nitrogen to form the corresponding sulfonamide salt. This activation is crucial as it enhances the nucleophilicity of the nitrogen atom, facilitating its attack on the carbonyl carbon of the urea molecule in the second stage. The reaction kinetics are carefully managed by controlling the temperature profile, initially heating to 80-100°C to ensure complete salt formation before introducing urea and raising the temperature to 100-120°C to drive the condensation to completion. This sequential addition prevents side reactions and ensures that the urea is consumed efficiently, minimizing the formation of biuret or other urea-derived impurities that often plague sulfonylurea syntheses.
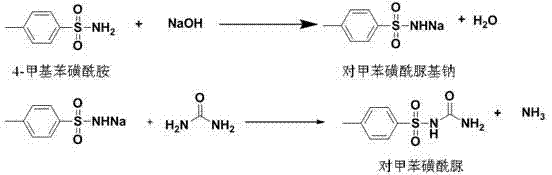
Following the condensation reaction, the workup procedure plays a pivotal role in defining the final purity profile of the high-purity OLED material or pharmaceutical intermediate. The reaction mixture is cooled and treated with water, which dissolves the inorganic salts and excess base, allowing for a clean phase separation where the organic product remains distinct or precipitates upon acidification. The patent specifies acidifying the aqueous phase to a pH of 6-7 using mineral acids, which protonates the sulfonylurea anion to precipitate the neutral product. This precise pH control is essential to prevent the hydrolysis of the sensitive sulfonylurea linkage, which can occur under strongly acidic or basic conditions. The ability to purify the resulting crude solid using hot water, as demonstrated in several embodiments, suggests that the impurity profile is significantly cleaner than in alcohol-based methods, likely due to the high solubility of inorganic byproducts in water versus the organic product. This mechanistic understanding underscores the robustness of the process for reducing lead time for high-purity pharmaceutical intermediates by simplifying the isolation steps.
How to Synthesize p-Toluenesulfonylurea Efficiently
The practical implementation of this synthesis requires strict adherence to the thermal profiles and stoichiometric ratios defined in the patent to maximize yield and purity. The process begins with the suspension of 4-methylbenzenesulfonamide and the inorganic base in toluene, followed by a controlled heating ramp to ensure complete conversion to the salt intermediate before urea addition. Operators must monitor the gas phase temperature closely, maintaining it between 80°C and 120°C during the initial activation phase to drive off any water formed and push the equilibrium toward salt formation. Once the salt is generated, urea is introduced, and the temperature is maintained at 100-120°C for a duration of 3 to 6 hours to ensure full conversion. Detailed standardized synthesis steps see the guide below.
- React 4-methylbenzenesulfonamide with an inorganic base (NaOH or KOH) in toluene or xylene at 80-120°C to form the sodium salt.
- Add urea to the reaction mixture and maintain temperature at 100-120°C for 3-6 hours to complete the condensation.
- Cool the mixture, add water for phase separation, acidify the aqueous layer to pH 6-7, and purify the resulting crude solid via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits centered around cost stability and risk mitigation. The substitution of specialized or hazardous reagents with commodity chemicals like sodium hydroxide and toluene creates a more resilient supply chain that is less susceptible to market volatility or regulatory restrictions on hazardous materials. By eliminating the need for high-pressure reactors and complex safety systems required for chlorobenzene processing, capital expenditure for new production lines can be significantly optimized, while existing facilities can be retrofitted with minimal disruption. The reduction in solvent toxicity also streamlines environmental compliance workflows, reducing the administrative burden and costs associated with hazardous waste disposal and emissions monitoring. These factors collectively contribute to a more predictable and cost-effective manufacturing model for reliable pharmaceutical intermediate suppliers.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the replacement of expensive potassium carbonate with low-cost sodium hydroxide or potassium hydroxide, which are globally available commodity chemicals. Additionally, the ability to utilize water for purification in place of large volumes of ethanol or methanol results in substantial savings on solvent procurement and recovery costs. The higher reaction yield, reported up to 93% compared to the historical 70-80% range, means that less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS). Furthermore, the lower energy requirements due to atmospheric pressure operation and moderate temperatures reduce utility costs, compounding the financial benefits over large-scale production runs.
- Enhanced Supply Chain Reliability: Sourcing 4-methylbenzenesulfonamide, urea, and sodium hydroxide is significantly more straightforward than managing the supply of chlorobenzene, which faces increasing regulatory scrutiny and transportation restrictions globally. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without catastrophic failure, ensures consistent batch output and minimizes the risk of production delays due to process upsets. This reliability is critical for maintaining continuous supply to downstream API manufacturers who depend on just-in-time delivery schedules. The simplified logistics of handling non-halogenated solvents also reduce the complexity of storage and transportation, further securing the supply chain against external disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent's embodiment of a 4m³ reactor setup producing over 500kg of product, indicating readiness for multi-ton annual production capacities. The elimination of chlorobenzene removes a major source of persistent organic pollutants, aligning the manufacturing process with increasingly stringent global environmental regulations such as REACH and EPA guidelines. The potential to recycle toluene and the reduced generation of hazardous liquid waste simplify the wastewater treatment process, lowering the overall environmental impact. This alignment with green chemistry principles not only future-proofs the production facility against tightening regulations but also enhances the corporate sustainability profile, which is increasingly valued by international pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this p-toluenesulfonylurea synthesis method. These answers are derived directly from the experimental data and technical specifications provided in patent CN102557997A, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does this method improve upon traditional chlorobenzene-based synthesis?
A: This method replaces toxic chlorobenzene with safer solvents like toluene or xylene, significantly reducing environmental hazards and explosion risks while maintaining high yields up to 93%.
Q: What are the purity specifications achievable with this process?
A: Through optimized acidification and recrystallization steps, the process consistently achieves HPLC purity levels greater than 99.7%, meeting stringent pharmaceutical standards.
Q: Can water be used for purification instead of alcohol?
A: Yes, unlike conventional methods requiring alcohol, this innovation allows for purification using hot water, which drastically lowers solvent costs and simplifies waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Toluenesulfonylurea Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving medications like Gliclazide. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and GC-MS instrumentation to guarantee that every batch of p-toluenesulfonylurea meets or exceeds the 99.7% purity benchmark established by leading industry standards. Our commitment to quality assurance ensures that our clients receive a product that is fully characterized and ready for immediate use in downstream API synthesis.
We invite pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production volume needs, ensuring a partnership built on transparency, technical excellence, and mutual growth.
