Advanced Green Synthesis of p-Toluenesulfonylurea for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, particularly for critical intermediates used in antidiabetic medications. Patent CN111747872A introduces a groundbreaking synthetic methodology for p-toluenesulfonylurea, a pivotal precursor in the production of Gliclazide, which addresses long-standing environmental and efficiency challenges in fine chemical synthesis. This innovative approach replaces hazardous organic solvents with a benign water-ethanol mixture, fundamentally altering the safety profile and economic viability of large-scale production. By leveraging sodium cyanate as a key reagent instead of traditional urea condensation, the process eliminates the generation of toxic ammonia gas, a significant occupational hazard in conventional facilities. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a substantial leap forward in process intensification and regulatory compliance. The method not only promises yields exceeding 90% under optimized conditions but also ensures a purity profile greater than 99%, meeting the stringent quality standards required for global API manufacturing. As we analyze the technical nuances of this patent, it becomes evident that adopting such green chemistry principles is no longer optional but essential for maintaining competitiveness in the modern fine chemical intermediates market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of p-toluenesulfonylurea has relied heavily on condensation reactions between p-toluenesulfonamide and urea, typically catalyzed by strong bases such as sodium hydroxide or potassium carbonate. These legacy processes, while chemically feasible, suffer from severe drawbacks that impact both operational safety and environmental sustainability. A primary concern is the extensive use of volatile organic compounds (VOCs) like toluene, chlorobenzene, or 1,2-dichloroethane as reaction media, which necessitate complex solvent recovery systems and pose significant fire and health risks to plant personnel. Furthermore, the thermal decomposition of urea at high reaction temperatures inevitably leads to the release of substantial quantities of ammonia gas, requiring specialized scrubbing equipment to prevent atmospheric pollution. From a quality perspective, these harsh conditions often promote the formation of difficult-to-remove impurities, specifically disulfonylurea by-products, which complicate downstream purification and reduce overall material throughput. The combination of toxic solvent waste, gaseous emissions, and moderate yields hovering around 80% creates a high-cost operational burden that is increasingly untenable in a regulatory environment focused on carbon footprint reduction and worker safety.
The Novel Approach
In stark contrast to these archaic methods, the technology disclosed in patent CN111747872A utilizes a remarkably simple yet effective aqueous-ethanol solvent system that fundamentally resolves the issues of toxicity and waste generation. By dissolving p-toluenesulfonamide and sodium cyanate in a mixture of water and ethanol, the reaction proceeds under much milder conditions, typically between 60°C and 80°C, thereby eliminating the need for high-energy heating and the associated safety risks of pressurized vessels. The substitution of urea with sodium cyanate is a strategic masterstroke, as it prevents the evolution of ammonia gas entirely, creating a closed-loop system that is inherently safer for operators and the surrounding community. Moreover, the solubility characteristics of the reactants in this binary solvent system allow for precise control over reaction kinetics, minimizing side reactions and ensuring that the target sulfonylurea is formed with exceptional selectivity. This novel approach not only simplifies the workflow by removing the need for anhydrous conditions but also drastically reduces the cost of waste disposal, as the aqueous mother liquor can be treated with standard neutralization protocols rather than expensive hazardous waste incineration. For cost reduction in pharmaceutical intermediate manufacturing, this shift from organic to aqueous-organic media offers a compelling value proposition that aligns perfectly with modern green chemistry mandates.
Mechanistic Insights into Sodium Cyanate-Mediated Urea Formation
The core of this synthetic breakthrough lies in the unique nucleophilic substitution mechanism facilitated by the cyanate ion, which differs significantly from the condensation pathways seen in traditional urea-based syntheses. As illustrated in the reaction scheme below, the process initiates with the nucleophilic attack of the lone pair electrons on the amino group of p-toluenesulfonamide onto the electrophilic carbon atom of the cyanate ion. This interaction forms a transient intermediate where the negative charge is delocalized, eventually undergoing a proton transfer and rearrangement to establish the stable urea linkage. Unlike acid-catalyzed condensations that rely on the elimination of small molecules like water or ammonia to drive equilibrium, this addition reaction is thermodynamically favorable and proceeds efficiently without the need for dehydrating agents. The absence of aggressive acidic or basic catalysts further protects the integrity of the sulfonamide moiety, preventing hydrolysis or degradation that could lead to complex impurity profiles. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters, as it highlights the importance of maintaining specific pH levels and temperature ranges to maximize the collision frequency between the nucleophile and the cyanate species.
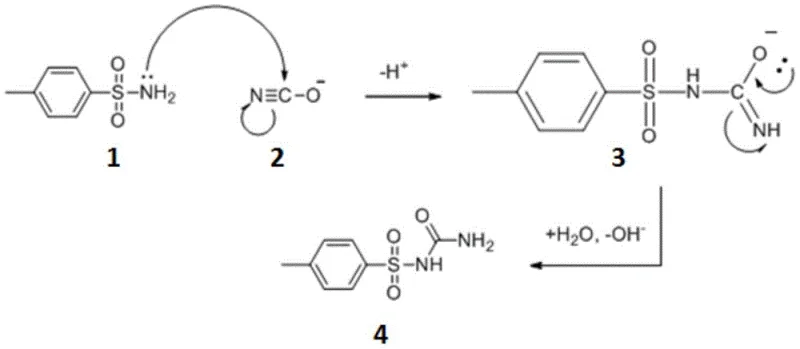
Beyond the primary reaction pathway, the impurity control mechanism inherent in this process is equally noteworthy for ensuring high-purity outputs suitable for sensitive pharmaceutical applications. The patent data suggests that the specific solvation environment provided by the water-ethanol mixture plays a critical role in suppressing the formation of disulfonylurea derivatives, which are common contaminants in conventional routes. By carefully controlling the stoichiometry, specifically maintaining a slight excess of sodium cyanate (molar ratio 1:1.2), the reaction drives towards complete consumption of the sulfonamide starting material without promoting over-reaction or polymerization. The subsequent workup procedure, involving hot filtration to remove unreacted solids followed by acidification of the mother liquor, acts as a powerful purification step that leverages the differential solubility of the product versus potential by-products. When the pH is adjusted to the acidic range of 1-2, the sodium salt of the intermediate undergoes an enolic rearrangement and precipitates as the free acid form of p-toluenesulfonylurea, leaving soluble impurities behind in the aqueous phase. This elegant integration of reaction chemistry and crystallization physics ensures that the final isolated material consistently achieves purity levels above 99%, significantly reducing the burden on downstream recrystallization steps.
How to Synthesize p-Toluenesulfonylurea Efficiently
Implementing this synthesis route requires precise attention to solvent ratios and thermal management to replicate the high yields reported in the patent literature. The process begins by preparing a homogeneous solution of the reactants, where the balance between water and ethanol is critical; water is necessary to dissolve the inorganic sodium cyanate, while ethanol is required to solubilize the organic p-toluenesulfonamide. Once the mixture is heated to reflux, typically around 78°C, the system must be maintained at this temperature for a sufficient duration, generally 7 to 8 hours, to ensure the reaction reaches completion without premature termination. Following the reaction period, a hot filtration step is employed to remove any trace amounts of unreacted starting materials that may have precipitated upon cooling, ensuring that the subsequent crystallization is not seeded with impurities. The detailed standardized synthesis steps, including specific stirring rates and cooling profiles, are outlined in the guide below to assist technical teams in replicating this robust protocol.
- Dissolve p-toluenesulfonamide and sodium cyanate in a mixed solvent of water and ethanol, maintaining a molar ratio of approximately 1: 1.2.
- Heat the reaction mixture to reflux temperatures between 60°C and 80°C for 7 to 8 hours to ensure complete conversion.
- Filter the hot mixture to remove unreacted solids, then acidify the mother liquor to pH 1-2 to precipitate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this green synthesis route offers tangible benefits that extend far beyond simple chemical yield, impacting the total cost of ownership and logistical reliability of the supply chain. The elimination of chlorinated and aromatic solvents removes the need for complex solvent recovery distillation columns and reduces the regulatory burden associated with storing and transporting hazardous flammable liquids. This simplification of the infrastructure directly translates to lower capital expenditure for new plants and reduced maintenance costs for existing facilities, allowing for more competitive pricing structures in the long term. Furthermore, the absence of ammonia gas generation eliminates the requirement for specialized gas scrubbing towers and continuous emission monitoring systems, thereby lowering operational overhead and minimizing the risk of production stoppages due to environmental compliance violations. By switching to a water-ethanol system, manufacturers can also leverage the widespread availability and low cost of these commodity solvents, insulating the production process from the volatility of petrochemical-derived solvent markets. These factors combined create a resilient supply chain capable of sustaining high-volume production without the bottlenecks typically associated with hazardous waste disposal and strict environmental permitting.
- Cost Reduction in Manufacturing: The transition to an aqueous-ethanol solvent system fundamentally alters the cost structure by removing the most expensive components of traditional chemical processing: hazardous waste disposal and solvent recovery. Since water and ethanol are significantly cheaper and easier to handle than toluene or chlorobenzene, the raw material costs are inherently lower, and the energy required for solvent distillation is drastically reduced due to the lower boiling point of ethanol compared to higher boiling aromatics. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to by-products, improving the overall atom economy and ensuring that a greater proportion of input costs are converted into saleable product. This efficiency gain allows for substantial cost savings that can be passed down to customers or reinvested into further process optimization initiatives.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like sodium cyanate and ethanol enhances supply chain security by reducing dependence on specialized reagents that may be subject to geopolitical shortages or transportation restrictions. The simplified process flow, which avoids the use of highly regulated toxic solvents, also accelerates the permitting process for new production lines, enabling faster scale-up from pilot to commercial volumes. This agility ensures that suppliers can respond more rapidly to fluctuations in market demand for antidiabetic intermediates, providing a stable and continuous supply stream for downstream API manufacturers. The robustness of the reaction conditions further reduces the likelihood of batch failures, ensuring consistent delivery schedules and strengthening the trust between suppliers and their pharmaceutical partners.
- Scalability and Environmental Compliance: The inherent safety of the water-ethanol system makes this process exceptionally scalable, as it avoids the thermal runaway risks associated with exothermic reactions in organic solvents. From an environmental standpoint, the process generates minimal hazardous waste, aligning perfectly with global initiatives to reduce the pharmaceutical industry's ecological footprint. The aqueous waste streams generated are biodegradable and easy to treat, significantly lowering the fees associated with third-party waste management services. This compliance advantage future-proofs the manufacturing asset against tightening environmental regulations, ensuring long-term operational continuity without the need for costly retrofits or process changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and practical application scenarios. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the cyanate-based approach over traditional urea condensation methods, focusing on safety, purity, and operational efficiency. We encourage stakeholders to review these insights to fully appreciate the strategic value of this technology.
Q: Why is the water-ethanol solvent system preferred over traditional organic solvents?
A: Traditional methods utilize toxic solvents like toluene or chlorobenzene which pose significant environmental and safety hazards. The water-ethanol system described in patent CN111747872A is non-toxic, reduces VOC emissions, and simplifies waste treatment protocols.
Q: How does this method prevent the formation of ammonia gas?
A: Unlike urea-based condensation methods that release ammonia as a by-product, this synthesis uses sodium cyanate as the nitrogen source. The reaction mechanism involves nucleophilic attack without the elimination of volatile ammonia, improving workplace safety.
Q: What is the expected purity profile of the final intermediate?
A: The patent data indicates that through careful filtration and acidification steps, the process achieves purity levels exceeding 99% (HPLC), effectively minimizing disulfonylurea impurities common in older synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Toluenesulfonylurea Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the long-term success of the global pharmaceutical supply chain. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one described in patent CN111747872A are translated into robust, industrial-grade realities. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By partnering with us, you gain access to a supply chain that prioritizes safety, sustainability, and consistency, allowing you to focus on your core drug development goals without worrying about raw material variability.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis route to your specific volume requirements and quality standards. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits of switching to this green manufacturing process. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how NINGBO INNO PHARMCHEM can become your strategic partner in the production of high-quality antidiabetic intermediates.
