Advanced Four-Step Synthetic Route for Agomelatine Intermediates Enhancing Commercial Scalability
Introduction to the Novel Agomelatine Synthesis Protocol
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for complex antidepressant agents like Agomelatine. Patent CN101792400A introduces a transformative four-step synthetic methodology that addresses the longstanding inefficiencies associated with earlier production techniques. By utilizing 7-methoxy-1-tetralone as a strategic starting material, this protocol leverages a sequence involving n-butyllithium-mediated cyanohydrin formation, followed by a critical DDQ-catalyzed aromatization, lithium aluminum hydride reduction, and final acetylation. This approach not only streamlines the manufacturing workflow but also eliminates the reliance on hazardous solvents such as benzene, which were prevalent in legacy processes. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for evaluating potential suppliers who can offer cost reduction in API manufacturing without compromising on regulatory compliance or product quality.
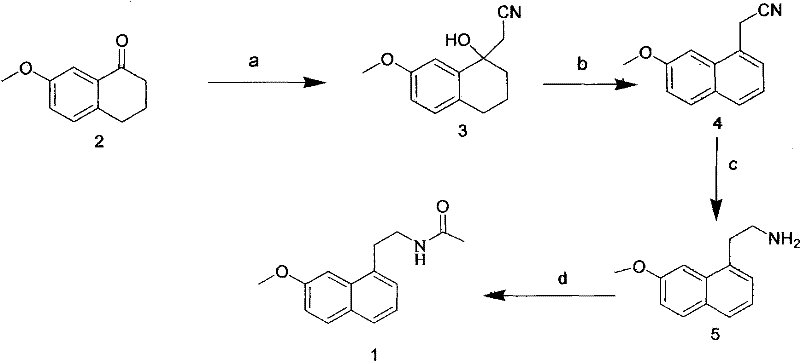
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Agomelatine, such as those described in patent EP0447285, relied heavily on an eight-step sequence initiating with a Reformatsky reaction between 7-methoxy-1-tetralone and ethyl bromoacetate. This antiquated approach suffered from severe drawbacks, including the necessity of using benzene, a solvent with significant toxicity and environmental impact, which complicates waste disposal and worker safety protocols. Furthermore, the multi-step nature of the traditional pathway inherently accumulates yield losses at every stage, resulting in a low overall recovery rate that drives up the cost of goods sold. Another prior art method, disclosed in EP156420, attempted to address some issues but introduced new hazards by employing deleterious allyl methacrylate as a hydrogen acceptor at extreme temperatures exceeding 200°C. Such harsh thermal conditions not only consume excessive energy but also pose significant risks of thermal runaway and equipment degradation, making the process unsuitable for reliable commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN101792400A achieves the target molecule through a concise four-step sequence that prioritizes operational simplicity and safety. The innovation lies primarily in the efficient construction of the key intermediate, (7-methoxy-1-naphthyl) acetonitrile, achieved in just two steps with high controllability. By replacing the high-temperature dehydrogenation with a DDQ-mediated oxidation performed at moderate temperatures between 50°C and 150°C, the process drastically reduces energy consumption and eliminates the need for toxic hydrogen acceptors. The subsequent reduction and acylation steps utilize standard, commercially available reagents like lithium aluminum hydride and acetyl chloride, ensuring that the supply chain remains resilient and不受制于 exotic materials. This streamlined architecture allows for a reliable pharmaceutical intermediate supplier to maintain consistent batch-to-batch quality while significantly simplifying the post-reaction treatment and purification workflows.
Mechanistic Insights into DDQ-Catalyzed Aromatization and Reduction
The cornerstone of this synthetic strategy is the oxidative aromatization step, where the tetrahydronaphthalene ring system is converted into the fully aromatic naphthalene core. In this mechanism, 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) acts as a potent hydride acceptor, facilitating the removal of hydrogen atoms from the saturated ring without requiring the extreme thermal energy seen in catalytic dehydrogenation methods. The reaction proceeds smoothly in solvents such as acetic acid or toluene, where the DDQ abstracts hydrides to form the aromatic system while being reduced to its hydroquinone form, which is easily removed during the aqueous workup. This mechanistic elegance ensures that the sensitive nitrile functionality remains intact, preventing side reactions that could lead to difficult-to-remove impurities. For technical teams, this implies a cleaner reaction profile and a simplified isolation process, often requiring only filtration and recrystallization to achieve high purity specifications.
Following aromatization, the conversion of the nitrile group to the primary amine via lithium aluminum hydride (LiAlH4) reduction is a critical transformation that demands precise control. The mechanism involves the nucleophilic attack of hydride ions on the electrophilic carbon of the nitrile group, forming an imine-aluminum complex which is subsequently reduced to the amine upon hydrolysis. The patent specifies a careful quenching protocol involving the sequential addition of water and sodium hydroxide solution to safely decompose the aluminum salts and prevent exothermic spikes. This attention to detail in the reduction phase is vital for minimizing the formation of secondary amines or other reduction by-products, thereby ensuring that the final Agomelatine product meets the stringent purity specifications required for antidepressant medications. The final acylation with acetyl chloride then proceeds under mild conditions, capping the synthesis with a high-yielding amide bond formation.
How to Synthesize Agomelatine Efficiently
Implementing this synthesis requires strict adherence to the temperature profiles and stoichiometric ratios defined in the patent to maximize yield and safety. The process begins with the cryogenic generation of the lithiated acetonitrile species, followed by the controlled addition of the ketone substrate to form the cyanohydrin intermediate. Subsequent oxidation and reduction steps must be monitored closely to prevent over-reaction or decomposition, particularly during the hydride reduction phase. The detailed standardized synthesis steps below outline the precise operational parameters required for successful execution.
- React 7-methoxy-1-tetralone with n-butyllithium and acetonitrile at -78°C to 0°C to form the cyanohydrin intermediate.
- Oxidize the cyanohydrin using DDQ in acetic acid or toluene at 50°C to 150°C to achieve aromatization.
- Reduce the nitrile group using lithium aluminum hydride in THF at 0°C to 60°C to generate the primary amine.
- Acylate the amine with acetyl chloride in the presence of a base like triethylamine to obtain final Agomelatine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this four-step synthetic route offers profound advantages in terms of cost structure and logistical stability. By eliminating the need for eight distinct reaction stages, the process inherently reduces the consumption of solvents, reagents, and labor hours, leading to substantial cost savings in the overall manufacturing budget. The avoidance of benzene and high-temperature dehydrogenation agents not only lowers the environmental compliance burden but also mitigates the risk of supply disruptions caused by the restricted availability of hazardous chemicals. This robustness ensures a more predictable lead time for high-purity pharmaceutical intermediates, allowing downstream manufacturers to plan their production schedules with greater confidence and efficiency.
- Cost Reduction in Manufacturing: The consolidation of the synthesis into four high-yielding steps fundamentally alters the economic model of Agomelatine production. By removing four entire unit operations compared to the traditional Reformatsky route, the facility saves significantly on utility costs, reactor occupancy time, and waste treatment expenses. The use of DDQ, while a specialized reagent, is offset by the elimination of expensive palladium catalysts and the energy-intensive heating required for thermal dehydrogenation. Furthermore, the high yields reported in the patent examples, such as 95% for the first step and 80% for the oxidation, mean that less raw material is wasted, directly improving the margin profile for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: A major bottleneck in pharmaceutical supply chains is the dependency on exotic or highly regulated reagents. This protocol relies on commodity chemicals like acetonitrile, acetic acid, and acetyl chloride, which are readily available from multiple global sources. This diversification of the supply base reduces the risk of single-source failure and price volatility. Additionally, the simplified workup procedures, which often involve straightforward filtration and crystallization rather than complex chromatographic separations, accelerate the throughput of the manufacturing plant. This efficiency translates to shorter cycle times and the ability to respond more rapidly to fluctuations in market demand for antidepressant therapies.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but the mild conditions of this route facilitate a smoother transition from pilot plant to commercial production. The absence of benzene removes a significant regulatory hurdle, as facilities do not need to invest in specialized containment systems for carcinogenic solvents. The lower operating temperatures in the aromatization step reduce the thermal load on cooling systems and minimize the risk of thermal incidents. From an environmental perspective, the reduced solvent usage and the generation of less hazardous waste streams align with modern green chemistry principles, enhancing the corporate sustainability profile of the manufacturer and ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents a superior choice for industrial application. Understanding these details helps stakeholders make informed decisions regarding technology transfer and vendor qualification.
Q: Why is the DDQ oxidation method superior to previous dehydrogenation techniques?
A: Previous methods utilized toxic allyl methacrylate at temperatures exceeding 200°C, leading to low yields and safety hazards. The DDQ method operates at milder temperatures (50°C-150°C) with higher controllability and simpler workup.
Q: How does this four-step route improve upon the traditional eight-step Reformatsky synthesis?
A: The traditional route involves eight steps including hydrolysis, chlorination, and ammonification, resulting in low total recovery. This novel route condenses the process to four high-yielding steps, significantly reducing material loss and processing time.
Q: What are the critical safety considerations for the reduction step?
A: The reduction utilizes lithium aluminum hydride, which requires strict moisture control and careful quenching protocols. The patent specifies a controlled addition of water and sodium hydroxide solution to safely decompose excess hydride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Agomelatine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep technical expertise and rigorous process control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN101792400A route are fully realized in large-scale manufacturing. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor impurity profiles at every stage of the synthesis. This commitment to quality ensures that our Agomelatine intermediates meet the exacting standards required by global regulatory bodies, providing our partners with a secure and compliant supply source.
We invite pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this four-step route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to validate the superior performance and reliability of our manufacturing capabilities for your next project.
