Advanced Iridium-Catalyzed Asymmetric Reductive Amination for Commercial Scale Chiral Amine Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access chiral building blocks, which serve as the foundational scaffolds for numerous life-saving medications. Patent CN110862324A introduces a groundbreaking direct synthesis method for chiral secondary amine compounds, addressing a long-standing challenge in asymmetric catalysis. This technology leverages an iridium-catalyzed asymmetric reductive amination strategy, utilizing simple ketones and primary alkylamines as starting materials to produce high-value chiral amines in a single operational step. The significance of this innovation cannot be overstated, as chiral amines are critical pharmacophores found in major drugs such as Clopidogrel, Cinacalcet, Rivastigmine, and Selegiline. By enabling the direct use of primary alkylamines as the nitrogen source, this method bypasses the need for complex multi-step sequences or inefficient resolution processes, offering a streamlined route that is highly attractive for industrial scale-up.
Furthermore, the patent highlights the exceptional stereoselectivity achievable with this protocol, reporting enantiomeric excess values reaching up to 95%. This level of precision is vital for regulatory compliance in drug manufacturing, where impurity profiles must be tightly controlled. The methodology represents a significant leap forward in green chemistry principles, maximizing atom economy while minimizing waste generation. For procurement and supply chain leaders, this translates to a potential reduction in raw material costs and processing time, making it a compelling candidate for the production of reliable pharmaceutical intermediates. The robustness of the catalytic system, combined with the availability of inexpensive starting materials, positions this technology as a cornerstone for next-generation amine synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of optically pure chiral secondary amines has been fraught with inefficiencies and economic drawbacks. The most common historical approach involved the resolution of racemic mixtures, a process that inherently caps the maximum theoretical yield at 50%, resulting in substantial material waste and increased disposal costs. Alternatively, chemists often relied on multi-step synthetic routes involving the use of chiral auxiliaries or protecting groups, which necessitate additional reagents, solvents, and purification steps. These conventional methods not only extend the overall lead time for high-purity pharmaceutical intermediates but also introduce more opportunities for yield loss at each stage. Moreover, many existing catalytic methods struggle with primary alkylamines as substrates, often suffering from poor reactivity or low stereocontrol, thereby limiting their utility in synthesizing complex drug molecules.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a highly active iridium complex coordinated with a chiral phosphoramidite ligand to facilitate asymmetric reductive amination directly. This method allows for the one-pot conversion of ketones and primary alkylamines into chiral secondary amines with remarkable efficiency. By eliminating the need for resolution or excessive protection-deprotection cycles, the process drastically simplifies the manufacturing workflow. The use of hydrogen gas as the reducing agent is both cost-effective and environmentally benign, producing water as the only byproduct. This shift towards a direct catalytic hydrogenation strategy not only enhances the overall yield, with reported values exceeding 90% in specific embodiments, but also significantly reduces the environmental footprint associated with chemical manufacturing, aligning perfectly with modern sustainability goals.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Reductive Amination
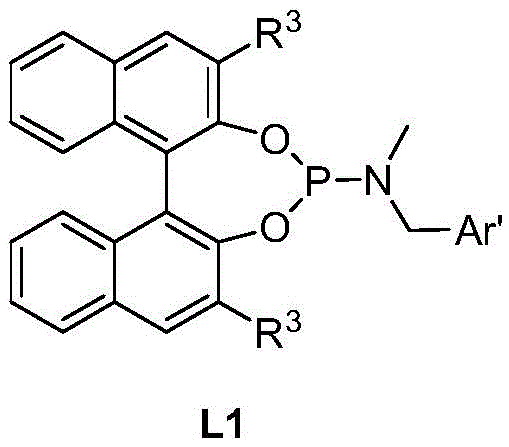
The core of this technological breakthrough lies in the sophisticated design of the catalytic system, which orchestrates the stereochemical outcome of the reaction with high fidelity. The catalyst is generated in situ from an iridium salt, such as (1,5-cyclooctadiene)iridium(I) dichloride dimer, and a specialized chiral phosphoramidite ligand. These ligands, often derived from binaphthyl or tetrahydronaphthyl backbones, create a rigid chiral pocket around the metal center. This steric environment dictates the approach of the substrate, ensuring that hydrogen addition occurs selectively to one face of the imine intermediate formed between the ketone and the amine. The presence of additives, such as titanates or organic bases, further modulates the electronic properties of the catalyst and facilitates the dehydration step required to form the imine species prior to reduction.
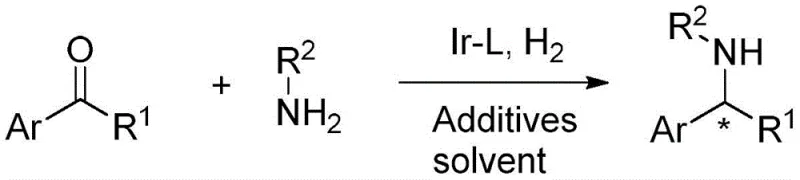
The versatility of this catalytic system is demonstrated by its tolerance to a wide range of functional groups on both the aromatic and aliphatic portions of the substrates. Whether the ketone bears electron-withdrawing groups like trifluoromethyl or electron-donating groups like methoxy, the catalyst maintains high activity and selectivity. The mechanism likely involves the formation of an iridium-hydride species which transfers hydride to the prochiral imine intermediate. The chiral ligand ensures that this transfer happens enantioselectively, locking in the desired configuration at the newly formed stereocenter. This precise control is essential for producing drug substances where the wrong enantiomer could be inactive or even toxic, underscoring the value of this method for producing high-purity OLED materials or pharmaceutical ingredients.
How to Synthesize Chiral Secondary Amines Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to maximize the benefits of the catalytic system. The process is designed to be operationally simple, typically involving the mixing of the ketone, amine, catalyst components, and additives in an ester solvent such as ethyl acetate. The reaction is then conducted under a hydrogen atmosphere in a high-pressure vessel. While the specific stoichiometry and conditions can be tuned based on the substrate, the general protocol provides a robust framework for generating diverse chiral amine libraries. For detailed standard operating procedures and optimization parameters, please refer to the technical guide below.
- Combine the ketone substrate, primary alkylamine, iridium catalyst precursor, chiral phosphoramidite ligand, and additives (such as titanates or organic bases) in a suitable ester solvent.
- Transfer the mixture to a high-pressure reactor, purge with hydrogen gas multiple times to ensure an inert atmosphere, and pressurize to approximately 50 atmospheres.
- Stir the reaction at 50°C for 20 hours, then quench with saturated sodium bicarbonate, separate the organic phase, and purify via distillation or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers transformative advantages for supply chain stability and cost management in the fine chemical sector. By shifting from resolution-based methods to direct asymmetric synthesis, manufacturers can effectively double their theoretical output from the same amount of starting material. This fundamental improvement in atom economy directly correlates to substantial cost savings in raw material procurement. Furthermore, the simplification of the process flow—removing multiple isolation and purification steps—reduces the consumption of solvents and energy, leading to a leaner and more sustainable manufacturing operation. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the use of inexpensive, commercially available ketones and primary amines significantly lower the bill of materials. Additionally, the high catalytic efficiency means that less catalyst loading may be required over time as the process is optimized, further driving down production costs without compromising quality. The reduction in processing steps also lowers labor and utility costs associated with extended reaction times and complex workups.
- Enhanced Supply Chain Reliability: Because the starting materials are simple commodity chemicals rather than specialized chiral pools, the risk of supply disruption is minimized. The robustness of the reaction conditions, operating at moderate temperatures and pressures, ensures consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous production schedules and meeting just-in-time delivery requirements for downstream drug manufacturers.
- Scalability and Environmental Compliance: The use of hydrogen gas and ester solvents aligns well with green chemistry initiatives, simplifying waste treatment and regulatory compliance. The process is inherently scalable, having been demonstrated to work efficiently in batch reactors, which facilitates the transition from laboratory discovery to commercial scale-up of complex pharmaceutical intermediates. This scalability ensures that supply can be rapidly ramped up to meet market demand spikes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this patented synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: What is the primary advantage of this iridium-catalyzed method over traditional resolution?
A: Unlike traditional resolution which theoretically limits yield to 50%, this asymmetric reductive amination allows for direct synthesis with high enantioselectivity (up to 95% ee) and high yields (over 90%), significantly improving atom economy.
Q: Can this method be used for synthesizing Cinacalcet intermediates?
A: Yes, the patent explicitly demonstrates the successful synthesis of Cinacalcet (1-1) intermediates using 1-naphthylethyl ketone and 3-(3-trifluoromethyl)-propylamine, achieving 93% yield and 95% enantioselectivity.
Q: What type of ligands are required for this transformation?
A: The process utilizes specialized chiral phosphoramidite ligands (such as L1 or L2 derivatives based on binaphthyl or tetrahydronaphthyl backbones) which are crucial for inducing the desired stereochemistry during the hydrogenation step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Secondary Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced catalytic technologies in maintaining a competitive edge in the pharmaceutical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative methods like this iridium-catalyzed reductive amination can be seamlessly transferred to large-scale manufacturing. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical purity.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral amine synthesis can accelerate your development timeline and optimize your supply chain.
