Advanced Synthesis of Cyclopropylamine: A Cost-Effective Route for Pharmaceutical Intermediates
The chemical industry is constantly evolving, driven by the relentless pursuit of more efficient, cost-effective, and environmentally sustainable synthetic pathways for critical intermediates. A recent breakthrough detailed in patent CN114989018A introduces a robust methodology for the synthesis of cyclopropylamine, a vital building block extensively utilized in the formulation of herbicides, pesticides, and various pharmaceutical active ingredients. This innovative approach fundamentally shifts the paradigm from traditional, resource-intensive routes that rely on scarce precursors like 1,3-propanediol, towards a streamlined process anchored by the abundant and economically favorable 1,3-dichloropropane. By leveraging a strategic combination of nucleophilic substitution, intramolecular cyclization, and a classic Hoffmann degradation, this technology not only addresses the longstanding technical challenges of harsh reaction conditions and low yields but also opens new avenues for supply chain optimization. For global procurement leaders and R&D directors, understanding the nuances of this patent is essential for securing a competitive edge in the sourcing of high-value cyclic amines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of cyclopropylamine has been fraught with significant logistical and chemical hurdles that have constrained supply reliability and inflated manufacturing costs. Traditional synthetic routes often commence with 1,3-propanediol, a raw material that is not only more expensive but also requires a complex series of activation steps, such as bromination, before it can undergo cyanation and subsequent cyclization. These multi-step sequences inherently accumulate inefficiencies, as each transformation introduces potential yield losses and generates substantial quantities of hazardous waste streams that require costly disposal protocols. Furthermore, the reaction conditions associated with these legacy methods are frequently severe, necessitating extreme temperatures or pressures that demand specialized, high-maintenance reactor infrastructure. The cumulative effect of these factors is a fragile supply chain vulnerable to raw material price volatility and regulatory scrutiny regarding environmental compliance, making the conventional production of cyclopropylamine a bottleneck for downstream manufacturers of agrochemicals and fine chemicals.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the methodology disclosed in CN114989018A presents a elegantly simplified trajectory that leverages the inherent reactivity of 1,3-dichloropropane to achieve superior outcomes with reduced operational friction. This novel approach initiates with a direct nucleophilic substitution using sodium cyanide, bypassing the need for prior halogenation of a diol and immediately establishing the carbon-nitrogen bond required for the final amine structure. The subsequent steps are designed to maximize atom economy, particularly through the recovery and recycling of unreacted 1,3-dichloropropane via rectification, which drastically minimizes raw material consumption. By operating within a moderate temperature window of 60-100°C, the process eliminates the need for energy-intensive heating or cryogenic cooling, thereby lowering the overall carbon footprint of the manufacturing facility. This strategic redesign of the synthetic route translates directly into tangible commercial benefits, offering a pathway to significantly reduced production costs and enhanced process stability that is critical for maintaining continuous supply in high-demand markets.
Mechanistic Insights into the Multi-Step Transformation
The core of this technological advancement lies in the precise orchestration of three distinct chemical transformations that convert a simple linear dichloride into a strained cyclic amine with high fidelity. The initial phase involves the reaction of 1,3-dichloropropane with a sodium cyanide solution under reflux conditions, where a selective nucleophilic substitution occurs to generate 4-chlorobutyronitrile. This step is critical as it sets the stage for the subsequent ring closure, and the use of excess 1,3-dichloropropane ensures that the mono-substituted product is favored over the bis-substituted byproduct, thereby simplifying the purification landscape. 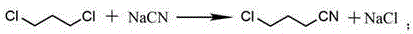
Following the formation of the nitrile intermediate, the process advances to a sophisticated cyclization and hydrolysis sequence mediated by sodium hydroxide. In this pivotal stage, the terminal chloride undergoes an intramolecular nucleophilic attack to close the three-membered ring, forming cyclopropanecarbonitrile, which is subsequently hydrolyzed under alkaline conditions to yield cyclopropanecarboxamide. 
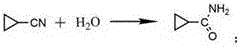 The control of pH and temperature during this phase is paramount to prevent ring-opening side reactions, ensuring that the strained cyclopropane motif remains intact. Finally, the synthesis culminates in a Hoffmann degradation reaction, where the amide is treated with sodium hypochlorite and sodium hydroxide to excise the carbonyl group and release the primary amine.
The control of pH and temperature during this phase is paramount to prevent ring-opening side reactions, ensuring that the strained cyclopropane motif remains intact. Finally, the synthesis culminates in a Hoffmann degradation reaction, where the amide is treated with sodium hypochlorite and sodium hydroxide to excise the carbonyl group and release the primary amine.  This final transformation is executed with rigorous temperature control, initially cooling the mixture to 0-10°C for reagent addition before heating to 100°C, a protocol that maximizes the conversion efficiency while minimizing the formation of degradation byproducts, ultimately delivering a product of exceptional purity.
This final transformation is executed with rigorous temperature control, initially cooling the mixture to 0-10°C for reagent addition before heating to 100°C, a protocol that maximizes the conversion efficiency while minimizing the formation of degradation byproducts, ultimately delivering a product of exceptional purity.
How to Synthesize Cyclopropylamine Efficiently
Implementing this synthesis route requires a disciplined adherence to the specific thermal and stoichiometric parameters outlined in the patent to ensure reproducibility and safety at scale. The process is designed to be robust, allowing for the recovery of valuable starting materials which contributes to the overall economic viability of the operation. Operators must pay close attention to the layering and separation steps following the initial cyanation, as the efficient recovery of unreacted 1,3-dichloropropane is a key driver for cost reduction. The detailed standardized synthesis steps below provide a comprehensive guide for technical teams looking to adopt this superior methodology for the commercial production of high-purity cyclopropylamine.
- Reflux 1,3-dichloropropane with sodium cyanide solution at 60-100°C to form 4-chlorobutyronitrile, followed by rectification to recover unreacted starting material.
- React the 4-chlorobutyronitrile intermediate with sodium hydroxide solution at 60-100°C to induce cyclization and hydrolysis, yielding cyclopropanecarboxamide.
- Perform Hoffmann degradation on the cyclopropanecarboxamide using sodium hypochlorite and NaOH at 100°C, collecting the fraction boiling at 49-51°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method represents a strategic opportunity to de-risk the sourcing of critical cyclic amine intermediates while simultaneously driving down the total cost of ownership. The shift away from exotic or tightly constrained raw materials like 1,3-propanediol to commodity chemicals like 1,3-dichloropropane fundamentally alters the cost structure of the final product, insulating buyers from the volatility associated with specialty diol markets. Moreover, the simplified reaction conditions reduce the burden on utility infrastructure, meaning that production can be scaled up in existing facilities without requiring capital-intensive retrofits for high-pressure or extreme-temperature capabilities. This flexibility ensures a more resilient supply chain capable of responding rapidly to fluctuations in market demand for agrochemical and pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic superiority of this process is primarily derived from the substitution of expensive precursor materials with widely available, low-cost industrial commodities. By utilizing 1,3-dichloropropane, manufacturers can bypass the costly bromination and activation steps inherent in diol-based routes, effectively stripping out multiple unit operations and their associated labor and energy costs. Additionally, the integrated rectification step allows for the continuous recovery and reuse of unreacted starting materials, creating a closed-loop system that dramatically minimizes raw material waste and further drives down the variable cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved because the key raw material, 1,3-dichloropropane, is produced on a massive global scale for diverse industrial applications, ensuring a consistent and reliable supply stream. Unlike niche precursors that may be subject to production bottlenecks at a single supplier, the commoditized nature of the feedstock means that procurement teams can leverage multiple sources to negotiate better terms and secure long-term contracts. This abundance of supply, coupled with the robustness of the synthetic route which tolerates minor variations in feedstock quality, guarantees a steady flow of intermediates to downstream customers, preventing production stoppages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard aqueous workups and distillation techniques that are easily transferred from pilot plants to multi-tonne commercial reactors. From an environmental perspective, the method offers substantial advantages by reducing the volume of hazardous wastewater generated; the alkaline hydrolysis step is efficient and complete, minimizing the load on effluent treatment plants. Furthermore, the avoidance of heavy metal catalysts or toxic halogenating agents in the later stages of the synthesis simplifies the regulatory compliance landscape, facilitating faster approvals for new manufacturing sites and reducing the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their supply chains. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring that the information provided is grounded in verified scientific results. Understanding these details is crucial for making informed decisions about vendor qualification and process adoption.
Q: What are the primary advantages of using 1,3-dichloropropane over 1,3-propanediol for cyclopropylamine synthesis?
A: The use of 1,3-dichloropropane offers significant economic advantages due to its wide availability and lower cost compared to 1,3-propanediol. Furthermore, the reaction conditions are milder, avoiding the harsh bromination steps required in traditional diol-based routes, which simplifies the operational complexity and reduces waste generation.
Q: How does the novel Hoffmann degradation step impact the purity of the final cyclopropylamine product?
A: The Hoffmann degradation step allows for the precise conversion of the amide intermediate to the amine with high selectivity. By controlling the temperature and utilizing fractional distillation to collect the specific boiling point fraction of 49-51°C, the process effectively removes impurities, resulting in a high-purity final product suitable for sensitive pharmaceutical applications.
Q: Is this synthesis method scalable for industrial production of agrochemical intermediates?
A: Yes, the method is highly scalable. The process utilizes common industrial reagents like sodium cyanide and sodium hypochlorite, and the reaction temperatures (60-100°C) are easily manageable in standard stainless steel reactors. The ability to recover and recycle unreacted 1,3-dichloropropane further enhances its viability for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is not just a technical upgrade but a strategic imperative for maintaining competitiveness in the global fine chemicals market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-volume manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cyclopropylamine meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to delivering not just a product, but a comprehensive solution that enhances your operational efficiency and product quality.
We invite forward-thinking organizations to collaborate with us to leverage this advanced synthesis technology for their specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your bill of materials. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to validate the superior quality and economic advantages of our cyclopropylamine supply chain firsthand.
