Revolutionizing L-tert-leucine Production: A Low-Cost Enzymatic Strategy for Global Pharma Supply Chains
The pharmaceutical industry's relentless pursuit of efficient, green synthesis routes for chiral amino acids has found a significant breakthrough in patent CN102978251A. This document details a highly optimized enzymatic method for producing L-tert-leucine, a critical non-natural alpha-amino acid serving as a pivotal chiral building block for antiviral medications, including HIV protease inhibitors and hepatitis C treatments. Unlike traditional chemical synthesis which often struggles with stereocontrol and environmental impact, this biocatalytic approach leverages a dual-enzyme system comprising Leucine Dehydrogenase (LeuDH) and Formate Dehydrogenase (FDH). The innovation lies not just in the biological agents used, but in the precise engineering of reaction conditions that overcome historical barriers such as substrate inhibition and excessive cofactor consumption. By integrating a strategic fed-batch substrate addition protocol, the method maintains high reaction velocities while minimizing the accumulation of inhibitory by-products, ultimately delivering a product with greater than 98% chemical purity and exceeding 99% optical purity.
For R&D directors and process chemists evaluating potential technology transfers, the significance of this patent extends beyond mere yield improvements; it represents a fundamental shift towards sustainable manufacturing paradigms. The ability to operate at mild temperatures between 35°C and 37°C eliminates the need for energy-intensive heating or cryogenic cooling often associated with organometallic catalysis. Furthermore, the use of ammonium formate as both the nitrogen source and the hydride donor for cofactor regeneration simplifies the reagent profile, reducing the complexity of waste stream management. This alignment with green chemistry principles ensures that the production of high-purity L-tert-leucine can be scaled without compromising environmental compliance, addressing a growing concern among global regulatory bodies and corporate sustainability officers alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of L-tert-leucine has been plagued by inherent inefficiencies that drive up costs and limit supply chain reliability. The most common traditional approach involves the chemical resolution of racemic DL-tert-leucine using lipases or chiral resolving agents. While chemically straightforward, this method suffers from a theoretical yield ceiling of 50%, as half of the starting material (the unwanted D-enantiomer) must be discarded or subjected to costly racemization cycles. Additionally, chemical synthesis routes often rely on heavy metal catalysts or harsh reducing agents like sodium borohydride, which introduce significant safety hazards and generate toxic heavy metal waste that requires expensive removal steps to meet pharmaceutical grade specifications. Previous attempts at biocatalysis also faced substantial hurdles; earlier patents disclosed processes requiring excessively high enzyme loads, sometimes utilizing whole-cell catalysts at concentrations as high as 80% of the reaction volume, which severely hampered reactor productivity and downstream processing efficiency.
The Novel Approach
The methodology described in CN102978251A dismantles these barriers through a sophisticated understanding of enzyme kinetics and process engineering. By implementing a fed-batch strategy where the substrate, trimethylpyruvate, is added in stages rather than all at once, the process effectively mitigates substrate inhibition—a phenomenon where high initial concentrations of the keto acid suppress enzyme activity. This allows the reaction to proceed at optimal rates even as product concentrations build up. Moreover, the optimization of catalyst loading is a standout feature; the total enzyme dosage is reduced to no more than 4% of the substrate weight, and the expensive NAD cofactor is minimized to just 0.2%. This drastic reduction in biocatalyst consumption, coupled with a space-time yield reaching 100 g/d/L, transforms the economic viability of the process, making it a superior alternative for cost reduction in pharmaceutical intermediate manufacturing compared to legacy resolution or chemical synthesis techniques.
Mechanistic Insights into LeuDH and FDH Coupled Catalytic Cycle
The core of this technological advancement lies in the elegant coupling of two distinct enzymatic reactions that drive the thermodynamic equilibrium towards product formation. As illustrated in the reaction scheme below, Leucine Dehydrogenase (LeuDH) catalyzes the reductive amination of trimethylpyruvate in the presence of ammonia and the reduced cofactor NADH to form L-tert-leucine and NAD+. However, since NADH is expensive and unstable in isolation, the process relies on Formate Dehydrogenase (FDH) to continuously regenerate the reduced cofactor. FDH oxidizes ammonium formate into carbon dioxide and ammonia, simultaneously reducing NAD+ back to NADH. This creates a closed catalytic loop where the cofactor acts merely as an electron shuttle, allowing minute quantities of NAD to drive the conversion of large amounts of substrate.
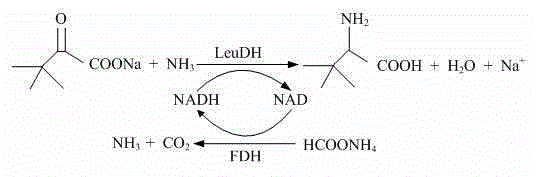
This coupled system is meticulously balanced to prevent the accumulation of inhibitory species. In many biocatalytic processes, the buildup of ammonia or the keto-acid substrate can denature the enzymes or shift the equilibrium unfavorably. The patent specifies the use of ammonium formate, which serves a dual purpose: it provides the necessary ammonia for the amination step while acting as the sacrificial reductant for the FDH cycle. The release of CO2 gas during the FDH step also helps drive the reaction forward by removing a product from the liquid phase, adhering to Le Chatelier's principle. For technical teams, understanding this mechanism is crucial for troubleshooting; maintaining the correct ratio of LeuDH to FDH and ensuring adequate mixing to vent CO2 are key operational parameters that ensure the high optical purity (>99% ee) and chemical yield (>90%) reported in the experimental data.
How to Synthesize L-tert-leucine Efficiently
The synthesis protocol outlined in the patent offers a robust framework for laboratory and pilot-scale production, emphasizing precise control over reaction timing and temperature to maximize efficiency. The process begins with the preparation of an aqueous reaction mixture containing the enzymes and cofactors, equilibrated to a physiological temperature range of 35°C to 37°C. Critical to the success of the operation is the staged addition of the trimethylpyruvate substrate; an initial charge is followed by supplemental feeds at the 2-hour and 4-hour marks. This temporal distribution of substrate prevents the local concentration spikes that typically lead to enzyme inhibition, ensuring a smooth reaction profile over the 24-hour cycle. Detailed standardized operating procedures for scaling this specific fed-batch enzymatic route are provided in the technical guide below.
- Prepare the reaction system by mixing Leucine Dehydrogenase, Formate Dehydrogenase, and NAD coenzyme in an ammonium formate buffer solution at 35-37°C.
- Implement a fed-batch strategy by initially adding trimethylpyruvate substrate, followed by sequential additions at 2 hours and 4 hours to prevent substrate inhibition.
- Monitor reaction progress via HPLC until full conversion (approx. 24 hours), then heat to 60°C, filter, and purify the product using methanol washes.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic process translates directly into enhanced margin protection and supply security. The primary economic driver is the substantial reduction in raw material costs associated with the biocatalysts. By lowering the enzyme loading to less than 4% and the cofactor to 0.2%, the process eliminates the need for purchasing bulk quantities of expensive biological reagents, which historically constituted a major portion of the variable cost in biotransformation. Furthermore, the simplicity of the downstream processing—requiring only heating, filtration through diatomite, and methanol washing—removes the need for complex chromatography columns or solvent-intensive extraction steps often required to remove metal catalysts or resolving agents. This streamlined workflow not only reduces utility consumption but also shortens the overall batch cycle time, increasing the throughput capacity of existing manufacturing infrastructure without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The drastic minimization of enzyme and cofactor usage fundamentally alters the cost structure of L-tert-leucine production. Unlike previous methods that required stoichiometric or near-stoichiometric amounts of resolving agents or high-load whole cells, this catalytic approach ensures that the cost per kilogram of product is significantly lowered. The elimination of heavy metal catalysts also removes the downstream costs associated with scavenging resins and rigorous heavy metal testing, further contributing to overall process economics and reducing the financial burden of quality control operations.
- Enhanced Supply Chain Reliability: The reliance on readily available substrates like trimethylpyruvate and ammonium formate, combined with stable enzyme formulations, mitigates the risk of supply disruptions often seen with specialized chiral reagents. The process operates under ambient pressure and mild temperatures, meaning it can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This flexibility allows for rapid scale-up and the ability to switch production lines quickly in response to market demand fluctuations, ensuring a consistent and reliable supply of this critical antiviral intermediate to downstream drug manufacturers.
- Scalability and Environmental Compliance: From a sustainability perspective, the aqueous nature of the reaction medium and the generation of benign by-products like CO2 and water align perfectly with modern environmental regulations. The absence of toxic organic solvents during the reaction phase and the reduction of waste streams simplify the permitting process for new production facilities. The high space-time yield of 100 g/d/L indicates that smaller reactor volumes can produce the same output as larger, less efficient systems, optimizing facility footprint and energy usage while ensuring that the commercial scale-up of complex pharmaceutical intermediates remains environmentally responsible and compliant with increasingly strict global standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in patent CN102978251A, providing clarity on how this method outperforms legacy technologies in terms of purity, yield, and operational simplicity. Understanding these specifics is vital for technical teams evaluating the feasibility of integrating this biocatalytic platform into their existing manufacturing portfolios.
Q: How does this enzymatic method improve yield compared to traditional resolution?
A: Traditional lipase resolution methods are theoretically limited to a maximum 50% yield because they separate racemic mixtures. This enzymatic reductive amination method converts the prochiral ketone directly into the L-enantiomer, achieving yields greater than 90% with optical purity exceeding 99%.
Q: What are the cost advantages of the catalyst loading in Patent CN102978251A?
A: Previous enzymatic processes often required excessive enzyme loads (up to 80% whole cell concentration) or high coenzyme usage (1% NAD). This optimized process reduces total enzyme loading to no more than 4% of substrate weight and coenzyme to 0.2%, drastically lowering raw material costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes a fed-batch approach to manage substrate inhibition, achieves a high space-time yield of 100 g/d/L, and requires no special pressure equipment, making it ideal for standard stainless steel reactors in commercial facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-tert-leucine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of L-tert-leucine in the synthesis of next-generation antiviral therapeutics. As a leading CDMO partner, we possess the technical expertise to translate the innovative enzymatic pathways described in CN102978251A into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle diverse biocatalytic transformations, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs that ensure every batch exceeds the >98% purity and >99% optical purity benchmarks required by top-tier pharmaceutical clients.
We invite global partners to collaborate with us to optimize their supply chains for chiral amino acids. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this enzymatic route can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our L-tert-leucine batches and to discuss route feasibility assessments for your upcoming projects, ensuring a secure and cost-effective supply of this vital pharmaceutical building block.
