Scalable Synthesis of Rasagiline Intermediates Using Atmospheric Aluminum-Nickel Alloy Reduction Technology
Scalable Synthesis of Rasagiline Intermediates Using Atmospheric Aluminum-Nickel Alloy Reduction Technology
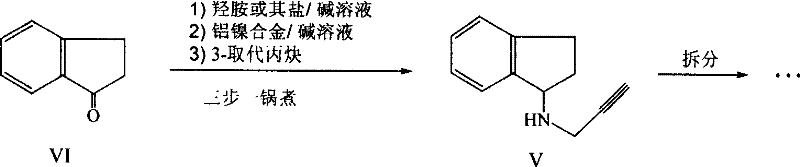
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for producing critical neurological agents. Patent CN101062897B, published in late 2011, introduces a transformative methodology for synthesizing 2,3-dihydro-1H-inden-1-amine, a pivotal intermediate for the anti-Parkinson's drug Rasagiline. This technology replaces hazardous high-pressure hydrogenation with a safer, atmospheric aluminum-nickel alloy reduction, offering a compelling value proposition for manufacturers aiming to optimize their supply chains. By leveraging heterogeneous catalysis in an alkaline medium, this process achieves high conversion rates while circumventing the stringent safety protocols associated with compressed hydrogen gas. For global procurement teams, this represents a shift towards more resilient manufacturing capabilities that are less dependent on specialized high-pressure infrastructure.
Furthermore, the patent elucidates a "one-pot" synthesis strategy that integrates multiple reaction steps—oximation, reduction, and alkylation—into a continuous sequence without intermediate isolation. This approach not only streamlines the operational workflow but also significantly reduces the environmental footprint by minimizing solvent usage and waste generation. As regulatory pressures on pharmaceutical manufacturing intensify, adopting such green chemistry principles becomes a strategic imperative. The following analysis dissects the technical merits of this innovation, providing R&D directors and supply chain heads with the insights needed to evaluate its potential for commercial adoption in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of 2,3-dihydro-1H-inden-1-one oxime to the corresponding amine has relied heavily on catalytic hydrogenation using Raney Nickel under pressure. As documented in prior art such as EP235590, this conventional route necessitates operating conditions of approximately 50 psi hydrogen pressure for extended durations, often exceeding 25 hours. Such requirements mandate the use of specialized autoclaves and rigorous safety monitoring systems, which inflate both capital expenditure and operational complexity. Additionally, alternative chemical reduction methods utilizing titanium tetrachloride and sodium borohydride demand strictly anhydrous conditions, presenting significant challenges for large-scale implementation due to the sensitivity of reagents and the high cost of solvent drying. These legacy processes introduce bottlenecks in production throughput and elevate the risk profile of the manufacturing facility.
The Novel Approach
In stark contrast, the methodology disclosed in CN101062897B utilizes an aluminum-nickel alloy in a basic aqueous-organic solvent system to effect reduction under atmospheric pressure. This innovation eliminates the need for external hydrogen sources and high-pressure vessels, allowing the reaction to proceed in standard glass-lined or stainless steel reactors commonly found in fine chemical plants. The process operates efficiently at moderate temperatures between 50°C and 60°C, utilizing inexpensive reagents like sodium hydroxide and ethanol. By shifting from a gas-phase hydrogenation to a liquid-phase heterogeneous reduction, the technology simplifies the engineering controls required for safe operation. This transition not only lowers the barrier to entry for contract manufacturing organizations but also enhances the overall flexibility of the production line to accommodate varying batch sizes without compromising safety or yield.

Mechanistic Insights into Aluminum-Nickel Alloy Mediated Reduction
The core of this technological advancement lies in the unique reactivity of the aluminum-nickel alloy within an alkaline environment. When introduced to a solution containing sodium or potassium hydroxide, the aluminum component of the alloy undergoes corrosion, reacting with water to generate nascent hydrogen species directly on the nickel surface. This in situ generation of active hydrogen creates a highly reactive microenvironment that facilitates the rapid reduction of the oxime functional group to the primary amine. Unlike traditional catalytic hydrogenation where gas-liquid mass transfer can be a rate-limiting step, this heterogeneous system ensures intimate contact between the reducing agent and the substrate. The nickel acts as a catalyst to activate the hydrogen species, while the aluminum serves as the stoichiometric source of electrons and protons, driving the reaction to completion with high selectivity.
From an impurity control perspective, this mechanism offers distinct advantages over borohydride reductions. The alkaline conditions suppress the formation of certain side products that might arise under acidic or neutral conditions. Furthermore, the use of a water-ethanol solvent system enhances the solubility of the inorganic byproducts, facilitating their removal during the aqueous workup phase. The reaction progress can be easily monitored via thin-layer chromatography (TLC), with typical completion times ranging from 7 to 9 hours depending on the specific batch parameters. The resulting amine can be isolated as a free base or converted directly into a stable salt, such as the hydrochloride, ensuring high purity suitable for subsequent coupling reactions. This robust mechanistic pathway provides R&D teams with a reliable foundation for scaling up the synthesis of complex amine intermediates.
How to Synthesize 2,3-Dihydro-1H-inden-1-amine Efficiently
Implementing this synthesis route requires precise control over reagent addition and temperature management to maximize efficiency and safety. The process begins with the preparation of a homogeneous alkaline solution, followed by the controlled introduction of the alloy to manage the exothermic evolution of hydrogen gas. Detailed operational parameters, including specific molar equivalents and solvent ratios, are critical for reproducing the high yields reported in the patent literature. Operators must adhere to strict batching protocols to prevent runaway reactions while ensuring complete consumption of the starting oxime. The following guide outlines the standardized procedural framework derived from the patent examples, serving as a baseline for process engineers to adapt to their specific manufacturing infrastructure.
- Prepare an alkaline solution by mixing sodium hydroxide or potassium hydroxide with a water-ethanol solvent system, maintaining a volume ratio optimized for solubility and reaction kinetics.
- Introduce 2,3-dihydro-1H-inden-1-one oxime into the reaction vessel and heat the mixture to a controlled temperature range of 50°C to 60°C to initiate activation.
- Add aluminum-nickel alloy powder in batches over a period of 2 to 3 hours to manage exothermic gas evolution, followed by continued stirring until TLC indicates complete conversion to the amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aluminum-nickel alloy reduction technology translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of high-pressure hydrogenation equipment removes a significant capital cost barrier, allowing for production in a wider range of facilities without the need for specialized safety certifications associated with compressed gas handling. This flexibility enhances supply chain resilience by diversifying the pool of potential manufacturing partners who can execute the synthesis. Moreover, the use of commodity chemicals like aluminum-nickel alloy and caustic soda reduces dependency on expensive, specialized reducing agents, stabilizing raw material costs against market volatility. The simplified workflow also shortens the overall production cycle time, enabling faster response to market demand fluctuations for Parkinson's disease medications.
- Cost Reduction in Manufacturing: The transition to atmospheric pressure processing drastically reduces energy consumption associated with compressing hydrogen and maintaining high-pressure systems. By utilizing inexpensive, commercially available aluminum-nickel alloy instead of precious metal catalysts or complex hydride reagents, the direct material cost per kilogram of the intermediate is significantly lowered. Additionally, the ability to perform the reaction in standard reactors minimizes maintenance costs and extends the lifespan of production equipment, contributing to long-term operational savings.
- Enhanced Supply Chain Reliability: Sourcing aluminum-nickel alloy is far more straightforward and secure compared to specialized catalysts that may face supply constraints. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by equipment failure or stringent safety shutdowns. This reliability ensures a consistent flow of high-purity intermediates to downstream API manufacturers, mitigating the risk of stockouts that could impact the availability of finished neurological drugs.
- Scalability and Environmental Compliance: The "one-pot" capability described in the patent allows for the telescoping of multiple steps, which significantly reduces the volume of organic solvents required and the quantity of wastewater generated. This reduction in waste streams simplifies effluent treatment processes and lowers the environmental compliance burden. The process is inherently scalable from pilot plant to multi-ton commercial production without requiring fundamental changes to the reaction engineering, facilitating a smoother technology transfer and faster time-to-market for new generic formulations.
Frequently Asked Questions (FAQ)
Understanding the nuances of this synthesis technology is essential for stakeholders evaluating its integration into existing production portfolios. Common inquiries often revolve around the safety profile of the alloy, the purity of the final product, and the logistical implications of switching from traditional hydrogenation methods. The answers provided below are grounded in the technical data disclosed within the patent, offering clarity on how this method addresses common pain points in pharmaceutical intermediate manufacturing. These insights are designed to assist technical teams in making informed decisions regarding process validation and vendor qualification.
Q: What are the safety advantages of using Al-Ni alloy over Raney Nickel hydrogenation?
A: The Al-Ni alloy method operates at atmospheric pressure, eliminating the need for high-pressure hydrogenation equipment (50 psi+) required by traditional Raney Nickel methods. This significantly reduces capital expenditure on specialized reactors and mitigates explosion risks associated with handling compressed hydrogen gas.
Q: How does the 'One-Pot' method impact production costs?
A: By combining the oximation, reduction, and N-alkylation steps into a single vessel without intermediate isolation, the process drastically reduces solvent consumption, energy usage for drying, and labor hours. It minimizes material loss during transfer and filtration, leading to substantial overall cost savings.
Q: Is the aluminum-nickel alloy reusable or disposable?
A: In this specific reduction protocol, the aluminum-nickel alloy acts as a sacrificial reagent that generates active hydrogen species in situ. While the nickel component remains, the alloy is consumed during the reaction and is typically filtered off as solid waste, requiring careful disposal or recycling protocols rather than regeneration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydro-1H-inden-1-amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe manufacturing processes in the pharmaceutical sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the aluminum-nickel alloy reduction are implemented with the highest standards of quality and safety. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,3-dihydro-1H-inden-1-amine meets the exacting requirements of global regulatory bodies. Our commitment to continuous improvement allows us to offer cost-effective solutions without compromising on the integrity of the supply chain.
We invite you to collaborate with us to leverage these advanced synthetic methodologies for your Rasagiline projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can enhance your competitive advantage in the neurological therapeutics market.
