Advanced Industrial Synthesis of Adefovir: A Cost-Effective Route for Antiviral API Production
The global burden of Hepatitis B virus (HBV) infection remains a critical public health challenge, driving sustained demand for effective antiviral therapeutics such as Adefovir. As a nucleotide analog of adenosine monophosphate, Adefovir serves as a pivotal active pharmaceutical ingredient (API) that inhibits HBV DNA polymerase upon intracellular phosphorylation. However, the commercial viability of this life-saving medication has historically been constrained by complex, hazardous, and cost-prohibitive manufacturing processes. Patent CN102250146A introduces a transformative synthetic methodology that addresses these industrial bottlenecks by utilizing adenine as a starting material through a refined three-step nucleophilic substitution sequence. This technical breakthrough not only enhances reaction selectivity and yield but also fundamentally reshapes the economic landscape of antiviral API production by replacing dangerous reagents with benign, commercially abundant alternatives. For pharmaceutical manufacturers and supply chain strategists, this innovation represents a significant opportunity to optimize cost structures while ensuring a robust, continuous supply of high-purity antiviral agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
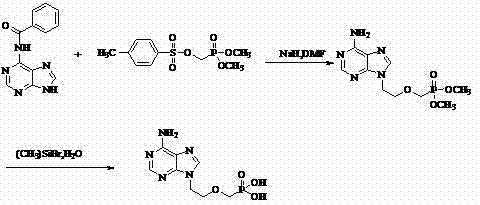
Historically, the industrial synthesis of Adefovir has been plagued by reliance on highly reactive and hazardous reagents that complicate scale-up and inflate operational expenditures. Traditional routes, such as those disclosed in earlier literature, frequently employ sodium hydride as a base and bromotrimethylsilane for deprotection, both of which pose severe safety risks including pyrophoricity and moisture sensitivity. Furthermore, these legacy processes often necessitate rigorous purification steps involving column chromatography and ion exchange resins to isolate the desired product from complex reaction mixtures. The requirement for such labor-intensive purification not only consumes vast quantities of organic solvents but also results in significant product loss, thereby depressing overall yields and increasing the environmental footprint of the manufacturing facility. Additionally, the use of expensive catalysts and difficult-to-handle intermediates creates supply chain vulnerabilities, making the consistent production of cost-effective Adefovir a persistent challenge for generic drug manufacturers.
The Novel Approach
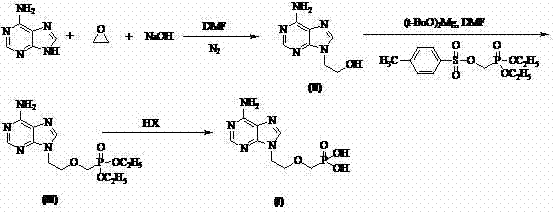
In stark contrast to these cumbersome legacy methods, the patented process outlined in CN102250146A offers a streamlined, industrially viable pathway that prioritizes safety, efficiency, and scalability. The core innovation lies in the strategic substitution of hazardous reagents with safer, more economical alternatives; specifically, ethylene oxide is utilized for the initial alkylation of adenine, and magnesium tert-butoxide replaces sodium hydride for the subsequent phosphonation step. This chemical redesign eliminates the need for column chromatography entirely, relying instead on straightforward solvent washing and recrystallization techniques to achieve high purity levels. By simplifying the workup procedures and utilizing robust reagents like hydroiodic acid for final deprotection, the new route significantly reduces processing time and waste generation. This approach not only lowers the barrier to entry for manufacturers but also ensures a more stable and predictable production cycle, essential for meeting the rigorous quality standards of the global pharmaceutical market.
Mechanistic Insights into Nucleophilic Substitution and Ester Hydrolysis
The success of this synthetic strategy hinges on the precise control of regioselectivity during the initial alkylation of the adenine ring system. In the first step, adenine undergoes nucleophilic attack on ethylene oxide under mild alkaline conditions, preferentially forming the N9-substituted 9-(2-hydroxyethyl)adenine isomer. The use of a specific base and solvent system, such as sodium hydroxide in DMF, facilitates this transformation while minimizing the formation of N3 or N7 isomers, which are common impurities in less optimized processes. This high degree of selectivity is crucial as it simplifies downstream purification and ensures that the subsequent phosphonation reaction proceeds with maximum efficiency. The second step involves the activation of the hydroxyl group via magnesium tert-butoxide, which acts as a mild yet effective base to generate the alkoxide intermediate in situ. This intermediate then attacks diethyl (tosyloxymethyl)phosphonate, installing the critical phosphonate side chain with high fidelity and minimal side reactions.
The final transformation involves the hydrolysis of the diethyl phosphonate ester to yield the free phosphonic acid, a step that traditionally requires harsh conditions. In this novel protocol, hydroiodic acid (HI) is employed as a cleavage agent, offering a superior alternative to bromotrimethylsilane. The mechanism likely involves the protonation of the ethoxy oxygen followed by nucleophilic attack by iodide, leading to the efficient removal of ethyl groups as ethyl iodide. This acid-mediated deprotection is not only cost-effective but also generates fewer toxic byproducts compared to silicon-based reagents. Following the reaction, careful pH adjustment to the range of 2.9 to 3.3 induces the precipitation of the zwitterionic Adefovir product, allowing for isolation via simple filtration. The final recrystallization from water-alcohol systems further refines the crystal lattice, removing trace impurities and ensuring the final API meets the stringent purity specifications required for clinical application.
How to Synthesize Adefovir Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to maximize yield and purity. The process begins with the alkylation of adenine in a nitrogen atmosphere to prevent oxidation, followed by a controlled addition of ethylene oxide gas. Subsequent steps involve the precise dosing of magnesium tert-butoxide and the phosphonate ester in polar aprotic solvents like DMF. The detailed standardized operating procedures, including specific molar ratios, reaction times, and workup protocols necessary for reproducible commercial manufacturing, are outlined in the comprehensive guide below.
- React Adenine with ethylene oxide under alkaline conditions in DMF to form 9-(2-hydroxyethyl)adenine with high regioselectivity.
- Perform nucleophilic substitution using magnesium tert-butoxide and diethyl (tosyloxymethyl)phosphonate to install the phosphonate side chain.
- Hydrolyze the diethyl ester intermediate using hydroiodic acid (HI) followed by pH adjustment and recrystallization to obtain pure Adefovir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced supply security. By eliminating the need for specialized purification equipment such as chromatography columns and ion exchange resins, manufacturers can significantly lower their capital investment and maintenance costs. Furthermore, the substitution of expensive and volatile reagents with commodity chemicals like ethylene oxide and hydroiodic acid creates a more resilient supply chain that is less susceptible to market fluctuations and raw material shortages. This robustness is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of high-cost reagents and the simplification of purification steps. Traditional methods relying on bromotrimethylsilane and column chromatography incur substantial material and labor costs; by replacing these with hydroiodic acid and recrystallization, the overall cost of goods sold is significantly reduced. Additionally, the higher selectivity of the reaction minimizes the formation of byproducts, leading to improved overall yields and reduced waste disposal costs. The ability to recover and recycle solvents like DMF and ethyl acetate further contributes to long-term cost savings, making the production of Adefovir more economically sustainable.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as adenine and ethylene oxide ensures a stable and predictable supply chain. Unlike specialized reagents that may have limited suppliers or long lead times, the inputs for this process are commodity chemicals produced at scale globally. This accessibility reduces the risk of production delays caused by raw material scarcity. Moreover, the milder reaction conditions reduce the likelihood of equipment corrosion or failure, enhancing plant uptime and reliability. For supply chain heads, this means a more dependable source of API that can consistently meet volume requirements without the volatility associated with complex, hazard-prone chemistries.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a cleaner, greener alternative to legacy methods. The elimination of silicon-based waste streams and the reduction in organic solvent usage align with increasingly strict environmental regulations and corporate sustainability goals. The process generates fewer hazardous byproducts, simplifying waste treatment and reducing the environmental footprint of the manufacturing site. This compliance advantage facilitates faster regulatory approvals and reduces the risk of environmental fines or shutdowns. Furthermore, the simplicity of the unit operations—primarily stirring, heating, and filtration—makes the process highly scalable from pilot plant to multi-ton commercial production, ensuring that capacity can be expanded rapidly to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Adefovir synthesis technology. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits and feasibility of the new route for potential partners and licensees. Understanding these details is essential for evaluating the fit of this technology within existing manufacturing portfolios.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: The novel process eliminates the use of pyrophoric sodium hydride and moisture-sensitive bromotrimethylsilane, replacing them with safer magnesium tert-butoxide and hydroiodic acid, significantly reducing operational hazards and fire risks in the plant.
Q: Does this method require expensive purification techniques like column chromatography?
A: No, a key advantage of this patented method is the elimination of column chromatography and ion exchange columns. The product is purified through simple solvent washing and recrystallization, which drastically reduces production costs and solvent consumption.
Q: What is the expected purity profile of Adefovir produced via this route?
A: The process demonstrates excellent selectivity, particularly in the initial alkylation step, yielding intermediates with HPLC purity greater than 95% and a final API product with purity exceeding 99%, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adefovir Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthesis route requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this process, while our rigorous QC labs enforce stringent purity specifications to guarantee product quality. We are committed to delivering Adefovir and its intermediates with the consistency and reliability that top-tier pharmaceutical companies demand.
We invite you to explore how this advanced synthesis technology can optimize your supply chain and reduce your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative approach can become a cornerstone of your antiviral drug portfolio.
