Advanced Industrial Synthesis of Adefovir: Enhancing Purity and Scalability for Antiviral Production
Introduction to Next-Generation Adefovir Manufacturing
The global demand for effective antiviral therapies, particularly for Hepatitis B Virus (HBV), necessitates robust and scalable manufacturing processes for key intermediates like Adefovir. Patent CN102250146A introduces a transformative synthetic methodology that addresses critical bottlenecks in the production of 9-(2-(phosphonomethoxy)ethyl)adenine. This technical breakthrough shifts the paradigm from hazardous, low-yield batch processes to a streamlined, high-efficiency protocol suitable for modern reliable pharmaceutical intermediates supplier operations. By leveraging adenine as a starting material and employing ethylene oxide for side-chain construction, the invention achieves superior regioselectivity and minimizes toxic waste generation. For R&D directors and supply chain leaders, this represents a pivotal opportunity to optimize cost reduction in pharmaceutical intermediates manufacturing while ensuring consistent supply continuity. The following analysis dissects the chemical innovations and commercial implications of this proprietary route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
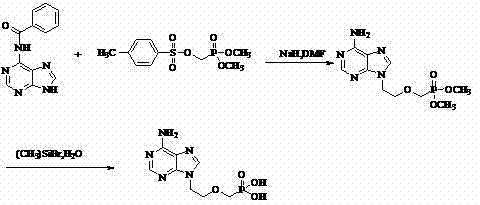
Historically, the synthesis of Adefovir has been plagued by reliance on dangerous reagents and complex purification protocols that hinder industrial viability. Traditional routes, such as those disclosed in earlier literature (e.g., Collect Czech Chem Commun, 1987), often necessitate the protection of amino groups and the use of bromotrimethylsilane and sodium hydride. These reagents not only escalate operational costs due to their high price and specialized handling requirements but also introduce significant safety hazards, including pyrophoric risks and corrosive volatility. Furthermore, legacy methods frequently depend on column chromatography and ion exchange columns for purification, which are notoriously difficult to scale beyond kilogram quantities and result in substantial product loss. The use of sodium hydride also promotes unwanted side reactions, complicating the impurity profile and reducing overall yield.
The Novel Approach
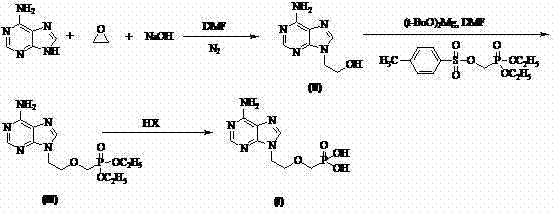
In stark contrast, the methodology outlined in CN102250146A offers a streamlined three-step sequence that bypasses these historical inefficiencies. The process initiates with the direct alkylation of adenine using ethylene oxide under mild alkaline conditions, eliminating the need for amino protection groups entirely. This is followed by a nucleophilic substitution using magnesium tert-butoxide, a safer and more selective base than sodium hydride, to couple the phosphonate side chain. The final deprotection step utilizes aqueous mineral acids (HI, HCl, or HBr) instead of expensive silylating agents. This strategic redesign allows for purification via simple washing and recrystallization, completely removing the need for chromatographic separation. The result is a process that is not only safer and more environmentally benign but also inherently scalable for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ethylene Oxide Alkylation and Acid Hydrolysis
The core chemical innovation lies in the regioselective N9-alkylation of adenine using ethylene oxide. Under controlled alkaline conditions (using NaOH in DMF at 0-50°C), the nucleophilic attack occurs preferentially at the N9 position of the purine ring, minimizing the formation of N3 or N7 isomers which are common impurities in less controlled reactions. This high selectivity is crucial for maintaining a clean impurity profile downstream. The subsequent coupling reaction employs magnesium tert-butoxide to deprotonate the hydroxyl group of the intermediate, generating an alkoxide that attacks the tosyloxymethyl phosphonate electrophile. The choice of magnesium over sodium cations appears to moderate the reactivity, reducing side reactions such as elimination or over-alkylation that typically plague sodium hydride-mediated processes.
Furthermore, the deprotection mechanism represents a significant departure from silyl-based cleavage. By utilizing strong mineral acids like hydroiodic acid (HI) at elevated temperatures (50-100°C), the diethyl phosphonate esters are hydrolyzed efficiently to the free phosphonic acid. This acid-mediated cleavage is robust and avoids the generation of silicon-containing waste streams, which can be difficult to remove and dispose of in compliance with environmental regulations. The final crystallization step, triggered by adjusting the pH to the isoelectric point (pH 2.9-3.3), ensures that the final high-purity Adefovir precipitates with exceptional clarity, effectively excluding organic impurities and residual salts without the need for extensive solvent exchanges.
How to Synthesize Adefovir Efficiently
The synthesis of Adefovir via this patented route involves a logical progression of alkylation, phosphorylation, and hydrolysis steps that are optimized for industrial throughput. The process begins with the preparation of 9-(2-hydroxyethyl)adenine, followed by coupling with a phosphonate building block, and concludes with acid hydrolysis to reveal the active phosphonic acid moiety. This sequence is designed to maximize yield at each stage while minimizing the accumulation of difficult-to-remove byproducts. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical control points for successful execution.
- React adenine with ethylene oxide under alkaline conditions (NaOH) in DMF at 0-50°C to form 9-(2-hydroxyethyl)adenine with high regioselectivity.
- Perform nucleophilic substitution by reacting the hydroxy-intermediate with diethyl (tosyloxymethyl)phosphonate using magnesium tert-butoxide as the base.
- Hydrolyze the diethyl ester intermediate using aqueous HI, HCl, or HBr at 20-120°C, followed by pH adjustment and recrystallization to obtain pure Adefovir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates directly into tangible operational efficiencies and risk mitigation. The elimination of chromatography is perhaps the most significant economic driver, as it removes a major bottleneck in production throughput and drastically reduces solvent consumption. Additionally, the substitution of expensive and hazardous reagents like bromotrimethylsilane and sodium hydride with commodity chemicals like ethylene oxide and mineral acids leads to a substantial reduction in raw material costs. This shift also simplifies the regulatory burden associated with handling and transporting dangerous goods, thereby enhancing supply chain resilience.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by removing the need for column chromatography and ion exchange resins, which are capital-intensive and slow. By replacing high-cost specialty reagents with bulk commodity chemicals, the variable cost per kilogram is drastically lowered. The high yields reported in the patent embodiments further contribute to cost efficiency by maximizing the output from each batch of starting material.
- Enhanced Supply Chain Reliability: Reliance on readily available starting materials such as adenine and ethylene oxide ensures a stable supply base, reducing the risk of shortages associated with exotic catalysts. The simplified purification workflow shortens the overall production cycle time, allowing for faster turnaround and more responsive inventory management. This reliability is critical for maintaining continuous production schedules in the competitive antiviral market.
- Scalability and Environmental Compliance: The absence of pyrophoric reagents and the use of standard aqueous workups make this process highly amenable to large-scale reactor operations. The reduction in hazardous waste generation, particularly silicon-based byproducts and spent chromatography media, aligns with increasingly stringent environmental regulations. This green chemistry approach not only lowers disposal costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Adefovir synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical feasibility for potential partners. Understanding these details is essential for evaluating the fit of this technology within your existing manufacturing infrastructure.
Q: How does this new synthesis method improve safety compared to conventional routes?
A: The novel process eliminates the use of sodium hydride, a pyrophoric and hazardous reagent, replacing it with magnesium tert-butoxide. Furthermore, it avoids bromotrimethylsilane, a volatile and corrosive deprotecting agent, utilizing safer mineral acids like hydroiodic acid instead.
Q: Does this method require column chromatography for purification?
A: No, a significant advantage of this patented method is that it eliminates the need for column chromatography and ion exchange columns. Purification is achieved through efficient washing, solvent extraction, and recrystallization, which drastically simplifies industrial scale-up.
Q: What is the expected purity of the final Adefovir product using this route?
A: The process is designed to achieve high purity levels, with experimental data indicating HPLC purity greater than 99% for the final API intermediate, ensuring it meets stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adefovir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the production of life-saving antiviral agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Adefovir meets the exacting standards required by global regulatory bodies. Our capability to implement the advanced chemistry described in CN102250146A positions us as a strategic partner for long-term supply security.
We invite you to engage with our technical team to explore how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume needs. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
