Advanced Reverse-Phase Purification Technology for High-Purity Adefovir Dipivoxil Manufacturing
The pharmaceutical industry continuously seeks robust purification strategies to ensure the safety and efficacy of antiviral agents, particularly for chronic conditions like Hepatitis B. Patent CN102143967A introduces a transformative approach to the purification of Adefovir Dipivoxil, a critical nucleotide reverse transcriptase inhibitor. Unlike traditional methods that struggle with scalability and impurity profiles, this invention utilizes a sophisticated reverse-phase column chromatography technique. By dissolving the crude reaction mixture in an aqueous or water-containing solvent system and adjusting the pH to an acidic range of 0.1 to 5.0, the process facilitates the selective retention and elution of the target molecule. This methodology not only achieves a remarkable purity level exceeding 99% but also streamlines the production of the amorphous form, which is preferred for its enhanced bioavailability. For global supply chains, this represents a significant leap forward in manufacturing reliability, offering a pathway to produce high-purity pharmaceutical intermediates without the bottlenecks associated with conventional crystallization or normal-phase chromatography.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Adefovir Dipivoxil has been fraught with technical challenges that hinder large-scale commercial viability. Prior art, such as the methods disclosed in U.S. Patent No. 5,663,159, relied heavily on normal-phase column technology using silica gel as the stationary phase. While effective on a small laboratory scale, these systems suffer from severe diffusion effects as the purification capacity increases, leading to a drastic reduction in separation efficiency. Furthermore, alternative crystallization methods described in Korean patents necessitate the use of substantial volumes of expensive organic solvents like n-butyl ether. These processes often require additional, labor-intensive filtration steps to remove salt by-products, such as triethylamine hydrochloride, introducing multiple points of failure and yield loss. The reliance on crystallization also poses a risk of producing inconsistent purity levels, and since the final product is often crystalline, an extra dissolution step is mandatory to achieve the desired amorphous state for pharmaceutical formulation, thereby inflating both time and operational costs.
The Novel Approach
The innovative method detailed in the patent data fundamentally shifts the paradigm by employing reverse-phase chromatography, specifically utilizing C1 to C18 alkyl-packed columns, with a strong preference for C18 octadecyl silica. This approach leverages the hydrophobic interactions between the stationary phase and the analyte in an aqueous environment, providing superior resolution even at higher loading capacities. By carefully controlling the pH of the mobile phase between 1.0 and 3.5 using acids like hydrochloric acid or methanesulfonic acid, the process ensures that the Adefovir Dipivoxil remains in a soluble salt or complex form during the loading phase, while impurities are effectively washed away or retained differently. Following elution, the pH is adjusted to a neutral or slightly basic range (2.5 to 10) to facilitate extraction into an organic solvent like dichloromethane or isopropyl acetate. This seamless transition from purification to isolation allows for the direct recovery of high-purity amorphous solids, bypassing the need for energy-intensive recrystallization and ensuring a consistent quality profile suitable for immediate downstream drug manufacturing.
Mechanistic Insights into Reverse-Phase Chromatographic Separation
The core of this purification success lies in the precise manipulation of physicochemical properties within the reverse-phase system. When the impure Adefovir Dipivoxil is introduced into the acidic aqueous environment, it forms salts or complexes that exhibit distinct polarity characteristics compared to the synthesis by-products. The C18 stationary phase acts as a non-polar barrier, retaining the target molecule based on its hydrophobic pivaloyloxy groups while allowing more polar impurities to pass through or be eluted with specific solvent gradients. The separation efficiency is further enhanced by the specific pH control; maintaining the pH between 0.1 and 5.0 ensures the protonation state of the adenine moiety and the phosphonate group optimizes interaction with the stationary phase. This selectivity is crucial because the synthesis of Adefovir Dipivoxil often generates structurally similar impurities, such as mono-esters or hydrolyzed derivatives, which are difficult to separate via standard precipitation.
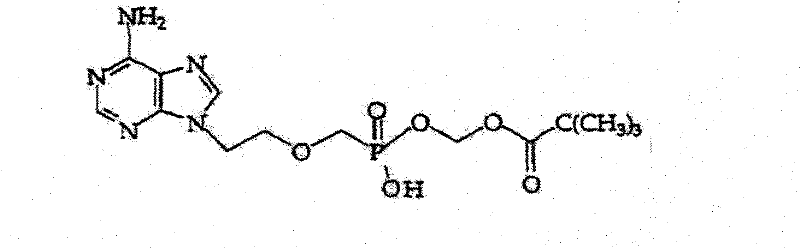
As illustrated by the structural complexity of potential by-products, such as the phosphonate derivatives shown in the patent data, the ability to distinguish between the target diester and its mono-ester or hydrolyzed counterparts is vital for regulatory compliance. The reverse-phase mechanism effectively resolves these close analogs by exploiting subtle differences in their hydrophobicity and ionization states. Once the target compound is eluted, the subsequent addition of a base triggers a phase transfer. By raising the pH to the 2.5 to 10 range, the Adefovir Dipivoxil converts back to its free base or neutral form, drastically increasing its solubility in organic solvents like dichloromethane. This pH-switch extraction strategy not only concentrates the product but also serves as a secondary purification step, leaving behind any remaining water-soluble inorganic salts or highly polar degradation products in the aqueous layer, thus guaranteeing the final amorphous solid meets the stringent high-purity pharmaceutical intermediate specifications required by global health authorities.
How to Synthesize High-Purity Adefovir Dipivoxil Efficiently
Implementing this purification protocol requires strict adherence to the defined parameters to ensure reproducibility and optimal yield. The process begins with the preparation of the crude feedstock, which is dissolved in a water-miscible organic solvent system adjusted to a specific acidic pH. This solution is then loaded onto a pre-equilibrated reverse-phase column, where the flow rate and mobile phase composition are critical for maximizing resolution. The eluate is collected and subjected to a controlled basification and extraction sequence, followed by solvent removal under reduced pressure to yield the final amorphous powder. The detailed standardized synthesis steps for implementing this reverse-phase purification workflow are outlined below.
- Dissolve impure Adefovir Dipivoxil containing synthesis by-products in water or a water-containing mixed solvent adjusted to pH 0.1-5.0.
- Pass the acidic aqueous solution through a reverse-phase column packed with C1-C18 alkyl groups, preferably C18 octadecyl silica.
- Elute the purified product, adjust the pH to 2.5-10 using a base, extract with an organic solvent like dichloromethane, and concentrate to obtain amorphous solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this reverse-phase purification technology translates into tangible operational improvements and risk mitigation. Traditional purification routes often involve volatile organic solvents and complex multi-step crystallizations that are sensitive to environmental fluctuations, leading to batch-to-batch variability. By contrast, the aqueous-based reverse-phase method offers a more robust and controllable process environment. The elimination of expensive crystallization solvents like n-butyl ether significantly reduces raw material costs and simplifies solvent recovery systems. Furthermore, the direct production of the amorphous form removes an entire unit operation from the manufacturing line, reducing the overall processing time and energy consumption. This streamlined workflow enhances the agility of the supply chain, allowing manufacturers to respond more quickly to market demands for antiviral intermediates without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this method are driven by the substitution of costly organic solvents with more economical aqueous systems and the reduction of unit operations. By eliminating the need for large volumes of n-butyl ether and the associated filtration steps for salt removal, the overall cost of goods sold is substantially decreased. Additionally, the higher purification efficiency means less product is lost to mother liquors or discarded fractions, improving the overall mass balance and yield of the facility. The ability to produce the final amorphous form directly also saves the costs associated with redissolving crystalline material, creating a leaner and more cost-effective manufacturing process for cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of purification processes that rely on specific crystal forms or hard-to-source reagents. This reverse-phase method relies on widely available C18 silica packing materials and common acids and bases, reducing the risk of raw material shortages. The robustness of the chromatographic separation ensures that even if the crude input varies slightly in impurity profile, the final output remains within specification, providing a buffer against upstream synthesis variations. This consistency is critical for maintaining long-term contracts with pharmaceutical clients who require guaranteed purity levels, thereby strengthening the reliability of the reliable pharmaceutical intermediate supplier relationship.
- Scalability and Environmental Compliance: Scaling chromatographic processes is generally more predictable than scaling crystallization, which can be sensitive to mixing and cooling rates. The reverse-phase column approach can be scaled from laboratory to commercial production with greater confidence, facilitating the commercial scale-up of complex pharmaceutical intermediates. Moreover, the reduced use of chlorinated and ether solvents aligns better with modern environmental, health, and safety (EHS) regulations. The aqueous waste streams generated are easier to treat compared to complex organic solvent mixtures, lowering the environmental footprint and disposal costs, which is increasingly important for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this purification method is essential for stakeholders evaluating its integration into their supply chain. The following questions address common inquiries regarding the mechanism, purity outcomes, and physical form of the product. These answers are derived directly from the technical disclosures within the patent documentation to provide accurate and actionable insights for decision-makers.
Q: What is the primary advantage of reverse-phase purification over normal-phase for Adefovir Dipivoxil?
A: Reverse-phase purification eliminates the diffusion effects seen in normal-phase silica columns at high capacities, allowing for consistent >99% purity suitable for mass production without expensive crystallization solvents.
Q: How does this method handle synthesis by-products?
A: The method effectively separates specific synthesis by-products (such as mono-esters or hydrolyzed derivatives) by leveraging pH-dependent solubility differences and hydrophobic interactions on the C18 stationary phase.
Q: Is the final product crystalline or amorphous?
A: The process directly yields high-purity amorphous Adefovir Dipivoxil, which offers superior dissolution rates and bioavailability compared to crystalline forms, removing the need for additional dissolution steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adefovir Dipivoxil Supplier
The technical advancements described in patent CN102143967A highlight the critical importance of advanced purification technologies in modern pharmaceutical manufacturing. At NINGBO INNO PHARMCHEM, we recognize that delivering high-quality antiviral intermediates requires more than just standard synthesis; it demands expertise in complex downstream processing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art chromatography systems and rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of Adefovir Dipivoxil we produce adheres to the highest international standards for safety and efficacy.
We invite pharmaceutical companies and procurement leaders to collaborate with us to leverage this superior purification technology for their projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
