Scalable Synthesis of Ticagrelor Key Intermediate A for Global API Production
Scalable Synthesis of Ticagrelor Key Intermediate A for Global API Production
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the demand for more efficient manufacturing processes for critical active pharmaceutical ingredients (APIs). A pivotal development in this domain is detailed in patent CN111087379B, which discloses a highly optimized preparation method for Ticagrelor Key Intermediate A. Ticagrelor, a potent P2Y12 receptor inhibitor, represents a cornerstone in modern antiplatelet therapy, offering superior efficacy compared to earlier generations of drugs. The structural complexity of this molecule necessitates precise synthetic strategies to ensure both economic viability and supply chain stability. 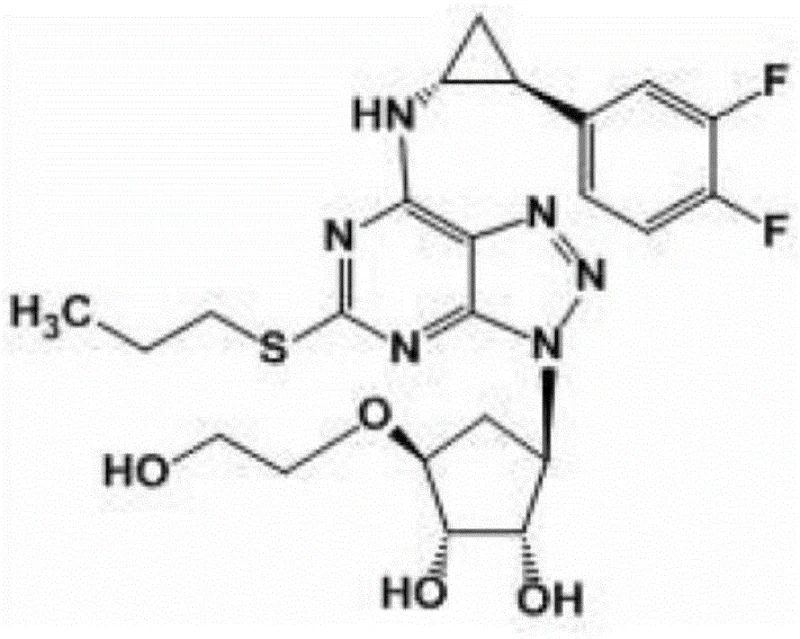 This patent introduces a streamlined approach that addresses the historical bottlenecks of long reaction sequences and low overall yields, positioning it as a vital technology for any reliable pharmaceutical intermediate supplier aiming to support the global demand for anticoagulant medications.
This patent introduces a streamlined approach that addresses the historical bottlenecks of long reaction sequences and low overall yields, positioning it as a vital technology for any reliable pharmaceutical intermediate supplier aiming to support the global demand for anticoagulant medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the cyclopentane core required for Ticagrelor has been fraught with inefficiencies that hinder cost reduction in API manufacturing. Prior art, including literature from Bioorganic & Medicinal Chemistry Letters (2012) and various patents like WO2013092900, typically relies on multi-step sequences involving extensive protecting group manipulations. 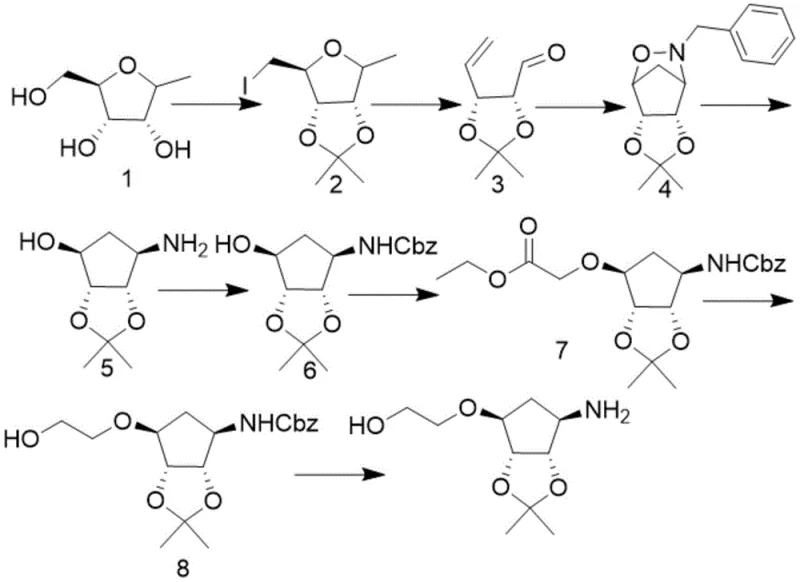 These conventional pathways often necessitate the installation and subsequent removal of bulky protecting groups, such as Cbz (carboxybenzyl), which adds significant time, material cost, and waste generation to the process. Furthermore, the cumulative yield losses across ten or more steps result in a final output that is economically unsustainable for large-scale production. The reliance on harsh conditions or expensive catalysts in these older routes further exacerbates the operational expenditure, making the final intermediate prohibitively expensive for generic drug manufacturers seeking to enter the market with competitive pricing structures.
These conventional pathways often necessitate the installation and subsequent removal of bulky protecting groups, such as Cbz (carboxybenzyl), which adds significant time, material cost, and waste generation to the process. Furthermore, the cumulative yield losses across ten or more steps result in a final output that is economically unsustainable for large-scale production. The reliance on harsh conditions or expensive catalysts in these older routes further exacerbates the operational expenditure, making the final intermediate prohibitively expensive for generic drug manufacturers seeking to enter the market with competitive pricing structures.
The Novel Approach
In stark contrast, the methodology described in CN111087379B offers a paradigm shift by condensing the synthesis into a concise three-step sequence starting from Intermediate 4. This innovative route bypasses the cumbersome protection-deprotection cycles entirely, leveraging a direct reductive cleavage strategy that preserves the stereochemical integrity of the molecule. By eliminating the need for CBZ protection after deprotection, the process drastically simplifies the workflow, reducing the number of unit operations and solvent exchanges required. This not only accelerates the timeline from raw material to finished intermediate but also significantly lowers the environmental footprint by minimizing solvent consumption and chemical waste. For procurement managers, this translates to a more robust supply chain with fewer points of failure, ensuring consistent availability of high-purity intermediates essential for downstream API synthesis.
Mechanistic Insights into Zinc-Mediated Reductive Cleavage
The cornerstone of this improved synthesis lies in the first step: the chemoselective reductive cleavage of the isoxazoline ring system. As illustrated in the reaction scheme, Intermediate 4 is treated with zinc powder in the presence of glacial acetic acid within a tetrahydrofuran (THF) medium. 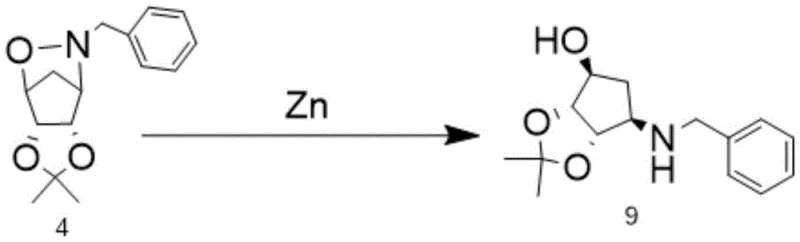 This transformation is critical as it opens the N-O bond to reveal the free amine functionality required for subsequent coupling, all while maintaining the delicate stereochemistry at the cyclopentane ring junctions. The reaction is conducted under mild thermal conditions, initially controlling the temperature between 5-10°C during acid addition to manage exothermicity, followed by stirring at 20-25°C for 24 hours. This gentle approach prevents epimerization or degradation of the sensitive chiral centers, ensuring that the resulting Intermediate 9 possesses the high optical purity necessary for biological activity. The use of zinc, a cheap and abundant metal, coupled with acetic acid, provides a safe and scalable alternative to more hazardous reducing agents like lithium aluminum hydride.
This transformation is critical as it opens the N-O bond to reveal the free amine functionality required for subsequent coupling, all while maintaining the delicate stereochemistry at the cyclopentane ring junctions. The reaction is conducted under mild thermal conditions, initially controlling the temperature between 5-10°C during acid addition to manage exothermicity, followed by stirring at 20-25°C for 24 hours. This gentle approach prevents epimerization or degradation of the sensitive chiral centers, ensuring that the resulting Intermediate 9 possesses the high optical purity necessary for biological activity. The use of zinc, a cheap and abundant metal, coupled with acetic acid, provides a safe and scalable alternative to more hazardous reducing agents like lithium aluminum hydride.
Following the reduction, the process flows seamlessly into an acid-catalyzed etherification and a final hydrogenation. The etherification step utilizes ethylene glycol and a solid acid catalyst (Nafion-H resin) to install the hydroxyethoxy side chain. This heterogeneous catalysis allows for easy removal of the catalyst via simple filtration, avoiding the contamination issues often associated with homogeneous acids. Finally, the benzyl group is removed via catalytic hydrogenation using palladium on carbon (Pd/C). This step is executed at moderate temperatures (50-60°C) and low hydrogen pressure (0.1 MPa), conditions that are easily manageable in standard industrial hydrogenation reactors. The combination of these mechanistic steps ensures that impurities are kept to a minimum, with the final product achieving GC purity levels of 99.5%, thereby meeting the stringent quality specifications demanded by regulatory bodies for cardiovascular drug substances.
How to Synthesize Ticagrelor Key Intermediate A Efficiently
The execution of this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process begins with the preparation of Intermediate 9, where the stoichiometry of zinc and acetic acid must be precisely controlled to ensure complete reduction without over-reduction or side reactions. Following isolation, the etherification step demands rigorous moisture control to drive the equilibrium towards the desired ether product, utilizing toluene as an azeotropic agent to remove water. The final hydrogenation step requires thorough purging of the reactor with nitrogen to ensure safety before introducing hydrogen gas. For R&D teams looking to implement this technology, the detailed standardized synthetic steps are provided below to facilitate immediate technology transfer and process validation.
- Perform reductive cleavage of the N-O bond in intermediate 4 using zinc powder and glacial acetic acid in THF at 20-25°C for 24 hours to obtain intermediate 9.
- Conduct etherification by reacting intermediate 9 with ethylene glycol in toluene using Nafion-H resin catalyst under reflux at 60°C for 5 hours to yield intermediate 10.
- Execute catalytic hydrogenation of intermediate 10 using Pd/C in absolute ethanol at 50-60°C under 0.1MPa hydrogen pressure to afford the final key intermediate A.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this three-step synthesis offers profound advantages for supply chain resilience and cost management. By reducing the number of synthetic steps from over ten to merely three, the manufacturing lead time is drastically compressed, allowing for faster response to market fluctuations and urgent API demands. The elimination of complex protecting group chemistry not only saves on reagent costs but also reduces the volume of hazardous waste generated, aligning with increasingly strict environmental regulations and sustainability goals. This streamlined approach enhances the overall reliability of the supply chain, as fewer processing steps mean fewer opportunities for batch failures or quality deviations that could disrupt production schedules.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant reduction in material and operational inputs. By omitting the CBZ protection and deprotection stages, manufacturers save substantially on the cost of protecting group reagents, additional solvents, and the energy required for extra heating and cooling cycles. Furthermore, the use of inexpensive reagents like zinc powder and ethylene glycol, combined with recoverable catalysts like Nafion-H resin, ensures that the variable cost per kilogram of the intermediate is minimized. This economic efficiency allows for competitive pricing strategies without compromising on the quality or purity of the final pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain security. The reagents employed are commodity chemicals with stable global availability, reducing the risk of raw material shortages that often plague specialized synthetic pathways. Additionally, the simplified workup procedures—primarily involving filtration, extraction, and crystallization—are easily scalable and less prone to technical difficulties during scale-up. This reliability ensures a consistent flow of high-quality intermediates to API manufacturers, mitigating the risk of production stoppages and ensuring continuity of supply for critical cardiovascular medications.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard equipment and safe operating conditions. The avoidance of cryogenic temperatures or high-pressure reactors (beyond standard hydrogenation) lowers the capital expenditure required for facility adaptation. Moreover, the reduction in solvent usage and chemical waste generation supports greener manufacturing practices. The ability to recycle solvents like toluene and ethyl acetate further enhances the environmental profile of the process, making it an attractive option for companies committed to sustainable chemical manufacturing and regulatory compliance.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common inquiries regarding yield, purity, and scalability, drawing directly from the experimental data provided in the patent documentation. These insights are intended to clarify the practical benefits of this method for technical decision-makers.
Q: What is the overall yield and purity of the new synthesis route?
A: The optimized process achieves high individual step yields: 92% for the zinc reduction step (Intermediate 9), 85% for the etherification step (Intermediate 10), and 98% for the final hydrogenation. The final product demonstrates exceptional purity, with HPLC analysis confirming levels exceeding 99.5%.
Q: How does this method improve upon conventional synthesis routes?
A: Unlike traditional methods requiring lengthy protection-deprotection sequences (such as CBZ protection), this novel route utilizes intermediate 4 as a starting material and achieves the target in only three steps. This significantly shortens the production cycle and eliminates complex purification stages associated with protecting group manipulation.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It employs robust, commercially available reagents like zinc powder and ethylene glycol, and utilizes standard unit operations such as filtration and distillation. The mild reaction conditions (e.g., 60°C reflux) and safe handling profiles make it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Intermediate A Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our team of expert chemists has extensively analyzed the pathway described in CN111087379B and is fully equipped to execute this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are supported by rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of Ticagrelor Intermediate A meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your production volumes. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
