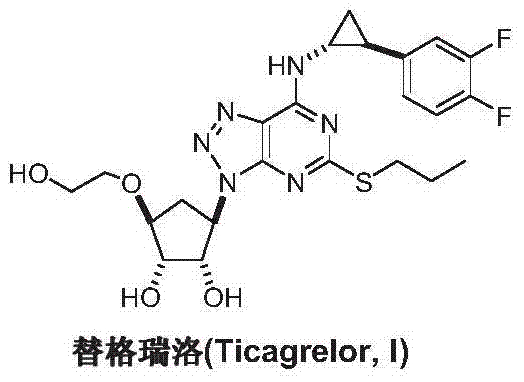
Advanced Green Synthesis of Ticagrelor Intermediate A for Commercial Scale-Up
Introduction to Next-Generation Ticagrelor Intermediate Manufacturing
The global demand for potent antiplatelet agents continues to surge, driven by the increasing prevalence of acute coronary syndromes. At the forefront of this therapeutic class is Ticagrelor, a reversible P2Y12 receptor antagonist that has revolutionized cardiovascular care since its approval by the FDA and EMEA. However, the complex molecular architecture of Ticagrelor presents significant challenges for supply chain stability and cost management. As detailed in patent CN103772295A, a breakthrough preparation method for the critical building block, 4,6-dichloro-2-(propylthio)-5-aminopyrimidine (Intermediate A), offers a transformative solution for the pharmaceutical industry. This innovative synthetic route bypasses traditional hazardous chemistries, replacing them with a streamlined, green chemistry approach that ensures robust supply continuity.
The strategic importance of Intermediate A cannot be overstated, as it serves as the foundational pyrimidine core upon which the entire drug molecule is constructed. Conventional manufacturing pathways have long been plagued by low overall yields and severe environmental burdens associated with diazotization processes. The technology disclosed in this patent represents a paradigm shift, utilizing a protected malonate cyclization strategy that not only enhances safety profiles but also drastically improves atom economy. For procurement leaders and R&D directors alike, adopting this methodology signifies a move towards more sustainable and economically viable API production. By leveraging this advanced chemistry, manufacturers can secure a reliable source of high-purity intermediates while mitigating the regulatory risks associated with older, more toxic synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the pyrimidine core for Ticagrelor has relied heavily on two primary strategies, both of which suffer from inherent inefficiencies and safety liabilities. The first conventional method, documented in various prior art patents, initiates with thiobarbituric acid and proceeds through a diazotization condensation with p-toluidine, followed by chlorination and catalytic reduction. This pathway is fundamentally flawed due to the generation of unstable diazonium intermediates, which pose explosion hazards and require stringent temperature controls that complicate scale-up. Furthermore, the subsequent reduction of the azo-compound often leads to over-reduction or incomplete conversion, resulting in difficult-to-remove impurities that compromise the quality of the final active pharmaceutical ingredient.
The second traditional approach involves nitration of the pyrimidine ring followed by nitro-reduction. While this avoids diazonium salts, it introduces equally severe challenges, particularly regarding regioselectivity and side reactions. The harsh conditions required for nitration can degrade the sensitive sulfur-containing moieties, and the subsequent hydrogenation step carries a high risk of dehalogenation, where the crucial chlorine atoms at the 4 and 6 positions are inadvertently removed. These side reactions not only lower the overall yield but also create a complex impurity profile that necessitates expensive and time-consuming purification steps. For a reliable pharmaceutical intermediate supplier, these unpredictabilities translate into inconsistent batch quality and extended lead times, ultimately jeopardizing the security of the drug supply chain.
The Novel Approach
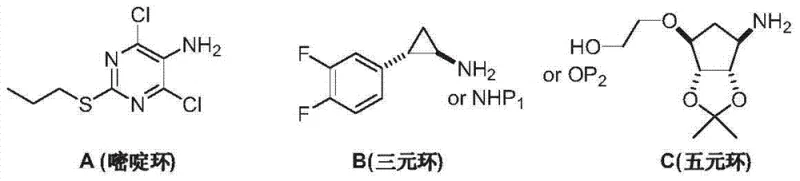
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a constructive build-up strategy starting from 2-protected amino-1,3-dialkyl malonate. This method elegantly constructs the pyrimidine ring through a cyclization reaction with thiourea, establishing the core scaffold with exceptional precision before introducing the side chains. By protecting the amino group early in the synthesis, the process prevents unwanted side reactions during the subsequent alkylation and chlorination steps, ensuring that the nitrogen functionality remains intact until the final deprotection stage. This logical progression eliminates the need for hazardous diazotization or aggressive nitration entirely, marking a significant advancement in process safety and environmental compliance.
Moreover, this new route offers superior control over the introduction of the propylthio group at the 2-position. Through a controlled S-alkylation reaction using halogenated propane, the process achieves high regioselectivity without affecting the protected amine or the carbonyl groups. The subsequent chlorination using phosphorus oxychloride proceeds smoothly on the dihydroxy intermediate, facilitated by the electron-withdrawing nature of the protected ring system. This sequence results in a much cleaner reaction profile, minimizing the formation of by-products and simplifying the downstream isolation procedures. For companies seeking cost reduction in API manufacturing, this streamlined workflow reduces solvent consumption, waste generation, and processing time, delivering a clear competitive advantage in the marketplace.
Mechanistic Insights into Protected Malonate Cyclization and Functionalization
The cornerstone of this innovative synthesis is the initial cyclization reaction between the protected malonate derivative and thiourea. Mechanistically, this transformation is driven by the nucleophilic attack of the thiourea nitrogen on the ester carbonyls of the malonate, facilitated by a strong base such as sodium methoxide. The reaction proceeds through a tetrahedral intermediate which collapses to eliminate alcohol, eventually forming the six-membered 2-thio-sodium barbiturate ring. The presence of the protecting group (such as acetyl or benzyloxycarbonyl) on the amino moiety is critical here; it modulates the electronic density of the nitrogen, preventing it from participating in the cyclization prematurely while remaining stable under the basic reaction conditions. This careful balancing of reactivity ensures that the ring closes exclusively to form the desired pyrimidine-2-thione scaffold with high fidelity.
Following the formation of the barbiturate core, the process moves to the S-alkylation step, which is pivotal for installing the propylthio side chain. In this phase, the sulfur atom at the 2-position acts as a potent nucleophile, attacking the electrophilic carbon of the alkyl halide (e.g., n-propyl bromide). The use of a mixed solvent system, typically comprising water and an organic co-solvent like methanol or acetonitrile, is essential to solubilize both the ionic sodium salt of the barbiturate and the organic alkyl halide. This biphasic-like environment enhances the reaction kinetics, allowing the alkylation to proceed at room temperature with excellent conversion rates. The subsequent chlorination step replaces the hydroxyl groups at the 4 and 6 positions with chlorine atoms via an activation mechanism involving phosphorus oxychloride, creating the highly reactive dichloro-pyrimidine intermediate ready for final coupling.
How to Synthesize 4,6-Dichloro-2-(propylthio)-5-aminopyrimidine Efficiently
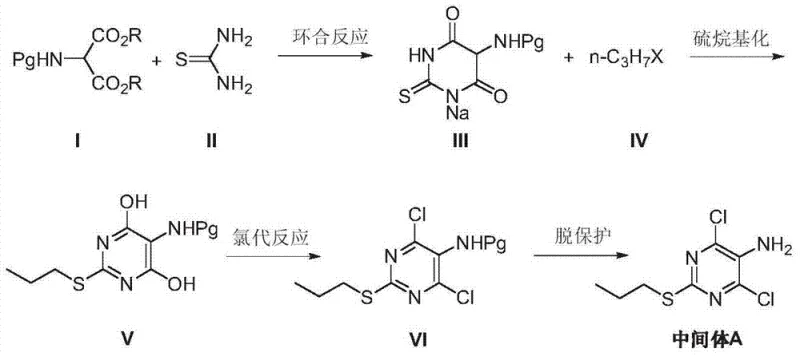
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity at every stage. The process begins with the cyclization of the protected malonate and thiourea under reflux conditions, followed by a carefully monitored S-alkylation at ambient temperatures to prevent over-alkylation. The chlorination step demands strict temperature control, typically initiated under ice-bath conditions to manage the exotherm before heating to drive the reaction to completion. Finally, the deprotection step must be tailored to the specific protecting group used; for instance, acetyl groups are removed via alkaline hydrolysis, while Cbz groups require catalytic hydrogenation or zinc reduction. The detailed standardized synthetic steps see the guide below.
- Perform a cyclization reaction between 2-protected amino-1,3-dialkyl malonate and thiourea using sodium methoxide to generate 5-protected amino-2-thio-sodium barbiturate.
- Conduct an S-alkylation reaction of the sodium barbiturate intermediate with a halogenated propane (such as n-propyl bromide) in a mixed solvent system to introduce the propylthio group.
- Execute a chlorination reaction using phosphorus oxychloride to replace hydroxyl groups with chlorine atoms, followed by a final deprotection step via hydrolysis or reduction to yield the target amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers profound strategic benefits that extend far beyond simple chemical efficiency. By eliminating the reliance on hazardous diazotization and nitration reagents, the process significantly reduces the regulatory burden and insurance costs associated with handling explosive or toxic materials. This translates directly into a more resilient supply chain, as the manufacturing facilities do not require specialized containment infrastructure for high-risk reactions. Furthermore, the use of commodity starting materials like diethyl malonate derivatives and thiourea ensures that raw material availability is never a bottleneck, providing a stable foundation for long-term production planning and inventory management.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by its high step-wise efficiency and simplified workup procedures. Traditional routes often suffer from cumulative yield losses due to multiple purification steps and side reactions; in contrast, this method achieves high conversion rates in each step, minimizing the loss of valuable intermediates. The elimination of expensive catalysts and the reduction in solvent usage for purification further drive down the cost of goods sold (COGS). Additionally, the avoidance of heavy metal catalysts in the reduction steps removes the need for costly metal scavenging and testing, streamlining the quality control workflow and reducing overall production expenses.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical industry, and this route enhances reliability by simplifying the manufacturing workflow. The reactions are robust and tolerant of minor variations in conditions, reducing the risk of batch failures that can disrupt supply schedules. The intermediates formed are stable solids that can be easily isolated, stored, and transported, allowing for flexible production scheduling and the creation of strategic buffer stocks. This stability ensures that downstream customers receive consistent quality material on time, safeguarding their own production timelines against upstream volatility.
- Scalability and Environmental Compliance: From an environmental perspective, this green chemistry approach aligns perfectly with modern sustainability goals. The process generates significantly less hazardous waste compared to nitration-based routes, simplifying wastewater treatment and disposal. The reactions are inherently scalable, having been demonstrated to work effectively from gram to kilogram scales without loss of efficiency. This scalability allows manufacturers to respond rapidly to market demand surges without the need for extensive process re-engineering. Moreover, the reduced environmental footprint enhances the corporate social responsibility profile of the supply chain, a factor increasingly valued by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Ticagrelor Intermediate A using this advanced methodology. These insights are derived directly from the patent data and practical manufacturing experience, aiming to clarify the operational benefits and technical feasibility of the process for potential partners. Understanding these details is crucial for making informed sourcing decisions and optimizing your supply chain strategy.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: Traditional methods rely on hazardous diazotization or nitration reactions which pose significant environmental and safety risks. This novel route utilizes a protected malonate cyclization strategy, completely eliminating the need for dangerous diazonium salts or nitro-reduction steps, thereby enhancing operational safety and reducing waste treatment costs.
Q: What are the key advantages regarding yield and purity for this intermediate?
A: The patented process demonstrates consistently high yields across all four steps, with cyclization yields exceeding 90% and final deprotection yields approaching 90%. The use of crystallization and standard extraction workups ensures high purity without the need for complex chromatographic purification, making it ideal for GMP manufacturing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes readily available commodity chemicals like thiourea and diethyl malonate derivatives. The reaction conditions involve standard heating and stirring without requiring extreme pressures or exotic catalysts, facilitating easy transfer from laboratory to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Dichloro-2-(propylthio)-5-aminopyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex cardiovascular drugs like Ticagrelor depends on a partner who can deliver both technical excellence and supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the market phase. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Intermediate A meets the highest international standards for API synthesis. Our facility is equipped to handle the specific nuances of this green synthesis route, from the initial cyclization to the final deprotection, ensuring optimal yield and consistency.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this superior synthetic route. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your unique production needs. Let us help you secure a sustainable, cost-effective, and high-quality supply of this critical pharmaceutical intermediate.
