Scalable Synthesis of 3-Benzazepine Derivatives for CNS Disorder Treatments via Optimized Cyclization
Introduction to Advanced Benzazepine Synthesis Technology
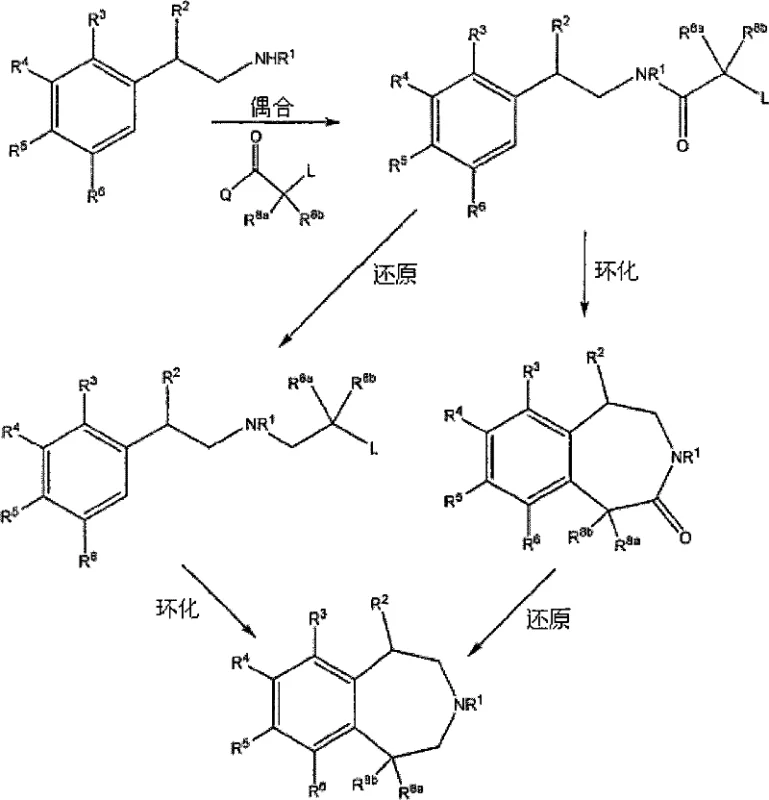
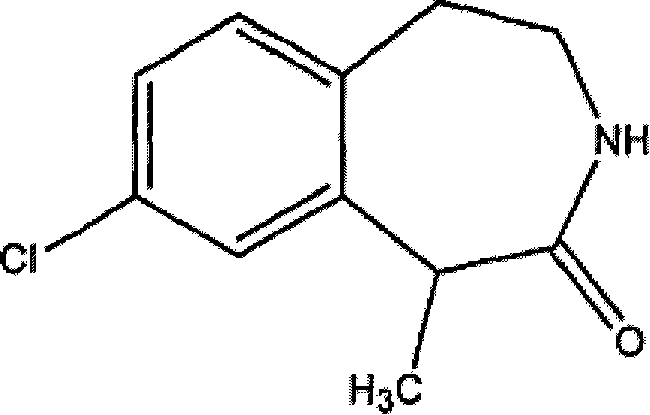
The pharmaceutical industry's relentless pursuit of effective treatments for central nervous system (CNS) disorders, particularly obesity, has placed a significant spotlight on serotonin (5-HT) receptor agonists. Patent CN1805939B introduces a groundbreaking methodology for the preparation of 3-benzazepines and their salts, which serve as potent 5-HT2C receptor agonists. This technology addresses the critical need for efficient, scalable, and high-purity synthetic routes to these complex heterocyclic structures. Unlike traditional methods that often suffer from low yields or require hazardous reagents, the disclosed processes leverage robust organic transformations such as Friedel-Crafts cyclization and selective borane reduction. For R&D directors and procurement specialists, understanding the nuances of this patent is essential, as it outlines a pathway to producing high-quality pharmaceutical intermediates that can streamline the supply chain for next-generation anti-obesity medications. The versatility of the substituents allowed on the benzazepine core further enhances its value as a platform technology for diverse drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-benzazepine cores has been fraught with challenges that hinder commercial viability. Conventional routes often rely on multi-step sequences involving sensitive organometallic reagents or harsh conditions that compromise overall yield and purity. Many prior art methods struggle with the formation of the seven-membered azepine ring, frequently resulting in significant amounts of polymeric byproducts or incomplete cyclization. Furthermore, controlling the stereochemistry at the C1 position has traditionally required expensive chiral catalysts or cumbersome chromatographic separations, which are impractical for large-scale manufacturing. The reliance on transition metals also introduces the risk of heavy metal contamination, necessitating costly purification steps to meet stringent regulatory standards for active pharmaceutical ingredients (APIs). These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for pharmaceutical companies aiming to bring CNS therapeutics to market quickly.
The Novel Approach
The methodology presented in CN1805939B offers a transformative solution by utilizing a streamlined sequence of amide formation, Lewis acid-catalyzed cyclization, and reduction. This approach bypasses the need for precious metal catalysts, instead employing widely available and cost-effective reagents like aluminum chloride and borane. The cyclization step, performed at elevated temperatures (120-150°C), drives the formation of the benzazepinone ring with high efficiency, minimizing side reactions. Moreover, the patent describes a highly effective chiral resolution strategy using L-(+)-tartaric acid, which allows for the isolation of single enantiomers with exceptional purity (ee > 98%) through simple crystallization techniques. This not only reduces the environmental footprint by avoiding complex chromatography but also significantly lowers the cost of goods sold (COGS). By integrating these robust chemical transformations, the novel approach ensures a reliable supply of high-purity intermediates suitable for commercial scale-up.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this synthetic strategy lies in the intramolecular Friedel-Crafts acylation or alkylation, facilitated by strong Lewis acids. In the conversion of amide precursors to the benzazepinone core (Formula II), aluminum chloride (AlCl3) acts as a potent activator. The mechanism involves the coordination of the Lewis acid to the carbonyl oxygen or the halogen leaving group, generating a highly electrophilic species. This activated intermediate then undergoes an electrophilic aromatic substitution with the pendant phenyl ring, closing the seven-membered ring. The reaction conditions, typically involving heating in solvents like 1,2-dichlorobenzene or decalin, provide the necessary thermal energy to overcome the activation barrier for ring closure while maintaining the stability of the substrate. This step is critical as it establishes the rigid scaffold required for biological activity. The use of excess Lewis acid ensures complete conversion, and the subsequent aqueous workup effectively quenches the reaction, allowing for the isolation of the crude lactam which can be further purified by recrystallization.
Following cyclization, the reduction of the lactam carbonyl to the corresponding amine is achieved using borane complexes, such as BH3·THF. This reduction is chemoselective, targeting the amide functionality without affecting other sensitive groups on the aromatic ring. The mechanism proceeds through the formation of an iminium ion intermediate, which is subsequently reduced by hydride transfer from the boron species. The presence of Lewis acid additives like BF3·TBME can further accelerate this reduction, enhancing reaction rates and yields. Crucially, this reduction step preserves the stereochemical integrity of adjacent chiral centers if present, or sets the stage for subsequent resolution. The combination of these mechanistic steps results in a robust process that minimizes impurity formation, ensuring that the final benzazepine product meets the rigorous purity specifications demanded by the pharmaceutical industry for clinical applications.
How to Synthesize 3-Benzazepine Derivatives Efficiently
The practical implementation of this synthesis involves a carefully controlled sequence of reactions designed to maximize yield and safety. The process begins with the coupling of a substituted phenethylamine with an alpha-halo acyl chloride to form the linear amide precursor. This step is typically conducted in polar aprotic solvents like acetonitrile at low temperatures to control exotherms. Following isolation, the amide undergoes the critical cyclization step using aluminum chloride at high temperatures, followed by quenching and extraction. The resulting benzazepinone is then reduced using borane-THF to yield the target amine. For chiral compounds, the racemic amine is subjected to resolution with tartaric acid in a tert-butanol/water mixture. Detailed standardized operating procedures for each of these steps, including specific molar ratios, temperature profiles, and workup protocols, are essential for reproducibility. The detailed standardized synthesis steps are outlined in the guide below.
- Perform amide coupling between a phenethylamine derivative and a halo-acyl chloride in the presence of a base like triethylamine.
- Execute intramolecular Friedel-Crafts cyclization using aluminum chloride at elevated temperatures (120-150°C) to form the benzazepinone core.
- Reduce the lactam carbonyl group using borane-THF complex followed by chiral resolution using L-(+)-tartaric acid to isolate the desired enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic routes described in CN1805939B presents substantial strategic advantages. The primary benefit lies in the drastic simplification of the supply chain for raw materials. By utilizing commodity chemicals such as aluminum chloride, borane, and tartaric acid, manufacturers can avoid the volatility and high costs associated with specialized transition metal catalysts. This shift not only stabilizes pricing but also mitigates the risk of supply disruptions caused by the limited availability of precious metals. Furthermore, the robustness of the high-temperature cyclization step allows for processing in standard glass-lined or stainless steel reactors without the need for exotic equipment, facilitating easier technology transfer between sites. The ability to achieve high purity through crystallization rather than chromatography significantly reduces solvent consumption and waste generation, aligning with modern green chemistry initiatives and reducing disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts in favor of aluminum-based Lewis acids results in a direct and significant reduction in raw material expenses. Additionally, the high yields reported in the patent examples minimize the loss of valuable intermediates, further driving down the cost per kilogram of the final product. The use of simple aqueous workups and crystallization purifications reduces the demand for large volumes of organic solvents and silica gel, lowering both material and waste treatment costs. This economic efficiency makes the process highly attractive for the commercial production of generic APIs where margin pressure is intense.
- Enhanced Supply Chain Reliability: The reliance on globally available, non-regulated reagents ensures a stable and continuous supply of inputs for manufacturing. Unlike processes dependent on single-source chiral ligands or custom-synthesized catalysts, this route uses bulk chemicals that can be sourced from multiple vendors worldwide. This diversification reduces the risk of production stoppages due to vendor-specific issues. Moreover, the scalability of the reaction conditions, demonstrated from gram to hundred-gram scales in the patent, provides confidence that the process can be seamlessly expanded to metric ton quantities to meet market demand without encountering unforeseen engineering hurdles.
- Scalability and Environmental Compliance: The process design inherently supports large-scale operations by avoiding extremely low-temperature steps or highly pyrophoric reagents that are difficult to handle in large vessels. The high-temperature cyclization is thermally manageable with standard cooling systems, and the borane reduction can be controlled through slow addition protocols. From an environmental perspective, the reduction in heavy metal usage simplifies the regulatory burden regarding residual metal limits in the final drug substance. The ability to recycle solvents like toluene and dichloromethane, combined with the generation of less hazardous waste streams, facilitates compliance with increasingly strict environmental regulations, thereby safeguarding the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of benzazepine derivatives based on the patented technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this chemistry into their existing production portfolios. The responses cover aspects ranging from reaction conditions to purity control measures.
Q: What is the primary advantage of the cyclization method described in CN1805939B?
A: The method utilizes robust Lewis acid catalysis (AlCl3) which allows for high-temperature cyclization without requiring expensive transition metal catalysts, significantly simplifying the purification process and reducing raw material costs.
Q: How is chirality controlled in the synthesis of these benzazepine derivatives?
A: Chirality is effectively managed through a classical resolution process using L-(+)-tartaric acid in a tert-butanol and water solvent system, achieving high enantiomeric excess (ee > 98%) through sequential recrystallization.
Q: Can this process be scaled for commercial API production?
A: Yes, the patent details both small-scale and large-scale examples (up to hundreds of grams) demonstrating that the exothermic reactions and workup procedures are manageable and suitable for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of complex pharmaceutical intermediates like 3-benzazepines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral purity analysis via HPLC, to guarantee that every batch meets the highest quality standards required for clinical and commercial use. Our commitment to excellence extends beyond mere manufacturing; we actively collaborate with clients to optimize processes for cost and efficiency.
We invite you to engage with our technical procurement team to discuss your specific requirements for benzazepine derivatives. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized synthetic routes can reduce your overall project costs. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you accelerate your drug development timeline with our proven expertise in heterocyclic chemistry and scalable manufacturing solutions.
