Advanced Industrial Synthesis of Tolvaptan via Silylation Protection Strategy
The pharmaceutical landscape for treating hyponatremia has been significantly advanced by the development of Tolvaptan, a potent non-peptide arginine vasopressin V2 receptor antagonist. As detailed in patent CN102260213A, a novel and highly efficient preparation method has been established that addresses critical bottlenecks in traditional manufacturing. This innovative approach utilizes 7-chloro-1,2,3,4,5-tetrahydro-1H-1-benzo-aza-5-one as the primary starting material, subjecting it to a streamlined four-step sequence involving reduction, silylating agent protection, amino acylation, and final deprotection. The strategic implementation of silyl protection groups serves as the cornerstone of this technology, effectively shielding reactive sites to prevent side reactions that typically plague the synthesis of such complex benzazepine derivatives. By optimizing reaction conditions and reagent selection, this method not only enhances the overall yield but also drastically simplifies the downstream purification processes, making it an ideal candidate for large-scale industrial application.
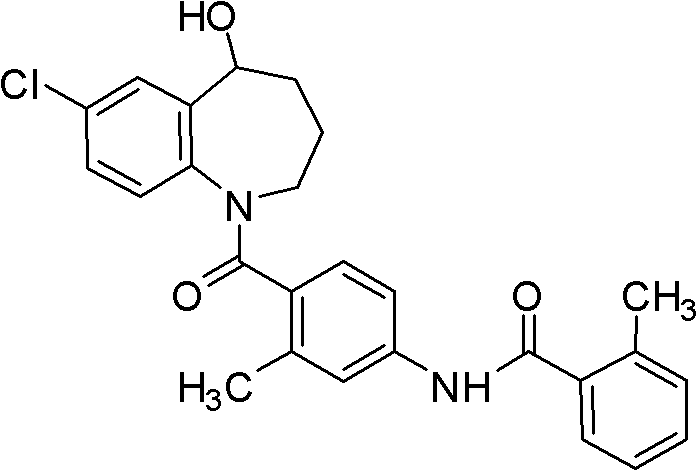
For R&D directors evaluating process feasibility, understanding the structural integrity and the specific functional group transformations is paramount. The target molecule, Tolvaptan, features a sensitive 5-hydroxy-2,3,4,5-tetrahydro-1H-1-benzazepine moiety linked to a substituted benzamide. Conventional synthetic routes often struggle with the regioselective acylation of the nitrogen atom in the presence of the hydroxyl group, leading to impurities that are difficult to separate. The methodology disclosed in CN102260213A elegantly solves this by temporarily masking these functionalities, ensuring that the subsequent acylation occurs exclusively at the desired nitrogen center. This level of control is essential for meeting the stringent purity specifications required for active pharmaceutical ingredients (APIs), particularly when targeting regulatory markets where impurity profiles are scrutinized heavily.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, several synthetic pathways were attempted, each fraught with significant industrial disadvantages that hindered cost-effective mass production. For instance, the route reported by Kazumi Kondo et al. relies heavily on the use of Platinum Dioxide (PtO2) for the catalytic reduction of the carbonyl group to a hydroxyl group. While chemically effective on a small scale, the reliance on precious metal catalysts like PtO2 introduces substantial cost burdens and necessitates complex removal procedures to ensure the final product is free of heavy metal residues. Furthermore, this legacy route involves multiple steps requiring column chromatography for purification, a technique that is notoriously difficult to scale up and results in significant material loss, with reported overall yields ranging merely from 30% to 50%. Similarly, other methods involving palladium catalysis or complex multi-step sequences starting from obscure raw materials like 4-chloroacetophenone base xanthate suffer from long production cycles and low market availability of starting materials.
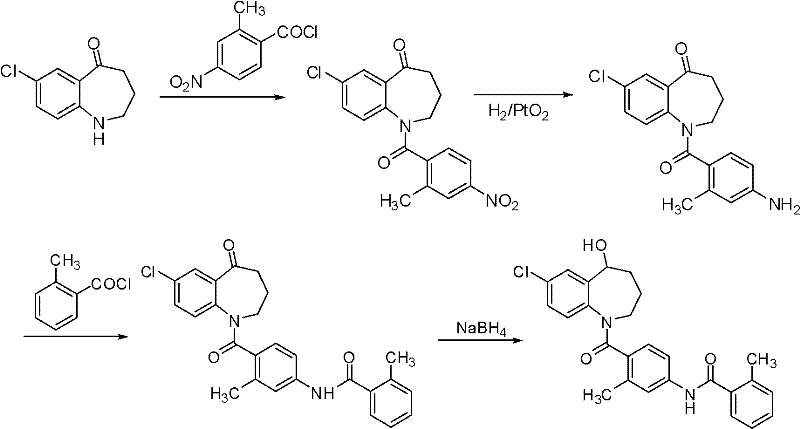
The Novel Approach
In stark contrast, the novel approach presented in the patent data revolutionizes the synthesis by replacing expensive catalytic hydrogenation with a simple chemical reduction using sodium borohydride (NaBH4). This substitution alone represents a massive leap forward in terms of cost reduction in pharmaceutical manufacturing, as it eliminates the need for high-pressure hydrogenation equipment and precious metal catalysts. The core innovation lies in the introduction of a silylation step immediately following the reduction. By converting the unstable 5-hydroxy intermediate into a stable bis-silylated derivative (Intermediate I), the process prevents the formation of O-acylated byproducts during the subsequent coupling reaction. This strategic protection allows the reaction to proceed with high chemoselectivity, enabling the direct use of crude intermediates in subsequent steps without the need for intermediate purification. Consequently, the production cycle is drastically shortened, and the overall yield is boosted to approximately 78%, providing a robust and economically viable pathway for commercial scale-up.
Mechanistic Insights into Silyl-Protection and Mixed Anhydride Coupling
The mechanistic elegance of this synthesis lies in the precise manipulation of nucleophilicity and electrophilicity through protecting group chemistry. In the initial reduction phase, sodium borohydride selectively reduces the ketone at the 5-position of the benzazepine ring to a secondary alcohol. Without protection, this alcohol would compete with the secondary amine during acylation, leading to a mixture of N-acyl and O-acyl products. The patent specifies the use of silylating agents such as trimethylchlorosilane (TMSCl) or hexamethyldisilazane (HMDS) in the presence of a base like potassium tert-butoxide. This reaction converts both the hydroxyl group and the amine nitrogen into their respective trimethylsilyl (TMS) ethers and amines. This dual protection renders the oxygen non-nucleophilic while maintaining the nitrogen's ability to act as a nucleophile under specific activation conditions, or alternatively, the nitrogen is deprotected selectively or reacts via a specific mechanism facilitated by the reaction conditions.
Following the protection, the coupling partner, 4-(2-toluylamino)-2-methyl-phenylformic acid, is activated in situ to form a mixed anhydride (Intermediate II) using reagents like pivaloyl chloride or trifluoroacetic anhydride at low temperatures (-10 to -5°C). This activation creates a highly electrophilic carbonyl carbon that is susceptible to nucleophilic attack. When Intermediate I is introduced under alkaline conditions (e.g., triethylamine), the nucleophilic nitrogen attacks the activated carbonyl, forming the critical amide bond. The bulky silyl groups sterically hinder any attack on the oxygen, ensuring exclusive N-acylation. Finally, the silyl groups are labile to acidic hydrolysis. Treatment with dilute hydrochloric acid or sulfuric acid at mild temperatures cleaves the Si-O and Si-N bonds, regenerating the free hydroxyl and amine groups to yield the final Tolvaptan molecule with high purity (>99.5%).
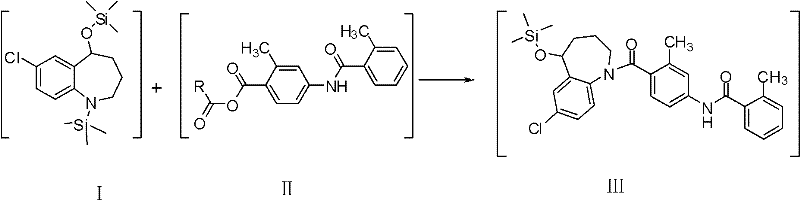
How to Synthesize Tolvaptan Efficiently
The synthesis of Tolvaptan via this patented route is designed for operational simplicity and high throughput, making it accessible for manufacturing facilities aiming to optimize their production lines. The process begins with the quantitative reduction of the ketone precursor, followed immediately by the silylation step which acts as a 'chemical lock' to preserve the integrity of the intermediate. The subsequent formation of the mixed anhydride and the coupling reaction are performed under controlled low-temperature conditions to minimize thermal degradation and side reactions. A key feature of this protocol is the 'telescoped' nature of the synthesis, where intermediates do not require isolation or chromatographic purification between steps, significantly reducing solvent consumption and waste generation. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures, please refer to the standardized guide below.
- Reduce 7-chloro-1,2,3,4,5-tetrahydro-1H-1-benzo-aza-5-one using sodium borohydride to obtain the corresponding alcohol.
- Protect the hydroxyl and amine groups of the alcohol intermediate using a silylating agent like trimethylchlorosilane to form Intermediate I.
- React 4-(2-toluylamino)-2-methyl-phenylformic acid with pivaloyl chloride to form a mixed anhydride (Intermediate II).
- Couple Intermediate I and Intermediate II under alkaline conditions to form the protected Tolvaptan precursor (Intermediate III).
- Perform acidic hydrolysis on Intermediate III to remove silyl groups and obtain the final Tolvaptan product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers transformative benefits that directly impact the bottom line and operational reliability. Traditional methods relying on precious metal catalysts and chromatographic purification create volatile cost structures and supply bottlenecks. By shifting to a base-metal-free, non-chromatographic process, manufacturers can achieve significant cost savings in pharmaceutical manufacturing. The elimination of Platinum Dioxide and Palladium catalysts removes the exposure to fluctuating precious metal markets and the costly logistics associated with metal recovery and disposal. Furthermore, the ability to use crude intermediates directly in subsequent steps reduces the number of unit operations, thereby lowering energy consumption, labor costs, and solvent usage. This streamlined workflow translates into a more predictable and lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global marketplace.
- Cost Reduction in Manufacturing: The replacement of expensive catalytic hydrogenation with chemical reduction using sodium borohydride represents a fundamental shift in cost structure. Sodium borohydride is a commodity chemical with a stable supply chain, unlike PtO2 which is subject to geopolitical and mining constraints. Additionally, the avoidance of column chromatography—a batch process that is slow and solvent-intensive—in favor of crystallization and filtration significantly reduces processing time and solvent recovery costs. The high yield of 78% compared to the 30-50% of older methods means that less raw material is required to produce the same amount of API, further driving down the variable costs per kilogram.
- Enhanced Supply Chain Reliability: The starting material, 7-chloro-1,2,3,4,5-tetrahydro-1H-1-benzo-aza-5-one, is chemically robust and readily available from multiple suppliers, reducing the risk of single-source dependency. The simplified process flow, which eliminates complex purification steps, reduces the likelihood of batch failures due to operational errors or equipment malfunctions. This robustness ensures a consistent supply of high-purity Tolvaptan, which is critical for maintaining uninterrupted production schedules for downstream drug formulations. The reduced production cycle time also allows for faster response to market demand fluctuations, enhancing the agility of the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory bench scale to multi-ton commercial production without the need for specialized high-pressure reactors or extensive metal scrubbing systems. The reduction in solvent usage and the elimination of heavy metal waste streams align with increasingly stringent environmental regulations and green chemistry principles. This not only minimizes the environmental footprint but also reduces the regulatory burden and costs associated with waste treatment and disposal, making the facility more sustainable and compliant with global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific advantages and procedural details outlined in the patent documentation, providing clarity on why this route is superior for industrial applications. Understanding these nuances helps stakeholders make informed decisions regarding technology transfer and process validation.
Q: How does the silylation strategy improve Tolvaptan purity?
A: The silylation step protects both the hydroxyl and amine functionalities, preventing unwanted O-acylation side reactions during the coupling phase. This chemoselectivity ensures the final product achieves purity levels exceeding 99.5% without requiring complex chromatographic purification.
Q: What is the yield advantage of this novel method compared to prior art?
A: While conventional methods reported in literature often suffer from yields between 30% and 50% due to harsh reduction conditions and purification losses, this patented process achieves a total yield of approximately 78%, significantly enhancing material efficiency.
Q: Does this process involve difficult-to-remove heavy metal catalysts?
A: No, unlike previous routes that utilized expensive Platinum Dioxide (PtO2) or Palladium catalysts which require rigorous removal steps, this method employs Sodium Borohydride for reduction and standard organic reagents, eliminating heavy metal contamination risks entirely.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tolvaptan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the pharmaceutical sector. Our team of expert chemists has thoroughly analyzed the patented route described in CN102260213A and possesses the technical capability to execute this process with precision and efficiency. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications (>99.5%) required for Tolvaptan, ensuring that every batch meets the highest international standards for safety and efficacy.
We invite potential partners to engage with us to explore how this cost-effective and high-yield synthesis route can benefit your supply chain. By leveraging our expertise in process optimization and scale-up, we can help you secure a reliable source of high-purity Tolvaptan intermediates and APIs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and partnership excellence.
