Advanced Manufacturing of Topiramate Intermediates: A Safe, High-Yield Sulfuryl Chloride Protocol
Advanced Manufacturing of Topiramate Intermediates: A Safe, High-Yield Sulfuryl Chloride Protocol
The pharmaceutical industry continuously seeks robust synthetic routes that balance safety, efficiency, and scalability, particularly for high-volume antiepileptic agents like topiramate. Patent CN101450951A introduces a transformative methodology for producing topiramate that addresses critical safety deficiencies inherent in legacy manufacturing processes. By shifting from hazardous sodium hydride-based chemistries to a controlled sulfuryl chloride pathway, this innovation offers a reliable topiramate intermediate supplier with a distinct competitive edge. The core breakthrough lies in the strategic substitution of toxic sulfamoyl chloride with readily available sulfuryl chloride, coupled with a novel dual-solvent system that enhances both reaction kinetics and product purity. This technical evolution not only mitigates the risk of industrial accidents but also streamlines the purification workflow, ensuring consistent access to high-purity topiramate for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of topiramate has been plagued by severe safety hazards and operational inefficiencies that hinder large-scale production. Traditional protocols, such as those described in earlier literature, often rely on the direct reaction of diacetone fructose with sulfamoyl chloride in the presence of sodium hydride (NaH) and dimethylformamide (DMF). This combination is notoriously unstable; the interaction between NaH and DMF is highly exothermic and difficult to control thermally, creating a tangible possibility of blasting or thermal runaway in a reactor setting. Furthermore, sulfamoyl chloride itself is a toxic reagent with limited commercial availability, introducing supply chain volatility and handling risks for operators. The reliance on such dangerous reagents necessitates expensive engineering controls and specialized containment infrastructure, driving up the overall cost of goods sold without guaranteeing superior yields.
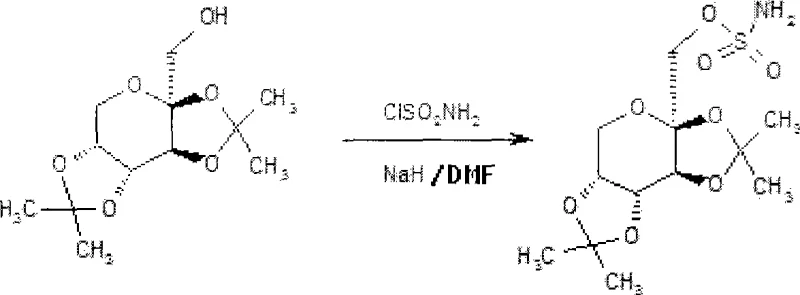
The Novel Approach
In stark contrast, the methodology disclosed in CN101450951A adopts a fundamentally safer and more economically viable route that eliminates the need for explosive reagents. This novel approach utilizes sulfuryl chloride, a non-explosive and widely accessible commodity chemical, to first generate a chlorosulfonic ester intermediate. This intermediate is subsequently subjected to ammonolysis using ammonia gas, a standard industrial reagent, to finalize the topiramate structure. The process is conducted in a carefully engineered mixed solvent system—toluene combined with methylene dichloride for the esterification step, and tetrahydrofuran mixed with methylene dichloride for the amination step. This specific solvent engineering dramatically improves the solubility of the starting material, diacetone fructose, which is poorly soluble in pure toluene, thereby facilitating smoother mass transfer and reaction homogeneity. The result is a process that is not only inherently safer but also capable of achieving yields exceeding 90%, representing a substantial improvement over the roughly 60% yields typical of older, less optimized pathways.
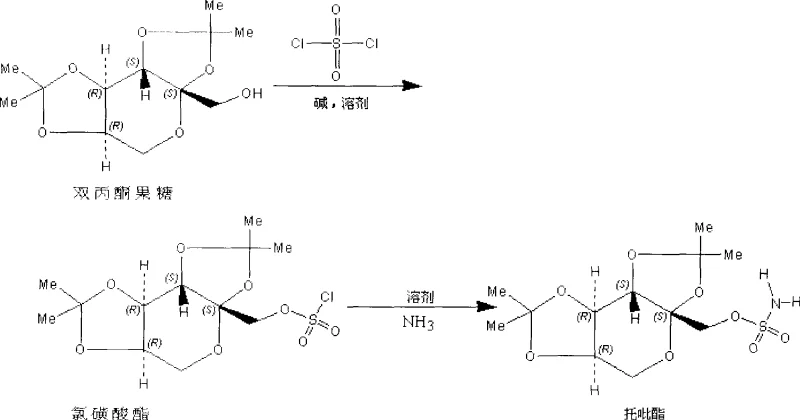
Mechanistic Insights into Sulfuryl Chloride-Mediated Esterification and Ammonolysis
The mechanistic elegance of this process lies in the sequential activation of the hydroxyl group on the diacetone fructose scaffold followed by nucleophilic displacement. In the initial esterification phase, sulfuryl chloride acts as an electrophilic sulfur source, reacting with the primary hydroxyl group of diacetone fructose in the presence of a weak base such as pyridine or triethylamine. The base serves a dual purpose: it scavenges the hydrogen chloride byproduct generated during the formation of the chlorosulfonic ester, preventing acid-catalyzed degradation of the sensitive acetonide protecting groups, and it activates the alcohol for nucleophilic attack. The use of a toluene/methylene dichloride mixture is critical here; while toluene provides a high-boiling medium suitable for thermal control, methylene dichloride ensures complete dissolution of the polar sugar derivative, preventing heterogeneous reaction conditions that could lead to incomplete conversion or side reactions.
Following the isolation or in-situ processing of the chlorosulfonic ester, the mechanism shifts to ammonolysis. Ammonia gas, introduced under mild pressure (0.1 atm to 0.5 atm), acts as a potent nucleophile, attacking the sulfur center of the chlorosulfonate moiety. This displaces the chloride ion and forms the stable sulfamate linkage characteristic of topiramate. The choice of a tetrahydrofuran (THF)/methylene dichloride solvent system for this step is equally strategic; THF is miscible with liquid ammonia and stabilizes the transition state, while the low reaction temperature of 15°C to 25°C suppresses potential elimination reactions or deprotection of the acetonide groups. This precise control over reaction parameters ensures that the impurity profile remains minimal, reducing the burden on downstream purification units and enhancing the overall purity of the final API intermediate.
How to Synthesize Topiramate Efficiently
The execution of this synthesis requires strict adherence to the solvent ratios and temperature profiles defined in the patent to maximize yield and safety. The process begins with the preparation of the chlorosulfonic ester, where maintaining the temperature between 0°C and 40°C is vital to prevent exothermic spikes. Following this, the ammonolysis step must be conducted in a closed system to maintain ammonia pressure, ensuring complete conversion of the intermediate. The detailed standardized synthesis steps, including specific reagent quantities and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency route.
- Perform esterification of diacetone fructose with sulfuryl chloride in a toluene/methylene dichloride mixed solvent at 0-40°C using pyridine as a base.
- Conduct ammonolysis by reacting the resulting chlorosulfonic ester with ammonia gas in a tetrahydrofuran/methylene dichloride solvent system at 15-25°C.
- Purify the crude topiramate product via recrystallization using anhydrous ethanol and cyclohexane precipitation at controlled temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond simple chemical yield. The shift away from hazardous reagents like sodium hydride and sulfamoyl chloride fundamentally alters the risk profile of the manufacturing facility, potentially lowering insurance premiums and reducing the need for specialized hazardous waste disposal services. By utilizing sulfuryl chloride and ammonia, which are bulk commodities with stable pricing and widespread availability, manufacturers can insulate themselves from the supply volatility associated with niche, high-toxicity reagents. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the elimination of expensive and dangerous reagents. Sodium hydride is a high-cost material that requires careful handling and quenching, adding significant operational overhead. By replacing it with pyridine or triethylamine and using cheap sulfuryl chloride, the direct material costs are significantly reduced. Furthermore, the high yield (>90%) minimizes the loss of valuable starting materials like diacetone fructose, effectively lowering the cost per kilogram of the final product. The simplified solvent recovery process, facilitated by the specific boiling points of the toluene and THF mixtures, also contributes to substantial cost savings in utility consumption.
- Enhanced Supply Chain Reliability: Reliability in the supply of topiramate intermediates is paramount for downstream API manufacturers. This process utilizes raw materials that are produced on a massive industrial scale globally, ensuring that supply disruptions are unlikely. The robustness of the reaction conditions, which tolerate slight variations in temperature and pressure better than the explosive NaH/DMF system, means that batch failures are rare. This consistency allows supply chain planners to forecast inventory levels with greater accuracy and reduce the need for excessive safety stock, optimizing working capital across the value chain.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The absence of pyrophoric reagents simplifies the engineering requirements for large reactors, allowing for faster technology transfer from pilot plant to full production. Additionally, the environmental footprint is reduced; the process avoids the generation of heavy metal waste or difficult-to-treat organic sludge associated with alternative routes. The use of recyclable solvents like toluene and THF aligns with modern green chemistry principles, facilitating easier regulatory approval and compliance with increasingly stringent environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this topiramate synthesis route. These answers are derived directly from the experimental data and technical specifications provided in patent CN101450951A, offering clarity on process safety, yield expectations, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: Why is the sulfuryl chloride route safer than the traditional sulfamoyl chloride method?
A: The traditional method utilizes sodium hydride (NaH) and DMF, which poses significant explosion risks due to uncontrollable exothermic reactions. The new protocol replaces hazardous reagents with stable sulfuryl chloride and ammonia, eliminating thermal runaway hazards.
Q: What yields can be expected from this optimized solvent system?
A: By optimizing the solvent ratios, specifically using a toluene/methylene dichloride mixture, the process achieves yields exceeding 90%, significantly higher than the 60% typical of older methods.
Q: How does the solvent choice impact the solubility of diacetone fructose?
A: Pure toluene has poor solvency for diacetone fructose, often requiring violent stirring or heating. The mixed solvent system dramatically increases solubility, allowing for smoother reaction kinetics and easier operational control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topiramate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the long-term sustainability of the pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101450951A are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of topiramate intermediate meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of solvent optimization and reaction control, delivering a product that supports the uninterrupted manufacture of life-saving antiepileptic medications.
We invite global partners to collaborate with us to leverage this advanced technology for their supply needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the confidence of a proven, safe, and economically superior manufacturing partner.
