Advanced One-Step Refining Technology for High-Purity Topiramate Manufacturing
Introduction to Advanced Topiramate Purification
The global pharmaceutical landscape demands increasingly stringent purity profiles for Active Pharmaceutical Ingredients (APIs), particularly for widely prescribed antiepileptic and antimigraine agents like Topiramate. As detailed in patent CN107778335B, a breakthrough refining methodology has been developed that addresses the critical bottleneck of impurity control in Topiramate manufacturing. This innovation is not merely a incremental improvement but a fundamental shift in downstream processing, enabling producers to consistently achieve single impurity levels below 0.1% and total impurities under 0.5% through a streamlined one-step operation. 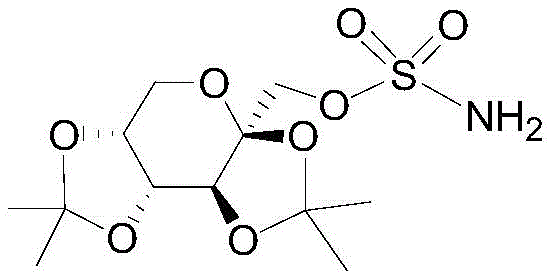 The structural complexity of Topiramate, specifically its bis-O-(1-methylethylidene)-β-D-fructopyranose sulfamate framework, makes it susceptible to specific degradation pathways during synthesis and isolation. Traditional methods often struggle to remove trace byproducts formed during the ammonolysis of the chlorosulfonate precursor, leading to batch failures or extensive rework. This new technical disclosure offers a robust solution that aligns perfectly with the needs of R&D directors seeking reliable, high-yield pathways for commercial API production.
The structural complexity of Topiramate, specifically its bis-O-(1-methylethylidene)-β-D-fructopyranose sulfamate framework, makes it susceptible to specific degradation pathways during synthesis and isolation. Traditional methods often struggle to remove trace byproducts formed during the ammonolysis of the chlorosulfonate precursor, leading to batch failures or extensive rework. This new technical disclosure offers a robust solution that aligns perfectly with the needs of R&D directors seeking reliable, high-yield pathways for commercial API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Topiramate has relied heavily on ammonolysis reactions followed by cumbersome purification sequences that introduce significant operational risks and costs. As illustrated in earlier patents such as U.S. Pat. No. 4,538,770, the conventional workflow involves reacting the chlorosulfonate precursor with ammonia gas under pressure, followed by solvent removal and a two-stage crystallization process using n-hexane and ethanol-water. 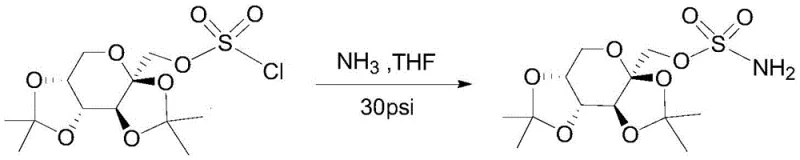 This multi-step approach is inherently inefficient; it necessitates long crystallization times, multiple drying phases, and the handling of large volumes of volatile hydrocarbons. Furthermore, alternative methods described in Chinese patent CN 200710068357.3 utilize ammonia water in cyclohexane but fail to adequately control anhydrous conditions, resulting in crude products with high impurity loads that require aggressive washing and concentration steps. These legacy processes are plagued by the persistent presence of degradation impurities, specifically those eluting at 7.6 minutes and 31 minutes in HPLC analysis, which conventional recrystallization techniques cannot reduce below the critical 0.1% threshold required for pharmaceutical grade material.
This multi-step approach is inherently inefficient; it necessitates long crystallization times, multiple drying phases, and the handling of large volumes of volatile hydrocarbons. Furthermore, alternative methods described in Chinese patent CN 200710068357.3 utilize ammonia water in cyclohexane but fail to adequately control anhydrous conditions, resulting in crude products with high impurity loads that require aggressive washing and concentration steps. These legacy processes are plagued by the persistent presence of degradation impurities, specifically those eluting at 7.6 minutes and 31 minutes in HPLC analysis, which conventional recrystallization techniques cannot reduce below the critical 0.1% threshold required for pharmaceutical grade material.
The Novel Approach
In stark contrast to these fragmented and hazardous legacy protocols, the refining method disclosed in CN107778335B introduces a unified, one-step crystallization strategy that dramatically simplifies the purification landscape. By leveraging a specific binary solvent system comprising an alcohol and an ether, coupled with precise pH modulation using ammonia water, this process effectively discriminates between the target API and its structurally similar impurities. The elimination of intermediate solvent swaps and the reduction of unit operations from multiple recrystallizations to a single cohesive step represents a paradigm shift in process chemistry. This approach not only mitigates the safety risks associated with handling large quantities of pure ether or hexane in separate stages but also drastically reduces the thermal history of the product, thereby minimizing thermal degradation. For supply chain managers, this translates to a significantly shortened production cycle and a more predictable manufacturing timeline, ensuring consistent availability of high-quality Topiramate for downstream formulation.
Mechanistic Insights into Solvent-Mediated Crystallization
The efficacy of this novel refining process lies in the sophisticated interplay between solvent polarity, temperature gradients, and pH control during the crystallization phase. When the crude Topiramate oily liquid is dissolved in an alcohol solvent and heated to reflux, the system achieves a state of maximum solubility where both the API and potential impurities are fully solvated. The subsequent cooling to a specific range of 55-65°C, followed by the adjustment of pH to 9-10 using ammonia water, is a critical mechanistic step. This pH adjustment likely serves to neutralize any acidic degradation byproducts or residual sulfonic acid species that may have formed during the upstream ammonolysis, converting them into more polar salts that remain in the mother liquor rather than co-crystallizing with the neutral Topiramate molecule.  The introduction of the ether solvent as an anti-solvent at this controlled temperature further manipulates the solubility curve, inducing supersaturation in a controlled manner that favors the nucleation of pure Topiramate crystals while excluding the kinetic traps that lead to impurity inclusion.
The introduction of the ether solvent as an anti-solvent at this controlled temperature further manipulates the solubility curve, inducing supersaturation in a controlled manner that favors the nucleation of pure Topiramate crystals while excluding the kinetic traps that lead to impurity inclusion.
Furthermore, the specific selection of ether solvents such as petroleum ether, ethylene glycol dimethyl ether, or methyl tert-butyl ether allows for fine-tuning the dielectric constant of the medium, which is essential for rejecting the stubborn 7.6-minute and 31-minute impurities. The final washing and grinding step with fresh ether solvent acts as a surface purification mechanism, stripping away any adhering mother liquor that contains concentrated impurities from the crystal lattice exterior. This comprehensive control over the thermodynamic and kinetic parameters of crystallization ensures that the resulting solid form possesses not only high chemical purity but also superior physical properties, such as consistent particle size distribution and flowability, which are vital for subsequent tablet compression or capsule filling operations in pharmaceutical manufacturing.
How to Synthesize Topiramate Efficiently
The implementation of this refining technology requires strict adherence to the optimized parameters regarding solvent ratios and temperature profiles to ensure reproducibility at scale. The process begins with the dissolution of the crude sulfamate oily liquid in a selected alcohol solvent, followed by the critical pH adjustment and anti-solvent addition sequence described in the mechanistic section. Operators must maintain precise thermal control during the dropwise addition of the ether solvent to prevent shock precipitation, which could trap impurities within the growing crystals. 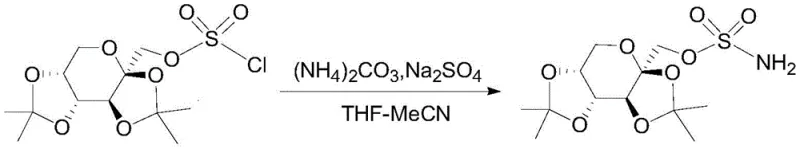 For a detailed breakdown of the specific operational parameters, solvent volumes, and safety protocols required to execute this synthesis successfully, please refer to the standardized technical guide provided below.
For a detailed breakdown of the specific operational parameters, solvent volumes, and safety protocols required to execute this synthesis successfully, please refer to the standardized technical guide provided below.
- Dissolve crude Topiramate oily liquid in an alcohol solvent, heat to reflux, cool to 55-65°C, and adjust pH to 9-10 using ammonia water.
- Dropwise add an ether solvent to the system while maintaining temperature between 50-65°C, then cool naturally to room temperature for crystallization.
- Filter the crystallized product, wash and grind the filter cake with fresh ether solvent, and dry under vacuum to obtain pure Topiramate crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-step refining methodology offers profound advantages that directly impact the bottom line and operational resilience of pharmaceutical manufacturers. By consolidating what was previously a multi-stage purification ordeal into a single, efficient unit operation, companies can realize substantial reductions in utility consumption, labor hours, and equipment occupancy time. The elimination of complex solvent exchange procedures and the reduction in overall processing time mean that production throughput can be significantly increased without the need for capital-intensive expansion of facility footprint. This efficiency gain is particularly valuable in the context of high-volume API manufacturing, where marginal improvements in cycle time compound into massive annual savings.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently lowers the cost of goods sold (COGS) by minimizing the consumption of expensive high-purity solvents and reducing energy loads associated with heating, cooling, and vacuum drying cycles. Unlike previous methods that required distinct recrystallization steps with different solvent systems, this unified approach allows for better solvent recovery and recycling potential, further driving down raw material costs. Additionally, the high yield exceeding 90% ensures that valuable starting materials are not lost to excessive mother liquor discard or degradation, maximizing the return on investment for every kilogram of precursor purchased.
- Enhanced Supply Chain Reliability: Simplifying the manufacturing process reduces the number of potential failure points, thereby enhancing the reliability of supply for downstream customers. The robustness of the crystallization parameters means that batch-to-batch variability is minimized, reducing the risk of out-of-specification (OOS) results that can delay shipments and disrupt inventory planning. Furthermore, the use of common, commercially available solvents like alcohols and ethers ensures that the supply chain is not vulnerable to shortages of exotic or highly regulated reagents, providing a stable foundation for long-term production planning.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard agitation and filtration equipment that is readily available in most GMP facilities, facilitating a smooth transition from pilot scale to commercial production. From an environmental standpoint, the reduction in solvent usage and the avoidance of hazardous reagents like pure ether in large uncontrolled quantities contribute to a greener manufacturing profile. This alignment with green chemistry principles not only reduces waste disposal costs but also supports corporate sustainability goals, which are increasingly becoming a prerequisite for partnerships with major global pharmaceutical companies.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this refining technology into their existing workflows, we have compiled answers to common inquiries regarding the process specifications and quality outcomes. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these details is crucial for making informed decisions regarding process validation and regulatory filing strategies.
Q: How does this refining method improve upon traditional Topiramate purification?
A: Unlike conventional methods requiring multi-step recrystallization and hazardous solvent exchanges, this novel approach achieves pharmaceutical grade purity (<0.1% single impurity) in a single refining step, significantly reducing processing time and energy consumption.
Q: What specific impurities does this process target?
A: The process is specifically optimized to reduce degradation impurities with retention times of 7.6 minutes and 31 minutes to below 0.1%, meeting strict regulatory standards for API manufacturing that previous methods failed to achieve.
Q: Is this process scalable for industrial production?
A: Yes, the method utilizes common industrial solvents like alcohols and ethers and avoids complex equipment requirements, making it highly suitable for commercial scale-up from pilot batches to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topiramate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably reproduced in your supply chain. We operate with stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities at the ppm level, guaranteeing that every batch of Topiramate meets the exacting standards required for global regulatory submission.
We invite you to engage with our technical procurement team to discuss how this advanced refining technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to move forward with confidence in securing a sustainable and high-quality supply of this critical antiepileptic ingredient.
