Advanced Synthesis of 2-Fluoro-4-Nitrobenzonitrile for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of 2-Fluoro-4-Nitrobenzonitrile for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously demands more efficient and safer pathways for synthesizing critical API intermediates, particularly for tyrosine kinase inhibitors used in oncology and cardiovascular treatments. Patent CN101648890B introduces a groundbreaking synthesis method for 2-fluoro-4-nitrobenzonitrile that fundamentally shifts the production paradigm from hazardous oxidation processes to a streamlined diazotization and cyanation sequence. This technical breakthrough addresses long-standing challenges regarding raw material toxicity, reaction condition severity, and overall process economics, positioning it as a superior choice for reliable pharmaceutical intermediate supplier networks seeking robust manufacturing solutions. By leveraging 3,4-difluoronitrobenzene as a starting point, the process eliminates the need for complex multi-step oxidations that traditionally plague this chemical class, offering a direct route to high-purity outputs essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-fluoro-4-nitrobenzonitrile has relied on arduous pathways that impose significant burdens on both safety protocols and production budgets. One prevalent conventional method involves the oxidation of 2-fluoro-4-nitrotoluene to the corresponding benzoic acid, followed by amidation and dehydration, a route famously documented in early medicinal chemistry literature.

This traditional approach, as illustrated in the reaction scheme above, necessitates the use of aggressive oxidants like chromium trioxide and hazardous dehydrating agents such as oxalyl chloride or phosphorus pentoxide, resulting in molar yields as low as 29% to 43% while generating substantial toxic waste streams. Furthermore, alternative routes attempting to utilize 2-fluoro-4-nitrobenzoic acid directly often suffer from the scarcity and high cost of the starting material, coupled with a lack of optimized reaction conditions in public literature, making them unsuitable for consistent commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a strategic diazotization-bromination followed by a copper-catalyzed cyanation, effectively bypassing the inefficiencies of direct oxidation. This innovative pathway begins with the ammonolysis of 3,4-difluoronitrobenzene to generate 2-fluoro-4-nitroaniline, which is then converted into the bromo-intermediate before final cyanation.
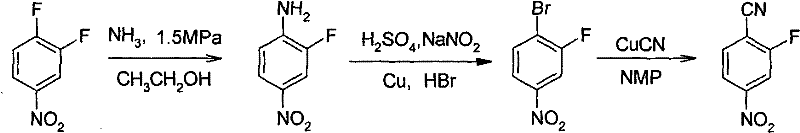
As depicted in the reaction flow, this sequence allows for precise control over impurity profiles and operates under significantly milder conditions compared to the harsh acidic environments of the past. The transition from an amine to a nitrile via a diazonium salt intermediate not only improves the overall atom economy but also utilizes N-methyl-2-pyrrolidone (NMP) as a solvent, which facilitates better solubility and reaction kinetics for the cyanation step, ultimately delivering a yellow crystalline product with exceptional purity specifications.
Mechanistic Insights into Diazotization and Copper-Catalyzed Cyanation
The core of this synthetic success lies in the meticulous execution of the Sandmeyer-type transformation, where the amino group is first converted into a highly reactive diazonium salt using sodium nitrite in an acidic medium at controlled low temperatures between 0°C and 5°C. This低温 step is critical for stabilizing the diazonium species and preventing premature decomposition, which could lead to phenolic by-products that compromise the integrity of the final API intermediate. Following the formation of the diazonium solution, the introduction of copper powder in hydrobromic acid at elevated temperatures triggers the replacement of the diazo group with a bromine atom, yielding 2-fluoro-4-nitrobromobenzene with high regioselectivity.
Subsequently, the cyanation step employs cuprous cyanide dissolved in a toluene and NMP mixture, creating a homogeneous catalytic environment that promotes the nucleophilic substitution of the bromine atom with a cyano group. The use of NMP is particularly advantageous as it stabilizes the copper-cyanide complex and enhances the solubility of the organic substrate, allowing the reaction to proceed efficiently at temperatures around 150°C to 160°C. This mechanistic pathway ensures that the fluorine atom at the ortho-position remains intact despite the presence of strong nucleophiles, a common failure point in other synthetic strategies, thereby guaranteeing the structural fidelity required for high-purity OLED material or pharmaceutical applications.
How to Synthesize 2-Fluoro-4-Nitrobenzonitrile Efficiently
To achieve optimal results in the laboratory or pilot plant, operators must strictly adhere to the temperature gradients and molar ratios defined in the patent examples, particularly during the exothermic diazotization phase. The process requires careful monitoring of the reaction progress via gas chromatography to ensure complete consumption of the amine before proceeding to the bromination stage, minimizing the formation of azo-coupling impurities. For a detailed breakdown of the specific operational parameters, reagent quantities, and workup procedures necessary to replicate this high-yield synthesis, please refer to the standardized protocol below.
- Prepare 2-fluoro-4-nitroaniline by reacting 3,4-difluoronitrobenzene with aqueous ammonia under pressure.
- Convert the amine to 2-fluoro-4-nitrobromobenzene via diazotization with sodium nitrite and subsequent Sandmeyer bromination with copper powder.
- Perform cyanation using cuprous cyanide in NMP solvent at elevated temperatures to yield the final nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthesis route offers transformative benefits by shifting the dependency from scarce, high-cost precursors to widely available commodity chemicals like 3,4-difluoronitrobenzene. The elimination of expensive and hazardous reagents such as oxalyl chloride and trifluoroacetic anhydride drastically simplifies the supply chain logistics, reducing the need for specialized storage and handling protocols that typically inflate operational expenditures. Moreover, the ability to source raw materials that are standard in the fine chemical industry ensures a more resilient supply chain, mitigating the risks associated with vendor shortages or geopolitical disruptions that often affect niche intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of costly oxidation steps with a more direct functional group interconversion that utilizes cheaper inorganic salts and solvents. By avoiding the multi-step degradation of toluene derivatives which often suffer from poor yields and high waste disposal costs, manufacturers can achieve substantial cost savings in pharma intermediate manufacturing without compromising on quality. The simplified purification process, which relies on standard recrystallization techniques rather than complex chromatographic separations, further lowers the cost of goods sold, making the final product highly competitive in the global market.
- Enhanced Supply Chain Reliability: Utilizing 3,4-difluoronitrobenzene as the primary feedstock leverages an existing, mature supply network, ensuring consistent availability and reducing lead time for high-purity pharmaceutical intermediates. Unlike routes dependent on custom-synthesized acids or chlorides that may have limited production capacity, this method allows for flexible scaling based on market demand. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by equipment failures or safety incidents, providing procurement managers with greater confidence in delivery timelines.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, utilizing solvents like toluene and NMP that are easily recovered and recycled in standard distillation units, aligning with modern green chemistry principles. The reduction in heavy metal waste, specifically by optimizing the copper usage and implementing efficient filtration steps for copper salt removal, significantly lowers the environmental burden and associated disposal fees. This compliance-friendly profile facilitates smoother regulatory approvals and supports the long-term sustainability goals of multinational chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent's experimental data and comparative analysis. These insights are intended to clarify the operational feasibility and quality benchmarks associated with this specific manufacturing route for decision-makers evaluating potential suppliers.
Q: What are the primary advantages of the diazotization route over traditional oxidation methods?
A: The diazotization route avoids the use of highly toxic and expensive reagents like oxalyl chloride and trifluoroacetic anhydride required in traditional oxidation pathways, significantly improving operational safety and reducing raw material costs.
Q: What purity levels can be achieved with this synthesis method?
A: The patented method consistently yields 2-fluoro-4-nitrobenzonitrile with a purity greater than 99.0% as determined by HPLC, meeting stringent requirements for pharmaceutical intermediate applications.
Q: Is the starting material 3,4-difluoronitrobenzene readily available for scale-up?
A: Yes, 3,4-difluoronitrobenzene is a commercially accessible and cost-effective raw material, which facilitates large-scale industrial production compared to harder-to-source precursors like 2-chloro-4-nitrobenzonitrile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-4-Nitrobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving therapies, and we have integrated this advanced synthesis technology into our production capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art HPLC and GC instrumentation to verify that every batch of 2-fluoro-4-nitrobenzonitrile meets the exacting standards required for tyrosine kinase inhibitor synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our specialized expertise. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this patented method can enhance your project's success and reliability.
